atoms
... elements composed of atoms atoms of the same element are alike different atoms can combine in ratios to form compounds chemical reactions can occur when atoms are separated, joined, or rearranged (but atoms are not created nor destroyed) ...
... elements composed of atoms atoms of the same element are alike different atoms can combine in ratios to form compounds chemical reactions can occur when atoms are separated, joined, or rearranged (but atoms are not created nor destroyed) ...
Atoms The smallest piece of matter that have specific properties of
... Found in the atomic nucleus. Neutron (no charge neutrons) No charge (neutral) Found in the nucleus. Electron (negative electrons) Negatively charged particles Found in the outer shells. Electrons determine properties of the atom. Chemical reactions involve sharing or exchanging electrons. ...
... Found in the atomic nucleus. Neutron (no charge neutrons) No charge (neutral) Found in the nucleus. Electron (negative electrons) Negatively charged particles Found in the outer shells. Electrons determine properties of the atom. Chemical reactions involve sharing or exchanging electrons. ...
Chapter 5 Review
... Of the elements Pt, Sc, V, Li, and Kr, which is/are nonmetal(s)? What is each vertical column of elements in the periodic table called? Approximately how many elements exist today? ...
... Of the elements Pt, Sc, V, Li, and Kr, which is/are nonmetal(s)? What is each vertical column of elements in the periodic table called? Approximately how many elements exist today? ...
Chapter 4 Study Guide
... Bohr’s Model is known as the __________________________ Model What is the purpose of energy levels in relation to an electron? ...
... Bohr’s Model is known as the __________________________ Model What is the purpose of energy levels in relation to an electron? ...
Atomic Number
... -Any material made of one type of atom is classified as an ______________________ -The Periodic Table is organized into rows and columns -Each vertical column is called a ____________________ or____________________ -Elements within the same group have similar properties EX. Au, Ag, Cu -Each horizont ...
... -Any material made of one type of atom is classified as an ______________________ -The Periodic Table is organized into rows and columns -Each vertical column is called a ____________________ or____________________ -Elements within the same group have similar properties EX. Au, Ag, Cu -Each horizont ...
Chapter 8. The Periodic Table
... chemical properties. The first column of active metals on the left is called the alkali metal family, the second alkaline earths,, the lanthanides and actinides together are called rare earths, and the last three representative families are called calcogens, halogens and noble gases. (The remaining ...
... chemical properties. The first column of active metals on the left is called the alkali metal family, the second alkaline earths,, the lanthanides and actinides together are called rare earths, and the last three representative families are called calcogens, halogens and noble gases. (The remaining ...
Atomic Review
... 5. Given the relative abundance of the following naturally occurring isotopes of oxygen, calculate the average atomic mass of oxygen. Assume that the atomic mass of each is the same as the mass number. oxygen- 16: 99.76% oxygen17: 0.037% oxygen-18: 0.204% ...
... 5. Given the relative abundance of the following naturally occurring isotopes of oxygen, calculate the average atomic mass of oxygen. Assume that the atomic mass of each is the same as the mass number. oxygen- 16: 99.76% oxygen17: 0.037% oxygen-18: 0.204% ...
Notes on Atomic Structure Structure of Atoms Atoms are composed
... The periodic table is a list of the elements that make up matter. It is organized by increasing atomic number. The Atomic Number shows the number of protons in the nucleus of an atom. It identifies the type of atom/element. The atomic number also equals the number of electrons whenever the atom is n ...
... The periodic table is a list of the elements that make up matter. It is organized by increasing atomic number. The Atomic Number shows the number of protons in the nucleus of an atom. It identifies the type of atom/element. The atomic number also equals the number of electrons whenever the atom is n ...
Document
... Dalton’s Atomic Theory - Summary 1. matter is composed, indivisible particles (atoms) 2. all atoms of a particular element are identical 3. different elements have different atoms 4. atoms combine in certain whole-number ratios 5. In a chemical reaction, atoms are merely rearranged to form new comp ...
... Dalton’s Atomic Theory - Summary 1. matter is composed, indivisible particles (atoms) 2. all atoms of a particular element are identical 3. different elements have different atoms 4. atoms combine in certain whole-number ratios 5. In a chemical reaction, atoms are merely rearranged to form new comp ...
Atomic Theory WS
... 18. List the 3 subatomic particles, their symbols and the rest of the information found in a table in the PowerPoint. ...
