Stoichiometry
... The propor?ons of elements in stoichiometric compounds. In H2O, hydrogen and oxygen is 2:1. Reac?on (Chemical Equa?on) Stoichiometry: The stoichiometric propor?ons by which species combine or are produced in chemical equa?ons. 2 H2 + O2 2 H20 Mass‐to‐Mass Stoichiometry: The mass stoichio ...
... The propor?ons of elements in stoichiometric compounds. In H2O, hydrogen and oxygen is 2:1. Reac?on (Chemical Equa?on) Stoichiometry: The stoichiometric propor?ons by which species combine or are produced in chemical equa?ons. 2 H2 + O2 2 H20 Mass‐to‐Mass Stoichiometry: The mass stoichio ...
AP Chemistry Curriculum Map - Belle Vernon Area School District
... orbitals with electrons, distribution of electrons in orbitals, shapes of orbitals). Anchor: CHEM.A.2.3 – Explain how periodic trends in the properties of atoms allow for the prediction of physical and chemical properties. Eligible Content CHEM.A.2.3.1 – Explain how the periodicity of chemical pro ...
... orbitals with electrons, distribution of electrons in orbitals, shapes of orbitals). Anchor: CHEM.A.2.3 – Explain how periodic trends in the properties of atoms allow for the prediction of physical and chemical properties. Eligible Content CHEM.A.2.3.1 – Explain how the periodicity of chemical pro ...
4.1 Defining the Atom
... If cathode rays are electrons given off by atoms, what remains of the atoms that have lost the electrons? • For example, after a hydrogen atom (the lightest kind of atom) loses an electron, what is left? ...
... If cathode rays are electrons given off by atoms, what remains of the atoms that have lost the electrons? • For example, after a hydrogen atom (the lightest kind of atom) loses an electron, what is left? ...
1ST CHAPTER Long-questions-basic-concept
... The term atom is derived from the Greek word “atoms” meaning indivisible. The smallest particle of an element which may or may not have independent existence is called an atom. For example ,the atoms of He,Ne and A r exist independently while the atoms of hydrogen ,nitrogen and oxygen do not have in ...
... The term atom is derived from the Greek word “atoms” meaning indivisible. The smallest particle of an element which may or may not have independent existence is called an atom. For example ,the atoms of He,Ne and A r exist independently while the atoms of hydrogen ,nitrogen and oxygen do not have in ...
Chemistry Challenge Problems
... its atoms. The noble gas elements, for example, all have similar chemical properties because the outermost energy levels of their atoms are completely filled. The chemical properties of ions also depend on the number of valence electrons. Any ion with a complete outermost energy level will have chem ...
... its atoms. The noble gas elements, for example, all have similar chemical properties because the outermost energy levels of their atoms are completely filled. The chemical properties of ions also depend on the number of valence electrons. Any ion with a complete outermost energy level will have chem ...
Get a clicker please
... of the following elements has the smallest first ionization energy? A potassium B magnesium C sodium D calcium ...
... of the following elements has the smallest first ionization energy? A potassium B magnesium C sodium D calcium ...
Chemistry Atoms, Molecules, and Ions 2.1 Multiple
... B) Rutherford's gold foil experiment. C) Thomson's cathode ray tube experiment. D) None of these Answer: B Topic: Section 2.4 Atomic Structure: Protons and Neutrons 18) The existence of neutrons in the nucleus of an atom was demonstrated by A) Millikan's oil drop experiment. B) Rutherford's gold foi ...
... B) Rutherford's gold foil experiment. C) Thomson's cathode ray tube experiment. D) None of these Answer: B Topic: Section 2.4 Atomic Structure: Protons and Neutrons 18) The existence of neutrons in the nucleus of an atom was demonstrated by A) Millikan's oil drop experiment. B) Rutherford's gold foi ...
Chemical Reactions
... • Law of Conservation of Mass • Matter cannot be created nor destroyed • Just like with math, both sides of the YIELDS sign must be equal! • So the same number of atoms must be on both sides of the equation ...
... • Law of Conservation of Mass • Matter cannot be created nor destroyed • Just like with math, both sides of the YIELDS sign must be equal! • So the same number of atoms must be on both sides of the equation ...
