Atomic Structure and the Periodic Table Vocabulary
... 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __________________________________________________________________ 4. ...
... 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __________________________________________________________________ 4. ...
Classifying Atoms
... The identity of an atom depends only on the number of protons, not on the number of neutrons. Many elements have atoms with the same number of protons but different numbers of neutrons. Atoms of an element that have different masses are isotopes (EYE-suh-TOHPS). The mass number of any given isotope ...
... The identity of an atom depends only on the number of protons, not on the number of neutrons. Many elements have atoms with the same number of protons but different numbers of neutrons. Atoms of an element that have different masses are isotopes (EYE-suh-TOHPS). The mass number of any given isotope ...
C2- Topic 1: Atomic structure and the periodic table. Assessable
... nucleus and that this number is unique to that element ...
... nucleus and that this number is unique to that element ...
Homework Geochem Test Review
... 2. What is smallest part of an element that has all the properties of that element? ...
... 2. What is smallest part of an element that has all the properties of that element? ...
C2 Topic 1 Can Do Sheet
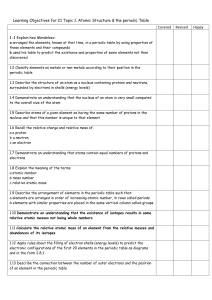
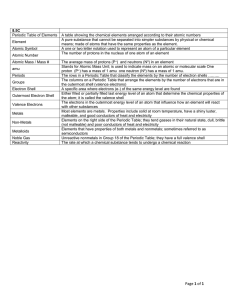
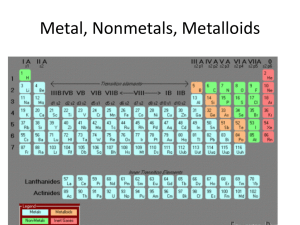
... a arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds b used his table to predict the existence and properties of some elements not then discovered 1.2 Classify elements as metals or non-metals according to their position in the pe ...
... a arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds b used his table to predict the existence and properties of some elements not then discovered 1.2 Classify elements as metals or non-metals according to their position in the pe ...
Chapter 3
... • Electrons moving through a glass tube with little pressure inside. • J.J. Thompson ...
... • Electrons moving through a glass tube with little pressure inside. • J.J. Thompson ...
Introduction to the Periodic Table
... The number of protons and neutrons in the nucleus of an atom. ...
... The number of protons and neutrons in the nucleus of an atom. ...
Chapter 3 Chemical Foundations
... Atomic Number, Mass Number, and Nucleus atomic number (Z) = mass number (A) = element symbol (X) = Note: mass number= Therefore …. mass number = ……. A= Z + number of neutrons ….. Number of neutrons = A-Z Note: For any given element on the periodic table: Number of protons = In order to symbolically ...
... Atomic Number, Mass Number, and Nucleus atomic number (Z) = mass number (A) = element symbol (X) = Note: mass number= Therefore …. mass number = ……. A= Z + number of neutrons ….. Number of neutrons = A-Z Note: For any given element on the periodic table: Number of protons = In order to symbolically ...
File
... • Heterogeneous Mixture-a mixture in which the presence of a t least two different substances is visible to the eye. • Homogenous Mixture-a mixture with a composition that is uniform throughout, all the way down to the molecular level. • Hydrocarbon-any molecule consisting of only hydrogen and carbo ...
... • Heterogeneous Mixture-a mixture in which the presence of a t least two different substances is visible to the eye. • Homogenous Mixture-a mixture with a composition that is uniform throughout, all the way down to the molecular level. • Hydrocarbon-any molecule consisting of only hydrogen and carbo ...
File
... nucleus of an atom. (The number of protons in the nucleus is the atomic number, which determines the identity of an element.) neutron: a subatomic particle that has no charge and that is located in the nucleus of an atom. electron: a subatomic particle that has a negative charge. element: a substanc ...
... nucleus of an atom. (The number of protons in the nucleus is the atomic number, which determines the identity of an element.) neutron: a subatomic particle that has no charge and that is located in the nucleus of an atom. electron: a subatomic particle that has a negative charge. element: a substanc ...
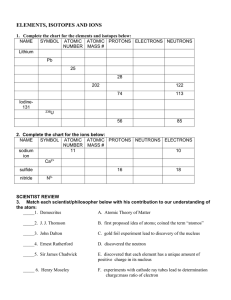
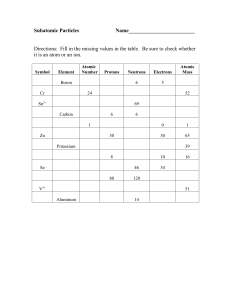
ELEMENTS, ISOTOPES AND IONS
... SYMBOL ATOMIC ATOMIC PROTONS NEUTRONS ELECTRONS NUMBER MASS # sodium ...
... SYMBOL ATOMIC ATOMIC PROTONS NEUTRONS ELECTRONS NUMBER MASS # sodium ...
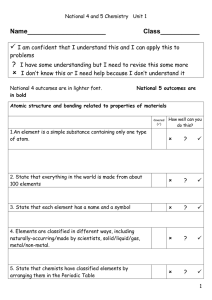
L.O.
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
Slide 1
... The multimedia approach of the standards is refreshing, and the focus on giving students a clear way to access the vocabulary of the standard is essential. This work reflects the new paradigms of teaching and learning that is being infused into educational classrooms of the 21st century. ...
... The multimedia approach of the standards is refreshing, and the focus on giving students a clear way to access the vocabulary of the standard is essential. This work reflects the new paradigms of teaching and learning that is being infused into educational classrooms of the 21st century. ...
The Chemical Basis of Life Chapter 4
... •Divided into pure substances or mixtures based on composition. –Pure substance •Uniform composition with same properties throughout •Can be classified as either an element or a compound ...
... •Divided into pure substances or mixtures based on composition. –Pure substance •Uniform composition with same properties throughout •Can be classified as either an element or a compound ...
Chemical Basis of Life
... 3- Atoms – the word comes from the Latin word atomos, which means indivisible. An atom is the simplest part of an element that contains all of the properties of the element. It would take 3 million carbon atoms lined next to each other to be the size of the period at the end of this sentence. 4- Al ...
... 3- Atoms – the word comes from the Latin word atomos, which means indivisible. An atom is the simplest part of an element that contains all of the properties of the element. It would take 3 million carbon atoms lined next to each other to be the size of the period at the end of this sentence. 4- Al ...
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.