12_chemistry_impq_CH13_amines_02
... This resonance accounts for the stability of the diazonium ion. Hence, diazonium salts of aromatic amines are more stable than those of aliphatic amines. (vii) Gabriel phthalimide synthesis is preferred for synthesising primary amines: Gabriel phthalimide synthesis results in the formation of 1° ami ...
... This resonance accounts for the stability of the diazonium ion. Hence, diazonium salts of aromatic amines are more stable than those of aliphatic amines. (vii) Gabriel phthalimide synthesis is preferred for synthesising primary amines: Gabriel phthalimide synthesis results in the formation of 1° ami ...
Higher Chemistry Resources Guide - Glow Blogs
... additional fourth column has been included which contains hyperlinks to useful resources. Please note: Practitioners are not required to use the resources listed – they are only included as helpful suggestions. Practitioners should also refer to the SQA website for the most up to date Course and Uni ...
... additional fourth column has been included which contains hyperlinks to useful resources. Please note: Practitioners are not required to use the resources listed – they are only included as helpful suggestions. Practitioners should also refer to the SQA website for the most up to date Course and Uni ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... Molecular compounds tend to be nonelectrolytes, except for strong acids (and weak acids & bases). Covalent Compounds: HCl, CO2, O2, P2O5, C6H14, C6H12O6, etc. Solubility Rules: 3. All salts containing the anions: NO3-, ClO3-, ClO4-, (C2H3O2-) are soluble. 4. All Cl-, Br-, and I- are soluble except f ...
... Molecular compounds tend to be nonelectrolytes, except for strong acids (and weak acids & bases). Covalent Compounds: HCl, CO2, O2, P2O5, C6H14, C6H12O6, etc. Solubility Rules: 3. All salts containing the anions: NO3-, ClO3-, ClO4-, (C2H3O2-) are soluble. 4. All Cl-, Br-, and I- are soluble except f ...
6 theoretical problems 2 practical problems
... Cement consists primarily of calcium silicates and calcium aluminates formed by heating and grinding of clay and limestone. In the later steps of cement production a small amount of gypsum, CaSO4 · 2 H2O, is added to improve subsequent hardening of the concrete. The use of elevated temperatures duri ...
... Cement consists primarily of calcium silicates and calcium aluminates formed by heating and grinding of clay and limestone. In the later steps of cement production a small amount of gypsum, CaSO4 · 2 H2O, is added to improve subsequent hardening of the concrete. The use of elevated temperatures duri ...
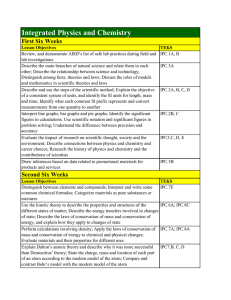
Integrated Physics and Chemistry
... atoms transfer their valence electrons to form ionic bonds, while other atoms share valence electrons to form covalent bonds; Differentiate between ionic, covalent, and metallic bonds; Compare the properties of substances with different types of bonds Name simple ionic and covalent compounds; Predic ...
... atoms transfer their valence electrons to form ionic bonds, while other atoms share valence electrons to form covalent bonds; Differentiate between ionic, covalent, and metallic bonds; Compare the properties of substances with different types of bonds Name simple ionic and covalent compounds; Predic ...
analytical chemistry - Львівський національний медичний
... form various acids and acids force increases with increasing of element oxidation degree. For oxidising-reducing properties the anions divide on anions-oxidisers with high oxidation degree (nitrate-anion), anions-reducers with lower oxidation degree (chlorides, bromides, iodides) and neutral anions, ...
... form various acids and acids force increases with increasing of element oxidation degree. For oxidising-reducing properties the anions divide on anions-oxidisers with high oxidation degree (nitrate-anion), anions-reducers with lower oxidation degree (chlorides, bromides, iodides) and neutral anions, ...
+ (aq)
... The d-block metals are comparatively small, and the metallic atoms are closely packed in the metallic lattice. Besides, both the 3d and 4s electrons of the d-block metals participate in metallic bondingAnswer by delocalizing into the electron sea. The strength of metallic bond in these metals is thu ...
... The d-block metals are comparatively small, and the metallic atoms are closely packed in the metallic lattice. Besides, both the 3d and 4s electrons of the d-block metals participate in metallic bondingAnswer by delocalizing into the electron sea. The strength of metallic bond in these metals is thu ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... Why might these be important? Give examples. • Aqueous reactions cannot take place without water. What do you already know about water that will help us understand aqueous reactions? Aqueous Reactions ...
... Why might these be important? Give examples. • Aqueous reactions cannot take place without water. What do you already know about water that will help us understand aqueous reactions? Aqueous Reactions ...
