PDF - mockies – Mockiesgateacademy
... Towards the end of the eighteenth century, pioneering work by Antoine and Marie Lavoisier and by John Dalton on the chemistry of air and the atomic nature of matter paved the way for modern chemistry. During the nineteenth century chemists worked steadily towards an understanding of the relationship ...
... Towards the end of the eighteenth century, pioneering work by Antoine and Marie Lavoisier and by John Dalton on the chemistry of air and the atomic nature of matter paved the way for modern chemistry. During the nineteenth century chemists worked steadily towards an understanding of the relationship ...
chapter 4 - reactions in solution
... attractions of the ions for each other and the interactions of the ions for water molecules. If ion-dipole interactions are strong, compounds will not dissolve in water. Salt does not dissolve in nonpolar solvent, such as gasoline, because interactions between solvent molecules and ions are very w ...
... attractions of the ions for each other and the interactions of the ions for water molecules. If ion-dipole interactions are strong, compounds will not dissolve in water. Salt does not dissolve in nonpolar solvent, such as gasoline, because interactions between solvent molecules and ions are very w ...
7. A timeline of symbols and signs in chemistry
... Chemists use representations as part of their explanations. We use representations to communicate ideas even though the representations have limitations. When we draw a chemical formula, for example, we focus on some aspects of it. These might be just the elements in the formula, or the numbers of e ...
... Chemists use representations as part of their explanations. We use representations to communicate ideas even though the representations have limitations. When we draw a chemical formula, for example, we focus on some aspects of it. These might be just the elements in the formula, or the numbers of e ...
Chemistry 11 – Course Review
... Malleability, Ductility, Lustre, Viscosity and Diffusion. Review the Phases of Matter. ...
... Malleability, Ductility, Lustre, Viscosity and Diffusion. Review the Phases of Matter. ...
Sugar Amino Acids - The Krasavin research group
... A recognized strategy in drug discovery for generating new bioactive molecules takes into account the vast array of natural products and fundamental building blocks used by nature, like amino acids, sugars and nucleosides, to produce new chemical entities with multifunctional groups anchored on a si ...
... A recognized strategy in drug discovery for generating new bioactive molecules takes into account the vast array of natural products and fundamental building blocks used by nature, like amino acids, sugars and nucleosides, to produce new chemical entities with multifunctional groups anchored on a si ...
BSc (Hons) Chemistry (Optional Minor: Forensic Science)/MSc
... Chemical Thermodynamics I: kinetic theory of gases, 1st, 2nd & 3rd laws of thermodynamics, thermochemistry, ionic equilibria, acid/base reactions, gravimetry, redox reactions, solubility product. Quantum Chemistry I: wave-particle behaviour of the electron, Bohr’s theory of the H-atom, Schrödinger’s ...
... Chemical Thermodynamics I: kinetic theory of gases, 1st, 2nd & 3rd laws of thermodynamics, thermochemistry, ionic equilibria, acid/base reactions, gravimetry, redox reactions, solubility product. Quantum Chemistry I: wave-particle behaviour of the electron, Bohr’s theory of the H-atom, Schrödinger’s ...
P-BLOCK ELEMENTS
... carbon atoms. Each carbon form 3 sigma bonds. The most common is C-60. This ball shaped molecule has 60 vertices. It contains both single (143.5) and double (138.3) bonds. It has cage like structure like a soccer ball having hexagonal and pentagonal rings of carbon. It has 20-six membered ring and 1 ...
... carbon atoms. Each carbon form 3 sigma bonds. The most common is C-60. This ball shaped molecule has 60 vertices. It contains both single (143.5) and double (138.3) bonds. It has cage like structure like a soccer ball having hexagonal and pentagonal rings of carbon. It has 20-six membered ring and 1 ...
teaching and learning materials - UNESDOC
... R1 and R2 are hydrocarbon groups that may be the same or different. Esters have characteristic odours that differ substantially from the sometimes foul smelling alcohols and carboxylic acids from which they are derived. Many of the pleasant odours of fruits and flowers are due to the presence of low ...
... R1 and R2 are hydrocarbon groups that may be the same or different. Esters have characteristic odours that differ substantially from the sometimes foul smelling alcohols and carboxylic acids from which they are derived. Many of the pleasant odours of fruits and flowers are due to the presence of low ...
Chapter 04
... To determine the molecular, ionic and net ionic equations: 1) Write and balance the molecular equation, predicting the products by assuming that the cations trade anions. 2) Write the ionic equation by separating strong electrolytes into their constituent ions. 3) Write the net ionic equation by ide ...
