Syllabus Cambridge International A & AS Level Chemistry Syllabus code 9701
... 1.3 Cambridge Advanced International Certificate of Education (AICE) Cambridge AICE is the group award of Cambridge International Advanced Supplementary Level and Advanced Level (AS Level and A Level). Cambridge AICE involves the selection of subjects from three curriculum areas – Mathematics and Sc ...
... 1.3 Cambridge Advanced International Certificate of Education (AICE) Cambridge AICE is the group award of Cambridge International Advanced Supplementary Level and Advanced Level (AS Level and A Level). Cambridge AICE involves the selection of subjects from three curriculum areas – Mathematics and Sc ...
SyllAbuS - Cambridge International Examinations
... think about how to approach each syllabus topic in order to encourage learners to make links between topics and develop a deep overall understanding of the subject. The teaching support package gives teachers guidance on integrating the key concepts into their teaching. See page 8 for more informati ...
... think about how to approach each syllabus topic in order to encourage learners to make links between topics and develop a deep overall understanding of the subject. The teaching support package gives teachers guidance on integrating the key concepts into their teaching. See page 8 for more informati ...
Chapter - Imperial Valley College
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
FREE Sample Here
... DNA and RNA differ in that RNA has deoxyribose and DNA has ribose. RNA is double-stranded and DNA is single-stranded. DNA holds genetic information and RNA uses that information to synthesize protein. RNA is found only in the nucleus and DNA is found only in the cytoplasm. ...
... DNA and RNA differ in that RNA has deoxyribose and DNA has ribose. RNA is double-stranded and DNA is single-stranded. DNA holds genetic information and RNA uses that information to synthesize protein. RNA is found only in the nucleus and DNA is found only in the cytoplasm. ...
Synthetic Organic Chemistry - Name
... Organolithium compounds are the most versatile reagents in all fields of chemistry . Thus some of the important applications of these compounds are as follows: 1. Organolithium compounds are highly reactive nucleophiles and strong bases due to the presence of strongly polarized Li-C bond. 2. Organol ...
... Organolithium compounds are the most versatile reagents in all fields of chemistry . Thus some of the important applications of these compounds are as follows: 1. Organolithium compounds are highly reactive nucleophiles and strong bases due to the presence of strongly polarized Li-C bond. 2. Organol ...
An Introduction to Chemical Science
... allowed to take the book from the laboratory; nor can he use any other book on Chemistry while experimenting. By this means he learns to make his own observations and inferences. For the benefit of the science and the added interest in the study, it is earnestly recommended that teachers encourage p ...
... allowed to take the book from the laboratory; nor can he use any other book on Chemistry while experimenting. By this means he learns to make his own observations and inferences. For the benefit of the science and the added interest in the study, it is earnestly recommended that teachers encourage p ...
The Role of Medicinal Chemistry in Canadian Pharmacy
... 2735 B.C. – Babylonian, Chinese, Indian cultures 400 B.C. – Greek culture (ancient apothecary) ...
... 2735 B.C. – Babylonian, Chinese, Indian cultures 400 B.C. – Greek culture (ancient apothecary) ...
class XI CHEMISTRY - Kendriya Vidyalaya No.1 Ichhanath Surat
... An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements ar ...
... An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements ar ...
class XI CHEMISTRY - Kendriya Vidyalaya No.1 Harni Road
... An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements ar ...
... An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements ar ...
- Kendriya Vidyalaya No. 2 Raipur
... An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements ar ...
... An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements ar ...
FREE Sample Here - We can offer most test bank and
... http://gettestbank.eu/Test-Bank-for-Chemistry-in-Focus-A-Molecular-View-of-Our-World,-5th-Editio n--Tr 48. A sample of heptane always contains 84% carbon and 16% hydrogen. Which of these best explains this phenomena? a. Law of Constant Composition b. Law of Conservation of Mass c. Dalton's Atomic Th ...
... http://gettestbank.eu/Test-Bank-for-Chemistry-in-Focus-A-Molecular-View-of-Our-World,-5th-Editio n--Tr 48. A sample of heptane always contains 84% carbon and 16% hydrogen. Which of these best explains this phenomena? a. Law of Constant Composition b. Law of Conservation of Mass c. Dalton's Atomic Th ...
Recycling and Chemical Mathematics
... release oxygen gas in the process. The animal life of Biosphere 2, through the process of breathing (respiration), takes in atmospheric oxygen and releases carbon dioxide. If everything could be arranged to come out even, a stable atmosphere with desirable levels of oxygen and carbon dioxide would b ...
... release oxygen gas in the process. The animal life of Biosphere 2, through the process of breathing (respiration), takes in atmospheric oxygen and releases carbon dioxide. If everything could be arranged to come out even, a stable atmosphere with desirable levels of oxygen and carbon dioxide would b ...
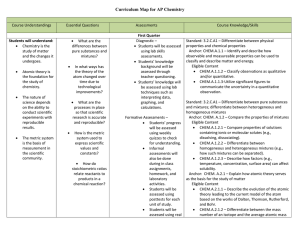
AP Chemistry Curriculum Map - Belle Vernon Area School District
... CHEM.A.2.2.3 – Explain the relationship between the electron configurations and the atomic structure of a given atom or ion (e.g., energy levels and/or orbitals with electrons, distribution of electrons in orbitals, shapes of orbitals). ...
... CHEM.A.2.2.3 – Explain the relationship between the electron configurations and the atomic structure of a given atom or ion (e.g., energy levels and/or orbitals with electrons, distribution of electrons in orbitals, shapes of orbitals). ...
Study Material - Class- XI- Chemistry
... An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements ar ...
