4 1 introduction to atoms 65-68
... 6. The term Rutherford gave to the positively charged particles in the nucleus of an atom was ________________________. 7. In the atomic model proposed by ________________________ , electrons move in specific orbits, similar to how planets orbit the sun. 8. What particle did Chadwick discover in 193 ...
... 6. The term Rutherford gave to the positively charged particles in the nucleus of an atom was ________________________. 7. In the atomic model proposed by ________________________ , electrons move in specific orbits, similar to how planets orbit the sun. 8. What particle did Chadwick discover in 193 ...
Elements, Compounds, and Atoms Video Notes
... 10. Neils Bohr – proposed that electrons orbit around the nucleus; each electron has a fixed amount of energy and electrons orbit in around the nucleus. (Think of the rungs on a ...
... 10. Neils Bohr – proposed that electrons orbit around the nucleus; each electron has a fixed amount of energy and electrons orbit in around the nucleus. (Think of the rungs on a ...
Atoms: The Building Blocks of Matter
... Dalton’s Atomic Theory • All matter composed of atoms Atoms… • of same element are identical; atoms of diff. elements are diff. • cannot be divided, created, destroyed • combine in simple whole # ratios to form compounds (Law of Definite Proportions) • are combined, separated or rearranged in chem. ...
... Dalton’s Atomic Theory • All matter composed of atoms Atoms… • of same element are identical; atoms of diff. elements are diff. • cannot be divided, created, destroyed • combine in simple whole # ratios to form compounds (Law of Definite Proportions) • are combined, separated or rearranged in chem. ...
Chapter 11 Modern Atomic Theory
... (indicates opposite spins). No more than 2 arrows can be in a box. S orbital has 1 box; p has 3; d has 5; f has 7. Example 11.2,, p. 342- write the complete electron configurations and orbital diagrams for elements aluminum through argon.(do above in the table under 11.9) 11.10 Electron Configuratio ...
... (indicates opposite spins). No more than 2 arrows can be in a box. S orbital has 1 box; p has 3; d has 5; f has 7. Example 11.2,, p. 342- write the complete electron configurations and orbital diagrams for elements aluminum through argon.(do above in the table under 11.9) 11.10 Electron Configuratio ...
Quantum-Mechanical Description of Mendeleev periodic table
... metallic gold film. If the model was correct, the massive α -particles (their mass is 7300 times that of the electron), would go through the film with minor deflections in their paths. The result was that many particles went just through but some particles were deflected at large angles [3]. Then he ...
... metallic gold film. If the model was correct, the massive α -particles (their mass is 7300 times that of the electron), would go through the film with minor deflections in their paths. The result was that many particles went just through but some particles were deflected at large angles [3]. Then he ...
Notepack - Hood River County School District
... Copy Figure 4.10, pg. 115 (calculating weighted averages) ...
... Copy Figure 4.10, pg. 115 (calculating weighted averages) ...
Name
... • The nucleus is made up of at least one positively charged particle called a proton and usually one or more neutral particles called neutrons. • Surrounding the nucleus is a region occupied by negatively charged particles called electrons. • Protons, neutrons, and electrons are often referred to as ...
... • The nucleus is made up of at least one positively charged particle called a proton and usually one or more neutral particles called neutrons. • Surrounding the nucleus is a region occupied by negatively charged particles called electrons. • Protons, neutrons, and electrons are often referred to as ...
Atomic Mass - AJS Phyiscs and Chemistry
... • The period number tells you how many energy levels you have. • Periods 6 and 7 have 14 additional elements that are listed at the bottom of the periodic table so it is easier to print the table on a standard page. • Properties change in 2 ways as you move from left to right across a period: – The ...
... • The period number tells you how many energy levels you have. • Periods 6 and 7 have 14 additional elements that are listed at the bottom of the periodic table so it is easier to print the table on a standard page. • Properties change in 2 ways as you move from left to right across a period: – The ...
Review of the Atom
... pathways around nucleus, held in place by protons (+) •worked only for hydrogen ...
... pathways around nucleus, held in place by protons (+) •worked only for hydrogen ...
The Atom
... Ex: atoms of liquids were thought to be smooth, which would allow the atoms to slide over each other. ...
... Ex: atoms of liquids were thought to be smooth, which would allow the atoms to slide over each other. ...
We know now that it is composed of protons and neutrons.
... The nucleus of an atom is positively charged and contains almost all the mass of the atom. We know now that it is composed of protons and neutrons. Electrons orbit around the nucleus in circular orbits. The nucleus holds the electrons by means of an electric force between positive and negative charg ...
... The nucleus of an atom is positively charged and contains almost all the mass of the atom. We know now that it is composed of protons and neutrons. Electrons orbit around the nucleus in circular orbits. The nucleus holds the electrons by means of an electric force between positive and negative charg ...
