No Slide Title
... properties of an element based on where it is in the periodic table. • Elements are listed in order of number of protons, because the periodic law states that when elements are arranged this way, similarities in their properties will occur in a regular pattern. ...
... properties of an element based on where it is in the periodic table. • Elements are listed in order of number of protons, because the periodic law states that when elements are arranged this way, similarities in their properties will occur in a regular pattern. ...
Chemistry Curriculum Map - Belle Vernon Area School District
... Diagnostic – Pretest on “The Periodic Table” (Given before Chapter 4), Pretest on “Bond Types” (Given before Chapter 5), Pretest on “Common Compounds” and Naming (Given before Chapter 5). Each posttest given after end of chapter. Benchmark – Study Island: Systems, Models, and Patterns (given During ...
... Diagnostic – Pretest on “The Periodic Table” (Given before Chapter 4), Pretest on “Bond Types” (Given before Chapter 5), Pretest on “Common Compounds” and Naming (Given before Chapter 5). Each posttest given after end of chapter. Benchmark – Study Island: Systems, Models, and Patterns (given During ...
Slide 1
... • Metals form cations • For each positive charge, the ion has 1 less electron than the neutral atom – Na atom = 11 p+ and 11 e-, Na+ ion = 11 p+ and 10 e– Ca atom = 20 p+ and 20 e-, Ca2+ ion = 20 p+ and 18 e- ...
... • Metals form cations • For each positive charge, the ion has 1 less electron than the neutral atom – Na atom = 11 p+ and 11 e-, Na+ ion = 11 p+ and 10 e– Ca atom = 20 p+ and 20 e-, Ca2+ ion = 20 p+ and 18 e- ...
Chapter 4
... • The number of protons in the nucleus of an atom is called the atomic number. • All atoms of any given element have the same atomic number. An element’s atomic number sets the atoms of that element apart from the atoms of all other elements. • Elements on the periodic table are ordered according to ...
... • The number of protons in the nucleus of an atom is called the atomic number. • All atoms of any given element have the same atomic number. An element’s atomic number sets the atoms of that element apart from the atoms of all other elements. • Elements on the periodic table are ordered according to ...
Chapter 4
... • The number of protons in the nucleus of an atom is called the atomic number. • All atoms of any given element have the same atomic number. An element’s atomic number sets the atoms of that element apart from the atoms of all other elements. • Elements on the periodic table are ordered according to ...
... • The number of protons in the nucleus of an atom is called the atomic number. • All atoms of any given element have the same atomic number. An element’s atomic number sets the atoms of that element apart from the atoms of all other elements. • Elements on the periodic table are ordered according to ...
Chemistry I Honors Semester I FINAL EXAM REVIEW Atomic
... c. having lost electrons owing to energy content. d. having a definite volume but not a definite shape. ____ 9. A mixture is a. a combination of pure substances bonded chemically. b. any substance with a uniform composition. c. a blend of any two or more kinds of matter, as long as each maintains it ...
... c. having lost electrons owing to energy content. d. having a definite volume but not a definite shape. ____ 9. A mixture is a. a combination of pure substances bonded chemically. b. any substance with a uniform composition. c. a blend of any two or more kinds of matter, as long as each maintains it ...
Phosphorus - Jimmy Lai
... Atomic Name For P is Phosphorus Atomic Number For Phosphorus is 15 Atomic Mass For Phosphorus is 30.97376 ...
... Atomic Name For P is Phosphorus Atomic Number For Phosphorus is 15 Atomic Mass For Phosphorus is 30.97376 ...
FREE Sample Here
... B) Covalent bonds involve the sharing of neutrons between atoms, and ionic bonds involve the sharing of electrons between atoms. C) Covalent bonds involve the sharing of electrons between atoms, and ionic bonds involve the electrical attraction between atoms. D) Covalent bonds involve the sharing of ...
... B) Covalent bonds involve the sharing of neutrons between atoms, and ionic bonds involve the sharing of electrons between atoms. C) Covalent bonds involve the sharing of electrons between atoms, and ionic bonds involve the electrical attraction between atoms. D) Covalent bonds involve the sharing of ...
