Periodic Trends - Barrington 220
... EXPLANATION: as electrons are added to more energy levels, the inner layers shield the outer electrons from the pull of the nucleus. The added electrons have less attraction to the nucleus due to this shielding effect, increasing the atom‛s size ...
... EXPLANATION: as electrons are added to more energy levels, the inner layers shield the outer electrons from the pull of the nucleus. The added electrons have less attraction to the nucleus due to this shielding effect, increasing the atom‛s size ...
The study of biology can help you better understand human
... Atoms of isotopes of an element have different number of protons The nucleus of an atom has positive charge. Atoms are mostly empty space. ...
... Atoms of isotopes of an element have different number of protons The nucleus of an atom has positive charge. Atoms are mostly empty space. ...
The diameter of a Ni atom is
... • Both are characterized by • Both are characterized by the their physical and chemical nature of their sub-atomic properties structure. ...
... • Both are characterized by • Both are characterized by the their physical and chemical nature of their sub-atomic properties structure. ...
Midterm Review File
... d. Identify the element in the oxygen group whose outermost electrons are in the fourth energy level____. e. Identify the name of a group of elements that contains only metals______________. f. Identify a characteristic that elements in the same period have in common_____________. 20. Answer the fol ...
... d. Identify the element in the oxygen group whose outermost electrons are in the fourth energy level____. e. Identify the name of a group of elements that contains only metals______________. f. Identify a characteristic that elements in the same period have in common_____________. 20. Answer the fol ...
Compounds Booklet Companion New 2013
... square. Atomic mass is always expressed as a decimal, because each element, except for Fluorine, can form isotopes, which are atoms with different numbers of neutrons in them and therefore different masses. Atomic mass is the average mass of all an element’s different isotopes. Mass is measured in A ...
... square. Atomic mass is always expressed as a decimal, because each element, except for Fluorine, can form isotopes, which are atoms with different numbers of neutrons in them and therefore different masses. Atomic mass is the average mass of all an element’s different isotopes. Mass is measured in A ...
CW07 Electron Structure of the Atom Click HERE for Puzzle Click
... The elements that have full s and p orbitals are the ___ elements (7.7; 2WWS) The amount of energy required to remove a valence electron from an atom is called ___ energy (7.7) The atomic size generally ___ as we move left to right in a period (7.7) The lowest energy electron configuration (7.3; 2WW ...
... The elements that have full s and p orbitals are the ___ elements (7.7; 2WWS) The amount of energy required to remove a valence electron from an atom is called ___ energy (7.7) The atomic size generally ___ as we move left to right in a period (7.7) The lowest energy electron configuration (7.3; 2WW ...
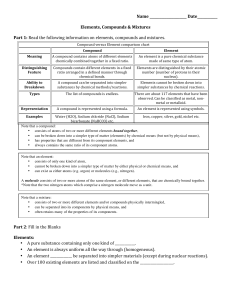
Compound vs Element chart
... • consists of atoms of two or more different elements bound together, • can be broken down into a simpler type of matter (elements) by chemical means (but not by physical means), • has properties that are different from its component elements, and • always contains the same ratio of its component at ...
... • consists of atoms of two or more different elements bound together, • can be broken down into a simpler type of matter (elements) by chemical means (but not by physical means), • has properties that are different from its component elements, and • always contains the same ratio of its component at ...
Unit 3 Note Outline
... Average Atomic Mass Atomic Mass Unit (AMU) Average Atomic Mass - The atomic mass of an element listed on the periodic table is a In order to calculate the average atomic mass of an element you must know: 1. The 2. The 3. The ...
... Average Atomic Mass Atomic Mass Unit (AMU) Average Atomic Mass - The atomic mass of an element listed on the periodic table is a In order to calculate the average atomic mass of an element you must know: 1. The 2. The 3. The ...
Chapter 4 4.1 Defining the Atom • Early Models of the Atom atom
... one element are different from those of any other element 3) Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. 4) Chemical reactions occur when atoms are separated from each other, joined, or rearranged in a different c ...
... one element are different from those of any other element 3) Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. 4) Chemical reactions occur when atoms are separated from each other, joined, or rearranged in a different c ...
Chapter 2
... John Dalton and the Atomic Theory of Matter • 1. All matter is composed of extremely small particles called atoms. • 2. All atoms of a given element are alike and differ from the atoms of any other element. • 3. Compounds are formed when atoms of different elements combine in fixed proportions. • 4 ...
... John Dalton and the Atomic Theory of Matter • 1. All matter is composed of extremely small particles called atoms. • 2. All atoms of a given element are alike and differ from the atoms of any other element. • 3. Compounds are formed when atoms of different elements combine in fixed proportions. • 4 ...
Preview from Notesale.co.uk Page 2 of 12
... usual in the order of their atomic number, from 2 to 20. The red numbers below each chemical symbol show its electronic structure. Moving across each period, you can see that the number of occupie ...
... usual in the order of their atomic number, from 2 to 20. The red numbers below each chemical symbol show its electronic structure. Moving across each period, you can see that the number of occupie ...
Remember Question words
... because it has gained or lost electrons cation = an atom with more protons than electrons is a positively charged ion anion = an atom with more electrons than protons is a negatively charged ion electrical charge = # of electrons - # of protons ...
... because it has gained or lost electrons cation = an atom with more protons than electrons is a positively charged ion anion = an atom with more electrons than protons is a negatively charged ion electrical charge = # of electrons - # of protons ...
Atomic Mass
... Atomic masses can be different for atoms of the same element if they have different numbers of neutrons Atoms with different masses are called Isotopes or Nuclides ...
... Atomic masses can be different for atoms of the same element if they have different numbers of neutrons Atoms with different masses are called Isotopes or Nuclides ...
Review Outline for Atomic Structure Test
... _P,As,Sb,Bi__8. Name of another element in the same family with Nitrogen _Li,Be,B,C,O,F,Ne____9. Name of another element in the same period with Nitrogen ...
... _P,As,Sb,Bi__8. Name of another element in the same family with Nitrogen _Li,Be,B,C,O,F,Ne____9. Name of another element in the same period with Nitrogen ...
- Lexington JHS
... • What makes an element reactive? – An incomplete valence electron level. – All atoms (except hydrogen) want to have 8 electrons in their very outermost energy level (This is called the rule of octet.) – Atoms bond until this level is complete. Atoms with few valence electrons lose them during bondi ...
... • What makes an element reactive? – An incomplete valence electron level. – All atoms (except hydrogen) want to have 8 electrons in their very outermost energy level (This is called the rule of octet.) – Atoms bond until this level is complete. Atoms with few valence electrons lose them during bondi ...
Document
... • What makes an element reactive? – An incomplete valence electron level. – All atoms (except hydrogen) want to have 8 electrons in their very outermost energy level (This is called the rule of octet.) – Atoms bond until this level is complete. Atoms with few valence electrons lose them during bondi ...
... • What makes an element reactive? – An incomplete valence electron level. – All atoms (except hydrogen) want to have 8 electrons in their very outermost energy level (This is called the rule of octet.) – Atoms bond until this level is complete. Atoms with few valence electrons lose them during bondi ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.