AP Chemistry
... 2.5.3.1 Three common conventions for labeling the groups; 2.5.3.1.1 Dr. V likes numbering them simply 1-18 (the IUPAC recommendation) 2.5.3.1.2 This text uses Arabic numerals to number the columns, using the letter A to signify the main block elements, and the letter B to distinguish the transition ...
... 2.5.3.1 Three common conventions for labeling the groups; 2.5.3.1.1 Dr. V likes numbering them simply 1-18 (the IUPAC recommendation) 2.5.3.1.2 This text uses Arabic numerals to number the columns, using the letter A to signify the main block elements, and the letter B to distinguish the transition ...
Elements and the Periodic Table
... stair-step line on the right side of the table. • Metals are found to the left of the line and nonmetals are found to the right of the line. • Elements that border the line on both sides are called semi-metals. ...
... stair-step line on the right side of the table. • Metals are found to the left of the line and nonmetals are found to the right of the line. • Elements that border the line on both sides are called semi-metals. ...
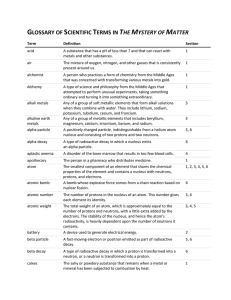
GLOSSARY OF SCIENTIFIC TERMS IN THE MYSTERY OF MATTER
... A type of science and philosophy from the Middle Ages that attempted to perform unusual experiments, taking something ordinary and turning it into something extraordinary. Any of a group of soft metallic elements that form alkali solutions when they combine with water. They include lithium, sodium, ...
... A type of science and philosophy from the Middle Ages that attempted to perform unusual experiments, taking something ordinary and turning it into something extraordinary. Any of a group of soft metallic elements that form alkali solutions when they combine with water. They include lithium, sodium, ...
Name: Period: _____ Date
... 2. _____ number of protons + neutrons; not on periodic table 3. _____ another name for a column on the periodic table, also called a family 4. _____ drawing of an atom that shows all the protons, neutrons and electrons 5. _____ positively charged particle in the atom 6. _____ the last column on the ...
... 2. _____ number of protons + neutrons; not on periodic table 3. _____ another name for a column on the periodic table, also called a family 4. _____ drawing of an atom that shows all the protons, neutrons and electrons 5. _____ positively charged particle in the atom 6. _____ the last column on the ...
Structure Changes of Matter
... b) Protons, each with positive charge, have about same mass as neutrons; Number of protons in atoms of each element is unique (= atomic no.). c) Neutrons, each with no charge, have about same size & mass as protons. Different numbers of neutrons in atom à different isotopes. d) Electrons, each with ...
... b) Protons, each with positive charge, have about same mass as neutrons; Number of protons in atoms of each element is unique (= atomic no.). c) Neutrons, each with no charge, have about same size & mass as protons. Different numbers of neutrons in atom à different isotopes. d) Electrons, each with ...
STRUCTURE_OF_THE_ATOM
... much changed for the next 1000 years. The ideas of Aristotle were still being taught. ...
... much changed for the next 1000 years. The ideas of Aristotle were still being taught. ...
Name: Period: _____ Date
... 2. _____ number of protons + neutrons; not on periodic table 3. _____ another name for a column on the periodic table, also called a family 4. _____ drawing of an atom that shows all the protons, neutrons and electrons 5. _____ positively charged particle in the atom 6. _____ the last column on the ...
... 2. _____ number of protons + neutrons; not on periodic table 3. _____ another name for a column on the periodic table, also called a family 4. _____ drawing of an atom that shows all the protons, neutrons and electrons 5. _____ positively charged particle in the atom 6. _____ the last column on the ...
File
... Atoms had too much mass - Scientists had discovered that atoms _____________________ more than they should if there was only _______________________ and _________________________. - There were also ________________________ of elements that weighed _________________________ amounts but had the ______ ...
... Atoms had too much mass - Scientists had discovered that atoms _____________________ more than they should if there was only _______________________ and _________________________. - There were also ________________________ of elements that weighed _________________________ amounts but had the ______ ...
Exam #2
... (a) Electron affinities decrease going down the group (from smaller to larger elements). (b) Ionization energies decrease going down the group (from smaller to larger elements). (c) Chemical reactivity decreases going down the group (from smaller to larger elements). (d) The second ionization energy ...
... (a) Electron affinities decrease going down the group (from smaller to larger elements). (b) Ionization energies decrease going down the group (from smaller to larger elements). (c) Chemical reactivity decreases going down the group (from smaller to larger elements). (d) The second ionization energy ...
Chapter 2 Law of
... 1. The cation name is given first followed by the anion name. 2. If the metal forms only one positive ion, the cation name is the element name. 3. If the metal forms more than one ppositive ion (transition/post-transitional elements), the cation name is the English name followed, without a space, by ...
... 1. The cation name is given first followed by the anion name. 2. If the metal forms only one positive ion, the cation name is the element name. 3. If the metal forms more than one ppositive ion (transition/post-transitional elements), the cation name is the English name followed, without a space, by ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.