The structure and mass of atoms - Brentwood Ursuline Convent
... Figure 1 Close-up of an atom. Note that the sizes of the protons, neutrons and electrons are not to scale. ...
... Figure 1 Close-up of an atom. Note that the sizes of the protons, neutrons and electrons are not to scale. ...
FREE Sample Here
... 41) A covalent chemical bond is one in which A) electrons are removed from one atom and transferred to another atom so that the two atoms become oppositely charged. B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms. C) outer-shell electrons of two atoms ...
... 41) A covalent chemical bond is one in which A) electrons are removed from one atom and transferred to another atom so that the two atoms become oppositely charged. B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms. C) outer-shell electrons of two atoms ...
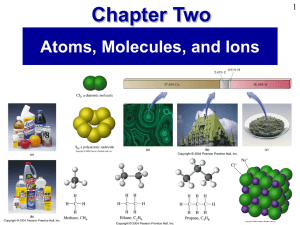
Prentice Hall Ch 02 Atoms Molecules Ions
... We usually write mass ratios in a form such as “the ratio of O to Mg is 0.6583:1.” The first number represents the mass of the first element named—in this case, a mass of oxygen, say 0.6583 g oxygen—and the second number represents the mass of the second element named—here a mass of magnesium. Altho ...
... We usually write mass ratios in a form such as “the ratio of O to Mg is 0.6583:1.” The first number represents the mass of the first element named—in this case, a mass of oxygen, say 0.6583 g oxygen—and the second number represents the mass of the second element named—here a mass of magnesium. Altho ...
FREE Sample Here
... 3) Three or four of the following statements are true and correct. Which one, if any, is false? If all the statements are true, choose answer E. A) Carbon, hydrogen, oxygen, and nitrogen make up approximately 96% of living matter. B) The trace element iodine is required only in very small quantities ...
... 3) Three or four of the following statements are true and correct. Which one, if any, is false? If all the statements are true, choose answer E. A) Carbon, hydrogen, oxygen, and nitrogen make up approximately 96% of living matter. B) The trace element iodine is required only in very small quantities ...
FREE Sample Here
... 41) A covalent chemical bond is one in which A) electrons are removed from one atom and transferred to another atom so that the two atoms become oppositely charged. B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms. C) outer-shell electrons of two atoms ...
... 41) A covalent chemical bond is one in which A) electrons are removed from one atom and transferred to another atom so that the two atoms become oppositely charged. B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms. C) outer-shell electrons of two atoms ...
Biology, 8e (Campbell) Chapter 2 The Chemical Context of Life
... 41) A covalent chemical bond is one in which A) electrons are removed from one atom and transferred to another atom so that the two atoms become oppositely charged. B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms. C) outer-shell electrons of two atoms ...
... 41) A covalent chemical bond is one in which A) electrons are removed from one atom and transferred to another atom so that the two atoms become oppositely charged. B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms. C) outer-shell electrons of two atoms ...
The Masses of Atoms
... I ask the indulgence of chemists and physicist for the approach I have taken with this book. The simplifications and short cuts that I have taken with their subjects, the anecdotal nature of this book as well as its cartoons, all have one sole aim: to allow a reader, who has not done much chemistry, ...
... I ask the indulgence of chemists and physicist for the approach I have taken with this book. The simplifications and short cuts that I have taken with their subjects, the anecdotal nature of this book as well as its cartoons, all have one sole aim: to allow a reader, who has not done much chemistry, ...
Biology, 8e (Campbell) Chapter 2 The Chemical Context of Life
... 3) Three or four of the following statements are true and correct. Which one, if any, is false? If all the statements are true, choose answer E. A) Carbon, hydrogen, oxygen, and nitrogen make up approximately 96% of living matter. B) The trace element iodine is required only in very small quantities ...