Synthesis and Characterization of Amorphous and Hybrid Materials
... processes as well. Those problems can be overcome by using local drug delivery methods to confine pharmaceuticals such as antibiotics, anti-inflammatory, anti-carcinogens, etc. (Arcos et. al., 2001; Ragel & Vallet-Regì, 2000; Vallet-Regì et al., 2000). The possibility of introducing certain drugs in ...
... processes as well. Those problems can be overcome by using local drug delivery methods to confine pharmaceuticals such as antibiotics, anti-inflammatory, anti-carcinogens, etc. (Arcos et. al., 2001; Ragel & Vallet-Regì, 2000; Vallet-Regì et al., 2000). The possibility of introducing certain drugs in ...
Synthesis and Structural Studies of Calcium and Magnesium
... calcium solids lie in the large metal diameters, the absence of energetically available dorbital to direct metal geometry, as well as the overall weakness of the metal-ligand bonds. A significant part of this project was concerned with the development of suitable reaction conditions to obtain X-ray ...
... calcium solids lie in the large metal diameters, the absence of energetically available dorbital to direct metal geometry, as well as the overall weakness of the metal-ligand bonds. A significant part of this project was concerned with the development of suitable reaction conditions to obtain X-ray ...
Chapter 9: Non-aqueous media
... (e.g. MeCO2 H, H2 SO4 ), ionization of bases is facilitated. Most acids are relatively weak under these conditions, and some even ionize as bases. We noted above that HCl, when dissolved in acetic acid, behaves as a weak acid. Hydrogen bromide and hydrogen iodide behave similarly but the extent of i ...
... (e.g. MeCO2 H, H2 SO4 ), ionization of bases is facilitated. Most acids are relatively weak under these conditions, and some even ionize as bases. We noted above that HCl, when dissolved in acetic acid, behaves as a weak acid. Hydrogen bromide and hydrogen iodide behave similarly but the extent of i ...
Chemical Reactions
... • The problem with word equations is they do not actually show the number of atoms or molecules of each substance… formulas would have to be written out for this to happen. (Absent? We looked at examples of these in class) ...
... • The problem with word equations is they do not actually show the number of atoms or molecules of each substance… formulas would have to be written out for this to happen. (Absent? We looked at examples of these in class) ...
fast pyrolysis characteristics of sugarcane bagasse hemicellulose
... CH4. It was observed that the yield of CO2 and CO was higher at lower temperatures, such as 550 °C, as well as that of H2 and CH4 at higher temperatures. The liquid products mainly contained furfural, cyclopentene-1 (2-methyl-2-cyclopentene-1, 2-cyclopentene-1, 3-methyl-2-cyclopentene), alcohols (1- ...
... CH4. It was observed that the yield of CO2 and CO was higher at lower temperatures, such as 550 °C, as well as that of H2 and CH4 at higher temperatures. The liquid products mainly contained furfural, cyclopentene-1 (2-methyl-2-cyclopentene-1, 2-cyclopentene-1, 3-methyl-2-cyclopentene), alcohols (1- ...
Chapter 22 - 2012 Book Archive
... Group 13 is the first group to span the dividing line between metals and nonmetals, so its chemistry is more diverse than that of groups 1 and 2, which include only metallic elements. Except for the lightest element (boron), the group 13 elements are all relatively electropositive; that is, they ten ...
... Group 13 is the first group to span the dividing line between metals and nonmetals, so its chemistry is more diverse than that of groups 1 and 2, which include only metallic elements. Except for the lightest element (boron), the group 13 elements are all relatively electropositive; that is, they ten ...
ANSWERS Problem Set 5a – Chemical Reactions
... Subscripts are underlined, Coefficients are boxed 3 H2SO4+ 2 Al Al2(SO4)3 + 3 H2 11) Write the word equation for the following balanced reactions: a. 2 Mg (s) + O2 (g) 2 MgO (s) solid magnesium reacts with oxygen gas to produce solid magnesium oxide in a synthesis reaction. b. HCl (aq) + NaOH (a ...
... Subscripts are underlined, Coefficients are boxed 3 H2SO4+ 2 Al Al2(SO4)3 + 3 H2 11) Write the word equation for the following balanced reactions: a. 2 Mg (s) + O2 (g) 2 MgO (s) solid magnesium reacts with oxygen gas to produce solid magnesium oxide in a synthesis reaction. b. HCl (aq) + NaOH (a ...
Brilliant Preparatory Section, Sitamarhi
... in the ratio 2:1, so that the reactants are completely consumed during the reaction. But atoms and molecules are so small in size that is not possible to count them individually. In order to overcome these difficulties, the concept of mole was introduced. According to this concept number of particle ...
... in the ratio 2:1, so that the reactants are completely consumed during the reaction. But atoms and molecules are so small in size that is not possible to count them individually. In order to overcome these difficulties, the concept of mole was introduced. According to this concept number of particle ...
aq - Haverford Alchemy
... Why might these be important? Give examples. • Aqueous reactions cannot take place without water. What do you already know about water that will help us understand aqueous reactions? Aqueous Reactions ...