Chemistry
... alchemy was nothing but a hoax? Although we can’t use chemistry to make gold or to live forever, modern chemistry is still very powerful. There may be no such thing as a potion that cures all diseases, but many chemists today are working to develop cures for specific diseases, including HIV/AIDS and ...
... alchemy was nothing but a hoax? Although we can’t use chemistry to make gold or to live forever, modern chemistry is still very powerful. There may be no such thing as a potion that cures all diseases, but many chemists today are working to develop cures for specific diseases, including HIV/AIDS and ...
FREE Sample Here - We can offer most test bank and
... b. The theory can still be used because the erroneous postulate does not have any effect on the physical properties of the elements. c. The theory can still be used because the erroneous postulate does not have any effect on the chemical properties of the elements. d. The theory can still be used be ...
... b. The theory can still be used because the erroneous postulate does not have any effect on the physical properties of the elements. c. The theory can still be used because the erroneous postulate does not have any effect on the chemical properties of the elements. d. The theory can still be used be ...
Some basic concepts of chemistry
... composition and properties of matter and the reactions by which matter converts from one form to the other. NEET: Chemistry (Vol. I) not only adds great value towards a progressive society but also contributes greatly to other branches of science like biology, physics, geology, astronomy, biotechnol ...
... composition and properties of matter and the reactions by which matter converts from one form to the other. NEET: Chemistry (Vol. I) not only adds great value towards a progressive society but also contributes greatly to other branches of science like biology, physics, geology, astronomy, biotechnol ...
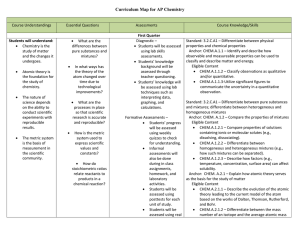
Subject Area Standard Area Organizing Category Grade Level
... CHEM.B.2.2.1: Utilize mathematical relationships to predict changes in the number of particles, the temperature, the pressure, and the volume in a gaseous system (i.e., Boyle’s law, Charles’s law, Dalton’s law of partial pressures, the combined gas law, and the ideal gas law). ...
... CHEM.B.2.2.1: Utilize mathematical relationships to predict changes in the number of particles, the temperature, the pressure, and the volume in a gaseous system (i.e., Boyle’s law, Charles’s law, Dalton’s law of partial pressures, the combined gas law, and the ideal gas law). ...
When wood, paper, and wax are burned, they ap
... In this chapter we will use what we have learned about chemical structure and formulas in studying the mass relationships of atoms and molecules. These relationships in turn will help us to explain the composition of compounds and the ways in which the composition changes. The mass of an atom is rel ...
... In this chapter we will use what we have learned about chemical structure and formulas in studying the mass relationships of atoms and molecules. These relationships in turn will help us to explain the composition of compounds and the ways in which the composition changes. The mass of an atom is rel ...
Learning Outcomes
... of s, p, d and f classification is not required; a copy of the Periodic Table will be available in Papers 1 and 2) ......................................................................................................................... 16 (c) define proton (atomic) number and nucleon (mass) number ...
... of s, p, d and f classification is not required; a copy of the Periodic Table will be available in Papers 1 and 2) ......................................................................................................................... 16 (c) define proton (atomic) number and nucleon (mass) number ...
Chapter 3: Elements, Compounds and the Periodic Table
... Number of protons that atom has in nucleus Unique to each type of element Element is substance whose atoms all contain identical number of protons ...
... Number of protons that atom has in nucleus Unique to each type of element Element is substance whose atoms all contain identical number of protons ...
Presentation
... are found in nature. • Some have special properties because they are unstable. • An unstable atom is an atom with a nucleus that will change over time. (This is called radioactive.) • They spontaneously fall apart after a certain amount of time. As they do, they give off smaller particles, as well a ...
... are found in nature. • Some have special properties because they are unstable. • An unstable atom is an atom with a nucleus that will change over time. (This is called radioactive.) • They spontaneously fall apart after a certain amount of time. As they do, they give off smaller particles, as well a ...
