Worked solutions to the problems
... your students need to show in Melbourne. We have tried to highlight the procedures in each exercise that need some particular caution, even for students of Olympiad level but our warnings cannot be comprehensive - your students will still need your careful supervision. We have also not included spec ...
... your students need to show in Melbourne. We have tried to highlight the procedures in each exercise that need some particular caution, even for students of Olympiad level but our warnings cannot be comprehensive - your students will still need your careful supervision. We have also not included spec ...
Sustainable Oxidation Catalysis for Synthesis
... * corresponding author: [email protected] Abstract Conventional wastewater treatment plants are not effective when wastewaters have to be treated due to the high COD load and the presence of recalcitrant compounds. An alternative to conventional water treatments are Advanced Oxidation Processes (A ...
... * corresponding author: [email protected] Abstract Conventional wastewater treatment plants are not effective when wastewaters have to be treated due to the high COD load and the presence of recalcitrant compounds. An alternative to conventional water treatments are Advanced Oxidation Processes (A ...
Unit 8: Reactions
... iv. Revise (remember, to use PENCIL) the coefficients where necessary. *Note: A coefficient is the number in front; a subscript is behind! When you write 2 Cl, that states there are TWO atoms of chlorine. When you write Cl2, that states there is ONE molecule of diatomic (2 atoms) chlorine. Diato ...
... iv. Revise (remember, to use PENCIL) the coefficients where necessary. *Note: A coefficient is the number in front; a subscript is behind! When you write 2 Cl, that states there are TWO atoms of chlorine. When you write Cl2, that states there is ONE molecule of diatomic (2 atoms) chlorine. Diato ...
Chemistry - BYU
... This course involves a study of structure, physical and chemical behavior, and bond theory as applied to inorganic chemistry. Using concepts, models, and experimental data, a variety of inorganic systems will be analyzed in a systematic and detailed fashion. Underlying trends in both the elements an ...
... This course involves a study of structure, physical and chemical behavior, and bond theory as applied to inorganic chemistry. Using concepts, models, and experimental data, a variety of inorganic systems will be analyzed in a systematic and detailed fashion. Underlying trends in both the elements an ...
PDF - Chemistry - University of Canterbury
... related, but in a somewhat different category, are the biological sciences, such as biochemistry, biotechnology, ecology and genetics. There is no sharp distinction between the physical and biological sciences, indeed they often overlap with each other – for example, chemical reactions are the basis ...
... related, but in a somewhat different category, are the biological sciences, such as biochemistry, biotechnology, ecology and genetics. There is no sharp distinction between the physical and biological sciences, indeed they often overlap with each other – for example, chemical reactions are the basis ...
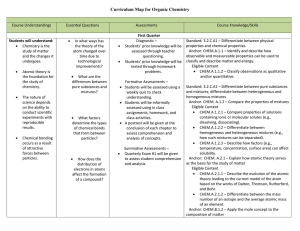
Organic Chemistry Curriculum Map - Belle Vernon Area School District
... CHEM.A.2.1.1 – Describe the evolution of the atomic theory leading to the current model of the atom based on the works of Dalton, Thomson, Rutherford, and Bohr. CHEM.A.2.1.2 – Differentiate between the mass number of an isotope and the average atomic mass of an element. Anchor: CHEM.B.1.2 – Appl ...
... CHEM.A.2.1.1 – Describe the evolution of the atomic theory leading to the current model of the atom based on the works of Dalton, Thomson, Rutherford, and Bohr. CHEM.A.2.1.2 – Differentiate between the mass number of an isotope and the average atomic mass of an element. Anchor: CHEM.B.1.2 – Appl ...
Syllabus Cambridge IGCSE Chemistry (US) Syllabus Code 0439 For examination in 2013
... Cambridge ICE is the group award of the IGCSE. It requires the study of subjects drawn from the five different IGCSE subject groups. It gives Centers the opportunity to benefit from offering a broad and balanced curriculum by recognizing the achievements of students who pass examinations in at least ...
... Cambridge ICE is the group award of the IGCSE. It requires the study of subjects drawn from the five different IGCSE subject groups. It gives Centers the opportunity to benefit from offering a broad and balanced curriculum by recognizing the achievements of students who pass examinations in at least ...
F:\Users\Steven\Documents\Chemistry\CHEM120\Problem Set
... When I went to write this problem I looked at the periodic table and saw that Rubidium had a mass of 85.467. Since the mass of all isotopes are even (or nearly so) and this average was uneven I knew immediately that rubidium had to have two major isotopes. When I looked up the isotopes sure enough t ...
... When I went to write this problem I looked at the periodic table and saw that Rubidium had a mass of 85.467. Since the mass of all isotopes are even (or nearly so) and this average was uneven I knew immediately that rubidium had to have two major isotopes. When I looked up the isotopes sure enough t ...
Low-Temperature Alkaline pH Hydrolysis of Oxygen-Free
... works was 10–25 wt % (Neish et al., 2009; Poch et al., 2012). However, a recent study shows that the fraction of ammonia in the subsurface ocean could be lower, around 2–3 wt % if taking into account the actual mass of nitrogen in Titan’s atmosphere (Tobie et al., 2012). Additionally, studies have p ...
... works was 10–25 wt % (Neish et al., 2009; Poch et al., 2012). However, a recent study shows that the fraction of ammonia in the subsurface ocean could be lower, around 2–3 wt % if taking into account the actual mass of nitrogen in Titan’s atmosphere (Tobie et al., 2012). Additionally, studies have p ...
Chemical Reactions
... • Law of Conservation of Mass • Matter cannot be created nor destroyed • Just like with math, both sides of the YIELDS sign must be equal! • So the same number of atoms must be on both sides of the equation ...