... Why might these be important? Give examples. • Aqueous reactions cannot take place without water. What do you already know about water that will help us understand aqueous reactions? Aqueous Reactions ...
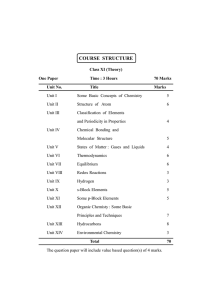
COURSE STRUCTURE
... Law of conservation of mass : ‘Mass can neither be created nor destroyed.’ In all physical and chemical changes, the total mass of reactants is equal to that of products. Law of constant composition : A chemical compound is always found to be made of same elements combined together in the same fixed ...
... Law of conservation of mass : ‘Mass can neither be created nor destroyed.’ In all physical and chemical changes, the total mass of reactants is equal to that of products. Law of constant composition : A chemical compound is always found to be made of same elements combined together in the same fixed ...
Elements Of Physical Chemistry 4th Edition Laidler
... by keith j laidler - physical chemistry solutions manual has 26 ratings and 3 reviews by keith j laidler fourth edition, download physical chemistry 4th edition laidler meiser and - physical chemistry 4th edition laidler meiser and 4 help pdf howlin rain the matrix the matrix the elements of user ex ...
... by keith j laidler - physical chemistry solutions manual has 26 ratings and 3 reviews by keith j laidler fourth edition, download physical chemistry 4th edition laidler meiser and - physical chemistry 4th edition laidler meiser and 4 help pdf howlin rain the matrix the matrix the elements of user ex ...
7.1 Describing Reactions
... If you examine this equation carefully, you will notice that the number of atoms on the left side does not equal the number of atoms on the right. The equation is not balanced. In order to show that mass is conserved during a reaction, a chemical equation must be balanced. You can balance a chemical ...
... If you examine this equation carefully, you will notice that the number of atoms on the left side does not equal the number of atoms on the right. The equation is not balanced. In order to show that mass is conserved during a reaction, a chemical equation must be balanced. You can balance a chemical ...
It`s Easy Being a Green Chemist
... way products are made. It is a science that aims to reduce or eliminate the use and/or generation of hazardous substances in the design phase of materials development. Considered a subset of sustainable chemistry, Green Chemistry is defined as: The utilization of a set of principles that reduces or ...
... way products are made. It is a science that aims to reduce or eliminate the use and/or generation of hazardous substances in the design phase of materials development. Considered a subset of sustainable chemistry, Green Chemistry is defined as: The utilization of a set of principles that reduces or ...
33 POLYMERS I OPTIONAL MODULE - 2
... 2. Synthetic Polymers : The polymers which are prepared in the laboratories are called synthetic polymers. These are also called man-made polymers. Polyethene, PVC, nylon, Teflon, bakelite, terylene, synthetic rubber, etc. are common examples. (b) Classification of Polymers on the Basis of structure ...
... 2. Synthetic Polymers : The polymers which are prepared in the laboratories are called synthetic polymers. These are also called man-made polymers. Polyethene, PVC, nylon, Teflon, bakelite, terylene, synthetic rubber, etc. are common examples. (b) Classification of Polymers on the Basis of structure ...
2014 Syllabus - Cambridge International Examinations
... In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Advanced Practical Skills 1 and Advanced Practical Skills 2. In other sessions only Advanced Practical Skills 1 will be available. These papers will be equivalent and each candidate wil ...
... In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Advanced Practical Skills 1 and Advanced Practical Skills 2. In other sessions only Advanced Practical Skills 1 will be available. These papers will be equivalent and each candidate wil ...
Pirogov National Medical Univercity of Vinnitsa
... 2. Key questions of the theme: In the chemical practice it is of great importance degree of purification of substances as the work with contaminated reagents may lead to error in results. Purity of substances used as medicines, determined by the State Pharmacopoeia of Ukraine and, therefore, pharmac ...
... 2. Key questions of the theme: In the chemical practice it is of great importance degree of purification of substances as the work with contaminated reagents may lead to error in results. Purity of substances used as medicines, determined by the State Pharmacopoeia of Ukraine and, therefore, pharmac ...
Subject Area Standard Area Organizing Category Grade Level
... CHEM.B.2.2.1: Utilize mathematical relationships to predict changes in the number of particles, the temperature, the pressure, and the volume in a gaseous system (i.e., Boyle’s law, Charles’s law, Dalton’s law of partial pressures, the combined gas law, and the ideal gas law). ...
... CHEM.B.2.2.1: Utilize mathematical relationships to predict changes in the number of particles, the temperature, the pressure, and the volume in a gaseous system (i.e., Boyle’s law, Charles’s law, Dalton’s law of partial pressures, the combined gas law, and the ideal gas law). ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.