... 3) Three or four of the following statements are true and correct. Which one, if any, is false? If all the statements are true, choose answer E. A) Carbon, hydrogen, oxygen, and nitrogen make up approximately 96% of living matter. B) The trace element iodine is required only in very small quantities ...
Theories of the constitution of gases in the early nineteenth century
... have been reserved for those devised merely to explain the behaviour of gases without helping the general progress of chemistry. Dalton’s theory, supported by the laws of constant, multiple and equivalent proportions and based on the combining weight of solids, gave enormous help in the classificati ...
... have been reserved for those devised merely to explain the behaviour of gases without helping the general progress of chemistry. Dalton’s theory, supported by the laws of constant, multiple and equivalent proportions and based on the combining weight of solids, gave enormous help in the classificati ...
Biology, 8e (Campbell)
... B) The trace element iodine is required only in very small quantities by vertebrates. C) Virtually all organisms require the same elements in the same quantities. D) Iron is an example of an element needed by all organisms. E) All of the other statements are true and correct. Answer: C Topic: Concep ...
... B) The trace element iodine is required only in very small quantities by vertebrates. C) Virtually all organisms require the same elements in the same quantities. D) Iron is an example of an element needed by all organisms. E) All of the other statements are true and correct. Answer: C Topic: Concep ...
FREE Sample Here - We can offer most test bank and
... a. All atoms of a given element have the same weight. b. Atoms of different elements combine in fixed whole number ratios. c. The weight of an object is neither created nor destroyed in a chemical reaction. d. All samples of a given compound have the same proportion of constituent elements. e. The s ...
... a. All atoms of a given element have the same weight. b. Atoms of different elements combine in fixed whole number ratios. c. The weight of an object is neither created nor destroyed in a chemical reaction. d. All samples of a given compound have the same proportion of constituent elements. e. The s ...
Chemistry - Summative Practice and Review for Chapter 4 and 5
... 54. Use the periodic table to determine the number of neutrons in nitrogen-14. 55. How many neutrons are present in an atom of the isotope 56. Calculate the number of neutrons in ...
... 54. Use the periodic table to determine the number of neutrons in nitrogen-14. 55. How many neutrons are present in an atom of the isotope 56. Calculate the number of neutrons in ...
Section 5 – Harry Moseley: Numbering the
... Radioactive isotopes are unstable and undergo spontaneous nuclear reactions, emitting particles and/or wavelike radiation. The decay of any one nucleus cannot be predicted, but a large group of identical nuclei decay at a predictable rate. This predictability can be used to estimate the age of mat ...
... Radioactive isotopes are unstable and undergo spontaneous nuclear reactions, emitting particles and/or wavelike radiation. The decay of any one nucleus cannot be predicted, but a large group of identical nuclei decay at a predictable rate. This predictability can be used to estimate the age of mat ...
Unit 1 Section 4 - Atomic Structure PPT
... the quantum mechanical model further It determined the allowed energies an electron can have Also, Schrodinger developed an equation to determine how likely it is to find an electron in a particular location around the nucleus of an atom. Return to TOC ...
... the quantum mechanical model further It determined the allowed energies an electron can have Also, Schrodinger developed an equation to determine how likely it is to find an electron in a particular location around the nucleus of an atom. Return to TOC ...
Follow this presentation to draw atoms 1-13
... electrons can have stable orbits around the nucleus. The Bohr Model is probably familiar as the "planetary model" of the atom. In the Bohr Model the neutrons and protons occupy a dense central region called the nucleus, and the electrons orbit the nucleus much like planets orbiting the Sun (but the ...
... electrons can have stable orbits around the nucleus. The Bohr Model is probably familiar as the "planetary model" of the atom. In the Bohr Model the neutrons and protons occupy a dense central region called the nucleus, and the electrons orbit the nucleus much like planets orbiting the Sun (but the ...
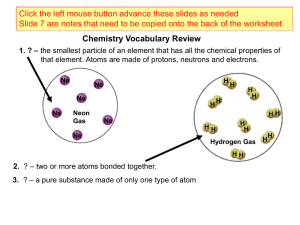
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.