Dr. Spencer`s PPT
... Democritus (460 BC) and John Dalton (1800s) - No (“atomos”means indivisible”) Chapt. 2.1 ...
... Democritus (460 BC) and John Dalton (1800s) - No (“atomos”means indivisible”) Chapt. 2.1 ...
Chemistry M.4 Lesson 1 Atom and Periodic Table
... Usually brittle solids or gases ,except for bromine(Br) Found on right side of periodic table AND hydrogen ...
... Usually brittle solids or gases ,except for bromine(Br) Found on right side of periodic table AND hydrogen ...
Slide 1
... 1. What are the reactants in this chemical equation? 2. What are the products in this chemical equation? 3. Are there the same number of atoms on both sides of the equation? a. Were any atoms destroyed or created? b. Was the Law of Conservation of Matter maintained? ...
... 1. What are the reactants in this chemical equation? 2. What are the products in this chemical equation? 3. Are there the same number of atoms on both sides of the equation? a. Were any atoms destroyed or created? b. Was the Law of Conservation of Matter maintained? ...
Chapter 3
... hydrogen atom, all atomic nuclei are made of protons and neutrons. • A proton has a positive charge equal in magnitude to the negative charge of an electron. • Atoms are electrically neutral because they contain equal numbers of protons and electrons. • A neutron is electrically neutral. Chapter men ...
... hydrogen atom, all atomic nuclei are made of protons and neutrons. • A proton has a positive charge equal in magnitude to the negative charge of an electron. • Atoms are electrically neutral because they contain equal numbers of protons and electrons. • A neutron is electrically neutral. Chapter men ...
Chemistry 121: Topic 2 - From Atoms to Stoichiometry Topic 2
... ¾ An Extensive Property is a property that depends on how much matter is being examined ie., mass ¾ An Intensive Property is a property that does not depend on how much matter is being examined ie., density ...
... ¾ An Extensive Property is a property that depends on how much matter is being examined ie., mass ¾ An Intensive Property is a property that does not depend on how much matter is being examined ie., density ...
Chapter 3 - Robinson Schools
... hydrogen atom, all atomic nuclei are made of protons and neutrons. • A proton has a positive charge equal in magnitude to the negative charge of an electron. • Atoms are electrically neutral because they contain equal numbers of protons and electrons. • A neutron is electrically neutral. Chapter men ...
... hydrogen atom, all atomic nuclei are made of protons and neutrons. • A proton has a positive charge equal in magnitude to the negative charge of an electron. • Atoms are electrically neutral because they contain equal numbers of protons and electrons. • A neutron is electrically neutral. Chapter men ...
Student Study Guide 1999
... For example: Chalk and water. A solid that does not dissolve when mixed in a liquid or a gas, forms a suspension. It forms a cloudy heterogenous mixture. The two substances will separate by themselves if left standing for long enough. They can also be separated by decantation and filtration. ...
... For example: Chalk and water. A solid that does not dissolve when mixed in a liquid or a gas, forms a suspension. It forms a cloudy heterogenous mixture. The two substances will separate by themselves if left standing for long enough. They can also be separated by decantation and filtration. ...
Summer Work: Independent Packet: Basics Of Atomic Structure
... almost any other course you have had to date. But, while the content must be mastered - that doesn’t imply that this packet must be 100% completed. Since, many of these ideas have been taught to you and/or touched upon since 6th grade you may be terrific at this work, and need to do very little revi ...
... almost any other course you have had to date. But, while the content must be mastered - that doesn’t imply that this packet must be 100% completed. Since, many of these ideas have been taught to you and/or touched upon since 6th grade you may be terrific at this work, and need to do very little revi ...
Chapter 3 PowerPoint Presentation
... • Every substance has its own unique set of physical and chemical properties. • Observations of properties may vary depending on the conditions of the immediate environment. • It is important to state the specific conditions in which observations are made because both chemical and physical propertie ...
... • Every substance has its own unique set of physical and chemical properties. • Observations of properties may vary depending on the conditions of the immediate environment. • It is important to state the specific conditions in which observations are made because both chemical and physical propertie ...
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.