... An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements ar ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... substance dissolves in water and the solvent pulls the individual ions from the crystal and solvates them. Recall: Ionic substances contain a metal & nonmetal or polyatomic ion. Examples: NH4Cl or KBr Aqueous Reactions © 2009, Prentice-Hall, Inc. ...
... substance dissolves in water and the solvent pulls the individual ions from the crystal and solvates them. Recall: Ionic substances contain a metal & nonmetal or polyatomic ion. Examples: NH4Cl or KBr Aqueous Reactions © 2009, Prentice-Hall, Inc. ...
Chemistry XII - Kendriya Vidyalaya IIM,Lucknow
... Rate Law or rate equation : It is the expression which relates the rate of reaction with concentration of the reactants. The constant of proportionality ‘k’ is known as rate constant. R = K [ A ]p [ B ]q Where , p and q are not stoichiometric coefficient but they are order of the reaction ...
... Rate Law or rate equation : It is the expression which relates the rate of reaction with concentration of the reactants. The constant of proportionality ‘k’ is known as rate constant. R = K [ A ]p [ B ]q Where , p and q are not stoichiometric coefficient but they are order of the reaction ...
Chapter 2 Matter and Components F11 110
... is found in a fixed amount in nature, and rarely are these amounts equal among the given isotopes of an element we must have a way to take this into account when talking about a naturally occurring element; enter Average Mass: ...
... is found in a fixed amount in nature, and rarely are these amounts equal among the given isotopes of an element we must have a way to take this into account when talking about a naturally occurring element; enter Average Mass: ...
Chapter 2 Matter and Components F11 110pt
... 1. Some compounds have been known and used for so long that their trivial (or common names) have become accepted by the IUPAC as official: ...
... 1. Some compounds have been known and used for so long that their trivial (or common names) have become accepted by the IUPAC as official: ...
Alkaloids
... 1- Direct titration of the alcoholic solution of the alkaloidal residue with standard acid, or 2- Back titration by dissolving the residue in a known amount of standard acid and back titration of residual acid against standard alkali. Non-aqueous titration: This method is suitable for determination ...
... 1- Direct titration of the alcoholic solution of the alkaloidal residue with standard acid, or 2- Back titration by dissolving the residue in a known amount of standard acid and back titration of residual acid against standard alkali. Non-aqueous titration: This method is suitable for determination ...
Novel Class of Heterometallic Cubane and Boride Clusters
... bound to molybdenum and boron. Cluster 5 represents an unprecedented metal-rich metallaborane cluster with a cubane core. The dimolybdaheteroborane 2 was found to be very reactive toward metal carbonyl compounds, and as a result, mild pyrolysis of 2 with [Fe2(CO)9] yielded distorted cubane cluster [ ...
... bound to molybdenum and boron. Cluster 5 represents an unprecedented metal-rich metallaborane cluster with a cubane core. The dimolybdaheteroborane 2 was found to be very reactive toward metal carbonyl compounds, and as a result, mild pyrolysis of 2 with [Fe2(CO)9] yielded distorted cubane cluster [ ...
Chemistry 30 June 2001 Grade 12 Diploma Examination
... Use the following information to answer the next question. Cold packs are commonly used by athletes to reduce swelling caused by injury. The packs consist of two plastic pouches: an inner pouch that contains a chemical and an outer pouch that contains water. When the inner pouch is broken, the chem ...
... Use the following information to answer the next question. Cold packs are commonly used by athletes to reduce swelling caused by injury. The packs consist of two plastic pouches: an inner pouch that contains a chemical and an outer pouch that contains water. When the inner pouch is broken, the chem ...
Boronic acids facilitate rapid oxime condensations at neutral pH
... with only 10% of pinacol ester oxime 2f observed; aer ninety minutes only 2a was present. The ketoximine pinacol product (entry 9), on the other hand, was more stable and comprised 97% of the product aer the rst injection (the remainder being 2e); aer thirty minutes 2g had also completely hydrol ...
... with only 10% of pinacol ester oxime 2f observed; aer ninety minutes only 2a was present. The ketoximine pinacol product (entry 9), on the other hand, was more stable and comprised 97% of the product aer the rst injection (the remainder being 2e); aer thirty minutes 2g had also completely hydrol ...
Fluorinated Butatrienes - diss.fu-berlin.de
... The present study provides deeper insight into the chemistry of fluorinated butatrienes both experimentally and theoretically. The four step synthesis of tetrafluorobutatriene starting from commercially available 1,1difluoroethylene could be successfully improved to an overall yield of up to 42% (fr ...
... The present study provides deeper insight into the chemistry of fluorinated butatrienes both experimentally and theoretically. The four step synthesis of tetrafluorobutatriene starting from commercially available 1,1difluoroethylene could be successfully improved to an overall yield of up to 42% (fr ...
Russell, M.J. and Hall, A.J. 2006.
... fides, mackinawite and greigite—sulfides which accommodate that effective and common catalytic metal, nickel. The primary carbon dioxide, which composed a proportion of the atmosphere/ ocean system (the volatisphere), is fixed by reaction with activated hydrothermal H2 emanating from the highly redu ...
... fides, mackinawite and greigite—sulfides which accommodate that effective and common catalytic metal, nickel. The primary carbon dioxide, which composed a proportion of the atmosphere/ ocean system (the volatisphere), is fixed by reaction with activated hydrothermal H2 emanating from the highly redu ...
Higher Chemistry Resources Guide - Glow Blogs
... A potential energy diagram can be used to show the energy pathway for a reaction. The enthalpy change is the energy difference between products and reactants. It can be calculated from a potential energy diagram. The enthalpy change has a negative value for exothermic reactions and a positive value ...
... A potential energy diagram can be used to show the energy pathway for a reaction. The enthalpy change is the energy difference between products and reactants. It can be calculated from a potential energy diagram. The enthalpy change has a negative value for exothermic reactions and a positive value ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.