... To determine the molecular, ionic and net ionic equations: 1) Write and balance the molecular equation, predicting the products by assuming that the cations trade anions. 2) Write the ionic equation by separating strong electrolytes into their constituent ions. 3) Write the net ionic equation by ide ...
SAMPLE QUESTION PAPER SIR.S.M.TAHIR CHEMISTRY Mob: 9557076999
... Miss Rita was asked to synthesized alcohol by acidic hydration of 1-butene. She was unaware of the fact that the vessel she used had some coating of a metal and in addition to alcohol (b.p. 373 K), compound X (b.p. 353 K) was also isolated. X form bisulphate compound as well as 2, 4-dinitrophenyl hy ...
... Miss Rita was asked to synthesized alcohol by acidic hydration of 1-butene. She was unaware of the fact that the vessel she used had some coating of a metal and in addition to alcohol (b.p. 373 K), compound X (b.p. 353 K) was also isolated. X form bisulphate compound as well as 2, 4-dinitrophenyl hy ...
Prep UK-intro.p65
... profitable discussions and enormous amount of work. Furthermore, I am indebted to my colleague lecturer Poul Thulstrup for his unremitting contribution to solving computer problems. I would also like to thank Martin Hancock for his assistance in proof-reading of the committees UK English. Funding f ...
... profitable discussions and enormous amount of work. Furthermore, I am indebted to my colleague lecturer Poul Thulstrup for his unremitting contribution to solving computer problems. I would also like to thank Martin Hancock for his assistance in proof-reading of the committees UK English. Funding f ...
SYLLABUS 5070 Cambridge O Level Chemistry
... educational experience for all students, whether or not they go on to study science beyond this level and, in particular, to enable them to acquire sufficient understanding and knowledge to 1.1 become confident citizens in a technological world, able to take or develop an informed interest in matter ...
... educational experience for all students, whether or not they go on to study science beyond this level and, in particular, to enable them to acquire sufficient understanding and knowledge to 1.1 become confident citizens in a technological world, able to take or develop an informed interest in matter ...
Wilhelm Ostwald, the Father of Physical Chemistry
... beginning. As time progresses, the small particles redeposit onto larger particles and grow in size. The small particles nucleate very easily and their formation is kinetically controlled. However, due to their large surface to volume ratio, they are not thermodynamically favourable. In order to red ...
... beginning. As time progresses, the small particles redeposit onto larger particles and grow in size. The small particles nucleate very easily and their formation is kinetically controlled. However, due to their large surface to volume ratio, they are not thermodynamically favourable. In order to red ...
Organic Chemistry
... Abstract: Disulfides are important compounds in biological and chemical processes which can be prepared by the oxidation of thiols. For this work, we have developed a new preparative method for the disulfide using inexpensive, recyclable, and relatively non-toxic polymer-supported (diacetoxyiodo)ben ...
... Abstract: Disulfides are important compounds in biological and chemical processes which can be prepared by the oxidation of thiols. For this work, we have developed a new preparative method for the disulfide using inexpensive, recyclable, and relatively non-toxic polymer-supported (diacetoxyiodo)ben ...
Metal disordering Cu(II) supramolecular polymers constructed from
... five metal sites, four are still disordered with the occupancy of 50%, and only Cu1 coordinated to the fixed pyridine molecules is fully occupied. Consequently, the whole structure also depends on the arrangement of the metal centers and several potential arrangements of the metal centers may exist, ...
... five metal sites, four are still disordered with the occupancy of 50%, and only Cu1 coordinated to the fixed pyridine molecules is fully occupied. Consequently, the whole structure also depends on the arrangement of the metal centers and several potential arrangements of the metal centers may exist, ...
On The Rocks - Técnico Lisboa
... Table 1- Major Classes of Copper Compounds, “On the Rocks” General method of synthesis from No of the Sample Compound, Classification from Box reference [1] or other 2 and Composition Dissolution of 56g/100 ml water; 1d CuSO4.5H2O, Hydrated ionic salt, Figs 1, 2 80 g/100 ml water if metal wires Any ...
... Table 1- Major Classes of Copper Compounds, “On the Rocks” General method of synthesis from No of the Sample Compound, Classification from Box reference [1] or other 2 and Composition Dissolution of 56g/100 ml water; 1d CuSO4.5H2O, Hydrated ionic salt, Figs 1, 2 80 g/100 ml water if metal wires Any ...
Molecules, Moles and Chemical Equations File
... that releases the same amount of energy. Careful examination of the progress of explosive chemical reactions reveals that they accelerate as they proceed. As a result, all of the available explosive is consumed in a very short period of time. As that happens, the energy from the explosion is also re ...