... • Law of Conservation of Mass • Matter cannot be created nor destroyed • Just like with math, both sides of the YIELDS sign must be equal! • So the same number of atoms must be on both sides of the equation ...
GCSE Chemistry Specification Specification for exams from 2014 2014
... GCSE Chemistry for certification June 2014 onwards (version 1.0) ...
... GCSE Chemistry for certification June 2014 onwards (version 1.0) ...
Stoichiometry - coercingmolecules
... e. How many formula units of sodium ascorbate are present? f. How many atoms of Na are present? ...
... e. How many formula units of sodium ascorbate are present? f. How many atoms of Na are present? ...
Naphtyl-imidazo-anthraquinones as novel colorimetric
... of great relevance for environment and human health care [8]. Due to the toxicity of the cyanide anion, highly harmful to the environment and human health, there is an interest to develop new and more selective chemosensors for this analyte. Cyanide compounds are largely applied in several areas suc ...
... of great relevance for environment and human health care [8]. Due to the toxicity of the cyanide anion, highly harmful to the environment and human health, there is an interest to develop new and more selective chemosensors for this analyte. Cyanide compounds are largely applied in several areas suc ...
Chapter
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
Chapter
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
Copper Chemistry
... Copper for Good Health Copper is one of many trace elements required for good health. It is part of the prosthetic groups of many proteins and enzymes and thus is essential to their proper function. Since the body can not synthesize copper it must be taken in the diet. Nuts, seeds, cereals, meat (e. ...
... Copper for Good Health Copper is one of many trace elements required for good health. It is part of the prosthetic groups of many proteins and enzymes and thus is essential to their proper function. Since the body can not synthesize copper it must be taken in the diet. Nuts, seeds, cereals, meat (e. ...
Synthetic Polymers
... reactions, terminations are relatively rare because the concentration of radicals is extremely low. Therefore, the probability of one radical encountering another is also quite low. This means that a typical polymer molecule is composed of thousands of monomers. For a hydrocarbon polymer such as pol ...
... reactions, terminations are relatively rare because the concentration of radicals is extremely low. Therefore, the probability of one radical encountering another is also quite low. This means that a typical polymer molecule is composed of thousands of monomers. For a hydrocarbon polymer such as pol ...
Holt Modern Chemistry Workbook
... because the sum of the partial charges on either end of the bond is zero. A molecular compound is any chemical compound whose simplest units are molecules. In other words, a single molecule of any molecular compound is an individual unit that is capable of existing on its own. A molecule may contain ...
... because the sum of the partial charges on either end of the bond is zero. A molecular compound is any chemical compound whose simplest units are molecules. In other words, a single molecule of any molecular compound is an individual unit that is capable of existing on its own. A molecule may contain ...
Some basic concepts of chemistry
... Chemistry plays a very important role in our everyday lives. Our daily needs of food, clothing, shelter, potable water, medicines, etc., are in one or the other manner connected with chemical compounds, processes and principles. There is no aspect of life that is not associated with chemistry. In fa ...
... Chemistry plays a very important role in our everyday lives. Our daily needs of food, clothing, shelter, potable water, medicines, etc., are in one or the other manner connected with chemical compounds, processes and principles. There is no aspect of life that is not associated with chemistry. In fa ...
w_4-3 Chemistry of Nitrogen Compounds
... Nitrogen–containing impurities (e.g., ammonia, amino acids, creatinine, uric acid, etc.) introduced into swimming pools by bathers react with free available chlorine to form combined chlorine compounds. Because these compounds do not readily hydrolyze to hypochlorous acid, they are poor disinfectant ...
... Nitrogen–containing impurities (e.g., ammonia, amino acids, creatinine, uric acid, etc.) introduced into swimming pools by bathers react with free available chlorine to form combined chlorine compounds. Because these compounds do not readily hydrolyze to hypochlorous acid, they are poor disinfectant ...
幻灯片 1
... periods and vertically in groups according to their chemical similarity - was developed in an empirical way in the 19th century. A more rigorous foundation came, first with the use of spectroscopy to determine atomic number and, second with the development of the quantum theory of atomic structure. ...
... periods and vertically in groups according to their chemical similarity - was developed in an empirical way in the 19th century. A more rigorous foundation came, first with the use of spectroscopy to determine atomic number and, second with the development of the quantum theory of atomic structure. ...
Problem 28. TUNNELING IN CHEMISTRY
... with the target containing californium-249 nuclei three cascades of -decay were detected, that started from the 118th element with the mass number 294. 2. Write the balanced equations of the nuclear reactions of: i) the synthesis and ii) the decay of the 118th element. ...
... with the target containing californium-249 nuclei three cascades of -decay were detected, that started from the 118th element with the mass number 294. 2. Write the balanced equations of the nuclear reactions of: i) the synthesis and ii) the decay of the 118th element. ...
Chemistry – 5071
... on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need of students to develop skills that will be of long term value in an increasing technological world rather than focusing on large quantities of actual material which m ...
... on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need of students to develop skills that will be of long term value in an increasing technological world rather than focusing on large quantities of actual material which m ...
REVIEWS Environmental remediation by photocatalysis R. Vinu AND Giridhar Madras
... used in conjunction with UV radiation and/or photocatalysis in order to accelerate the rate of degradation of the pollutants. Recently, ultrasound (US), microwave radiation and electrolysis are being coupled to the above AOPs in order to further enhance the decomposition of the pollutants. A common ...
... used in conjunction with UV radiation and/or photocatalysis in order to accelerate the rate of degradation of the pollutants. Recently, ultrasound (US), microwave radiation and electrolysis are being coupled to the above AOPs in order to further enhance the decomposition of the pollutants. A common ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.