... 18. List the 3 subatomic particles, their symbols and the rest of the information found in a table in the PowerPoint. ...
History of the Atom
... Foil Experiment and suggested the following characteristics of the atom: ...
... Foil Experiment and suggested the following characteristics of the atom: ...
Johnston Middle School Lesson Plan 2015-2016
... TSW describe the structure of atoms including the masses, electrical charges and locations of protons and neutrons in the nucleus and electrons in the electron cloud. TSW identify that protons determine an element's identity, and valence electrons determine its chemical properties and reactivity TSW ...
... TSW describe the structure of atoms including the masses, electrical charges and locations of protons and neutrons in the nucleus and electrons in the electron cloud. TSW identify that protons determine an element's identity, and valence electrons determine its chemical properties and reactivity TSW ...
Chemistry Unit 1 Revision
... • They are unreactive and do not easily form molecules because their atoms have stable arrangements of electrons. • The noble gases have eight electrons in their outer energy level, except for helium, which has only two electrons. • The boiling points of the noble gases increase with increasing rela ...
... • They are unreactive and do not easily form molecules because their atoms have stable arrangements of electrons. • The noble gases have eight electrons in their outer energy level, except for helium, which has only two electrons. • The boiling points of the noble gases increase with increasing rela ...
power point notes
... WAY! He thought that only 4 elements actually exist: water, air, fire and earth ...
... WAY! He thought that only 4 elements actually exist: water, air, fire and earth ...
The Nature of Molecules
... ***Inner energy shells (those closest to the nucleus) contain electrons with lower energy than the outer energy shells ***important concept as it will be discussed in the Light Dependent reaction of Photosynthesis ...
... ***Inner energy shells (those closest to the nucleus) contain electrons with lower energy than the outer energy shells ***important concept as it will be discussed in the Light Dependent reaction of Photosynthesis ...
Chapter 2
... • Chemical formula: atomic symbols with numeric subscripts that show relative proportions of atoms of different elements in a substance • Al2O3: ratio of Al atoms to O atoms is 2 : 3 • No subscript: ratio of 1 (NaCl, 1:1) ...
... • Chemical formula: atomic symbols with numeric subscripts that show relative proportions of atoms of different elements in a substance • Al2O3: ratio of Al atoms to O atoms is 2 : 3 • No subscript: ratio of 1 (NaCl, 1:1) ...
Station 1 - The Periodic Table, Molecules and Molecular
... 1. What is the distinction between atomic number and mass number? Between mass number and atomic mass? 2. Distinguish between the terms family and period in connection with the periodic table. For which of these is the term group also used. 3. When metals react with nonmetals, an ionic compound gene ...
... 1. What is the distinction between atomic number and mass number? Between mass number and atomic mass? 2. Distinguish between the terms family and period in connection with the periodic table. For which of these is the term group also used. 3. When metals react with nonmetals, an ionic compound gene ...
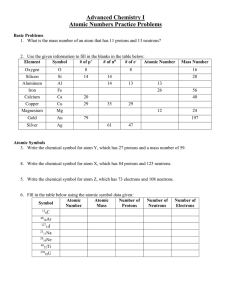
Atomic Numbers Practice Problems
... 7. With respect to the three fundamental particles of atoms, which numbers of particles are the same between two isotopes, and which ones are different? ...
... 7. With respect to the three fundamental particles of atoms, which numbers of particles are the same between two isotopes, and which ones are different? ...
Chemistry Study Guide What is matter made of? Matter is anything
... ● Atoms of nonmetals usually have more than 4 outer-shell electrons. ...
... ● Atoms of nonmetals usually have more than 4 outer-shell electrons. ...
5Periodic Table of Elements WB
... Valence electrons are how many electrons are in the outermost shell or period. Periodicity or PERIODIC LAW _________________________________________________________________ ...
... Valence electrons are how many electrons are in the outermost shell or period. Periodicity or PERIODIC LAW _________________________________________________________________ ...
Ch 4 and Ch 5 Study Guide (ICP) Multiple Choice Identify the choice
... 53. When an atom gains or loses energy, _______________________ jump between energy levels. 54. An electron that gains energy enters an excited state and absorbs a particle of light called a(n) ______________________. 55. The chemical properties of an atom are determined by _______________________, ...
... 53. When an atom gains or loses energy, _______________________ jump between energy levels. 54. An electron that gains energy enters an excited state and absorbs a particle of light called a(n) ______________________. 55. The chemical properties of an atom are determined by _______________________, ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.