Lecture 7
... distort or polarize nearby anions and so it is said to have polarizing power. Apart from lithium, none of the cations have enough polarizing power to react with water molecules, so any reactions of their compounds in water are due to the anions, not the cations. The elements show typical metallic pr ...
... distort or polarize nearby anions and so it is said to have polarizing power. Apart from lithium, none of the cations have enough polarizing power to react with water molecules, so any reactions of their compounds in water are due to the anions, not the cations. The elements show typical metallic pr ...
Chemistry - Tumkur University
... stalagmometer. b)Study of the variation of surface tension of a detergent solution with concentration. (II) Viscosity measurement (use of organic solvents excluded). a)Determination of the relative and absolute viscosity of a liquid or dilute solution using an Ostwald’s viscometer. b) Study of the v ...
... stalagmometer. b)Study of the variation of surface tension of a detergent solution with concentration. (II) Viscosity measurement (use of organic solvents excluded). a)Determination of the relative and absolute viscosity of a liquid or dilute solution using an Ostwald’s viscometer. b) Study of the v ...
Spectroscopy in Organic Chemistry….
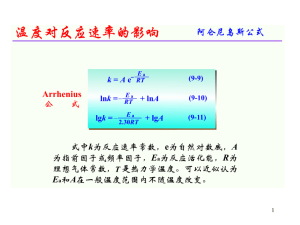
... If the Daddy gives a push every 3 seconds, the kid will go higher and energy will be absorbed. Every 2 seconds and the motion will get stalled and “interfered” with. Every 1.5 seconds and the energy will get absorbed but not as efficiently. The Daddy will get tired. This general principal applies in ...
... If the Daddy gives a push every 3 seconds, the kid will go higher and energy will be absorbed. Every 2 seconds and the motion will get stalled and “interfered” with. Every 1.5 seconds and the energy will get absorbed but not as efficiently. The Daddy will get tired. This general principal applies in ...
DEPARTMENT OF CHEMISTRY Course Book for M.Sc. in Chemistry
... Objective: The objective of the course is to cater the need of R&D division of various chemical and pharmaceutical industries, by imparting detailed knowledge of important name reactions, oxidizing-reducing agents, selective organometalic reagents used in organic synthesis and their industrial appli ...
... Objective: The objective of the course is to cater the need of R&D division of various chemical and pharmaceutical industries, by imparting detailed knowledge of important name reactions, oxidizing-reducing agents, selective organometalic reagents used in organic synthesis and their industrial appli ...
Chemistry - CBSE Academic
... at tertiary level. Therefore, there is a need to provide learners with sufficient conceptual background of Chemistry, which will make them competent to meet the challenges of academic and professional courses after the senior secondary stage. The new and updated curriculum is based on disciplinary a ...
... at tertiary level. Therefore, there is a need to provide learners with sufficient conceptual background of Chemistry, which will make them competent to meet the challenges of academic and professional courses after the senior secondary stage. The new and updated curriculum is based on disciplinary a ...
Chapters 1-4 Numbers and Measurements in Chemistry Units SI
... • Because carbon compounds can become quite large, organic compounds are described simply and unambiguously using line structures, where carbons and hydrogens are not explicitly shown. – Each corner or end of a line is a carbon. – Hydrogen y g atoms on carbon atoms are implied. p Carbon makes four b ...
... • Because carbon compounds can become quite large, organic compounds are described simply and unambiguously using line structures, where carbons and hydrogens are not explicitly shown. – Each corner or end of a line is a carbon. – Hydrogen y g atoms on carbon atoms are implied. p Carbon makes four b ...
01 Intro Chemistry
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
02Ch02chemistry2005
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
Chemistry I Syllabus 2011-2012
... reasoning The nuclear forces holding the nucleus together are many times larger than the electrostatic forces holding the atom together. When elements are listed by their atomic numbers, properties of the elements repeat over and over again. Nuclei can change through the process of fission, fusion, ...
... reasoning The nuclear forces holding the nucleus together are many times larger than the electrostatic forces holding the atom together. When elements are listed by their atomic numbers, properties of the elements repeat over and over again. Nuclei can change through the process of fission, fusion, ...