... that releases the same amount of energy. Careful examination of the progress of explosive chemical reactions reveals that they accelerate as they proceed. As a result, all of the available explosive is consumed in a very short period of time. As that happens, the energy from the explosion is also re ...
lecture ch1-3 chem161pikul
... – A = (number of protons)+(number of neutrons) = Z + N – For charge neutrality, number of electrons and protons must be equal Atomic Symbols = Summarize information about subatomic particles – Every isotope defined by two numbers Z and A Ex. What is the atomic symbol for helium? ...
... – A = (number of protons)+(number of neutrons) = Z + N – For charge neutrality, number of electrons and protons must be equal Atomic Symbols = Summarize information about subatomic particles – Every isotope defined by two numbers Z and A Ex. What is the atomic symbol for helium? ...
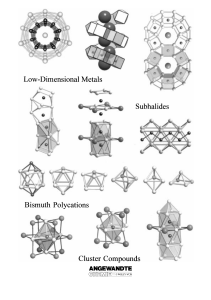
From the Metal to the Molecule
... oxidation state of the cation is smaller than would be expected from the (8 ÿ N) rule, have proved to be a rich source of unexpected structural and physical features. The extraordinary structural chemistry generally observed in subvalent compounds is a consequence of the low and often non-integer ox ...
... oxidation state of the cation is smaller than would be expected from the (8 ÿ N) rule, have proved to be a rich source of unexpected structural and physical features. The extraordinary structural chemistry generally observed in subvalent compounds is a consequence of the low and often non-integer ox ...
BASIC CONCEPTS OF CHEMISTRY
... bodies extracted from the space, is called the system . If no mass and heat transfer exists between the system and the surrounding environment, the system is called isolated. If this condition is not met, then the system is called open. If the system is only possible for the heat transfer , it is ca ...
... bodies extracted from the space, is called the system . If no mass and heat transfer exists between the system and the surrounding environment, the system is called isolated. If this condition is not met, then the system is called open. If the system is only possible for the heat transfer , it is ca ...
OCR AS Level Chemistry B (Salters) H033
... research to the concepts needed for the study of chemistry at AS Level. The specification is structured in a series of teaching modules that allow the concepts to unfold throughout the course. Each module is intended to be taught through a chemical ‘storyline’. The storylines address topics such as ...
... research to the concepts needed for the study of chemistry at AS Level. The specification is structured in a series of teaching modules that allow the concepts to unfold throughout the course. Each module is intended to be taught through a chemical ‘storyline’. The storylines address topics such as ...
major in Chemistry
... CHEMISTRY FOR THE SCIENCE MAJOR CHEM 11100. INTRODUCTORY CHEMISTRY (Biochemistry and Molecular Biology) Fundamental facts, concepts, and theories of chemistry and mathematical skills are emphasized. Topics include matter, measurements, calculations, elements, atomic theory, atomic mass, the mole, io ...
... CHEMISTRY FOR THE SCIENCE MAJOR CHEM 11100. INTRODUCTORY CHEMISTRY (Biochemistry and Molecular Biology) Fundamental facts, concepts, and theories of chemistry and mathematical skills are emphasized. Topics include matter, measurements, calculations, elements, atomic theory, atomic mass, the mole, io ...
guess paper class xii
... 21 How will you prepare the following compounds from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom. (i) Methyl benzoate (ii) m-nitrobenzoic acid (iii) Phenylacetic acid ...
... 21 How will you prepare the following compounds from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom. (i) Methyl benzoate (ii) m-nitrobenzoic acid (iii) Phenylacetic acid ...
IChO 2012
... (1875–1946) early in the 20 th century. That is, acids are electron-pair acceptors, whereas bases are electron-pair donors. There are thousands of molecules that can be classified as Lewis acids or bases, and hundreds of studies of the quantitative aspects of Lewis acid-base chemistry were carried o ...
... (1875–1946) early in the 20 th century. That is, acids are electron-pair acceptors, whereas bases are electron-pair donors. There are thousands of molecules that can be classified as Lewis acids or bases, and hundreds of studies of the quantitative aspects of Lewis acid-base chemistry were carried o ...
Module 2 Alcohols, halogenoalkanes and analysis
... o Alcoh and analysis Introduction Throughout the centuries, chemists have synthesised new substances and investigated their properties in the search for more useful materials. In the recent past, organic chemists have developed a broad range of original and exciting materials, such as pharmaceutical ...
... o Alcoh and analysis Introduction Throughout the centuries, chemists have synthesised new substances and investigated their properties in the search for more useful materials. In the recent past, organic chemists have developed a broad range of original and exciting materials, such as pharmaceutical ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.