Basic Atomic Theory
... • Approximation to the planetary model • Mathematically gravitational force similar to Coulomb forces • Strength of Coulomb forces much larger than gravitational • +ve and –ve charges cause attractive and repulsive ...
... • Approximation to the planetary model • Mathematically gravitational force similar to Coulomb forces • Strength of Coulomb forces much larger than gravitational • +ve and –ve charges cause attractive and repulsive ...
Bohr Models and Lewis Dot Structures
... • Periods go from left to • Groups go up and down right ...
... • Periods go from left to • Groups go up and down right ...
What is Everything Made Of?
... 1 Proton ( or Neutron)----1.67 x10-24 Grams =0.000000000000000000000001.67 Grams 1 electron-----9.1x10-28 Grams =0.0000000000000000000000000009.1Grams The Proton weighs 1800 x the Electron The Atom is about 5 X 10-8 Cm ...
... 1 Proton ( or Neutron)----1.67 x10-24 Grams =0.000000000000000000000001.67 Grams 1 electron-----9.1x10-28 Grams =0.0000000000000000000000000009.1Grams The Proton weighs 1800 x the Electron The Atom is about 5 X 10-8 Cm ...
Chemistry Vocab for Quiz 12/21 or 12/22 Atom – The smallest
... Chemistry Vocab for Quiz 12/21 or 12/22 Atom – The smallest particle of an element. Atomic number - The number of protons in the nucleus of an element Atomic mass – The average mass of one atom of an element Proton – A small positively particle in the nucleus Neutron – a small particle in the nucleu ...
... Chemistry Vocab for Quiz 12/21 or 12/22 Atom – The smallest particle of an element. Atomic number - The number of protons in the nucleus of an element Atomic mass – The average mass of one atom of an element Proton – A small positively particle in the nucleus Neutron – a small particle in the nucleu ...
國立屏東教育大學95學年度研究所碩士班入學考試
... 7. The diagram below is the Born-huber cycle for the formation of crystalline potassium fluoride. ...
... 7. The diagram below is the Born-huber cycle for the formation of crystalline potassium fluoride. ...
Atomic Structure/Electrons
... Sections: Atomic Structure Electrons Match the following to the correct letter: ...
... Sections: Atomic Structure Electrons Match the following to the correct letter: ...
HISTORY OF THE ATOM AND ATOMIC THEORY
... through the foil, a few would be slightly deflected. – most went through, but some were deflected by a large amount – this indicated there was a positive charge in the center of the atom, and the center was small compared to the size of the atom ...
... through the foil, a few would be slightly deflected. – most went through, but some were deflected by a large amount – this indicated there was a positive charge in the center of the atom, and the center was small compared to the size of the atom ...
DALTON`S ATOMIC THEORY - 1808: Publication of Dalton`s "A New
... The parts of Dalton's theory Matter is composed of small, chemically indivisible ATOMS ELEMENTS are kinds of matter that contain only a single kind of atom. All the atoms of an element have identical chemical properties. COMPOUNDS are kinds of matter that are composed of atoms of two or more ELEMENT ...
... The parts of Dalton's theory Matter is composed of small, chemically indivisible ATOMS ELEMENTS are kinds of matter that contain only a single kind of atom. All the atoms of an element have identical chemical properties. COMPOUNDS are kinds of matter that are composed of atoms of two or more ELEMENT ...
Atom Internet Scavenger Hunt
... its properties. Within atoms are different parts. These parts relate to patterns in the Periodic table, are instrumental for drawing a Bohr model and a Lewis electron dot diagram. To help you learn more about atoms and their relationship to the Periodic Table, please print out this form then go to e ...
... its properties. Within atoms are different parts. These parts relate to patterns in the Periodic table, are instrumental for drawing a Bohr model and a Lewis electron dot diagram. To help you learn more about atoms and their relationship to the Periodic Table, please print out this form then go to e ...
IPS Unit 2 Worksheet Packet
... The diagrams below show three squares from three different periodic tablse. On each diagram, label the following items: element name ...
... The diagrams below show three squares from three different periodic tablse. On each diagram, label the following items: element name ...
Unit B review - mvhs
... 21. The elements in which of the following have most nearly the same atomic radius? (A) Be, B, C, N (B) Ne, Ar, Kr, Xe (C) Mg, Ca, Sr, Ba (D) C, P, Se, I (E) Cr, Mn, Fe, Co 22. What is the correct order of decreasing first ionization energies for the elements Be, B, and C? (A) Be>B>C (B) B>Be>C (C) ...
... 21. The elements in which of the following have most nearly the same atomic radius? (A) Be, B, C, N (B) Ne, Ar, Kr, Xe (C) Mg, Ca, Sr, Ba (D) C, P, Se, I (E) Cr, Mn, Fe, Co 22. What is the correct order of decreasing first ionization energies for the elements Be, B, and C? (A) Be>B>C (B) B>Be>C (C) ...
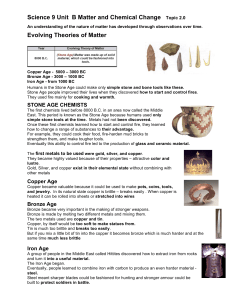
An understanding of the nature of matter has developed
... The number above the element’s symbol on the left is the atomic number. It shows how many protons are in the nucleus of one atom of the element. An oxygen atom, for example, always has eight protons. If you found six protons in an atom, the periodic table would show you that you were looking at carb ...
... The number above the element’s symbol on the left is the atomic number. It shows how many protons are in the nucleus of one atom of the element. An oxygen atom, for example, always has eight protons. If you found six protons in an atom, the periodic table would show you that you were looking at carb ...
Chapter 7 Periodic Properties of the Elements
... • Elements gain or lose electrons to acquire the electron configuration of the noble gases. Periodic Properties of the Elements ...
... • Elements gain or lose electrons to acquire the electron configuration of the noble gases. Periodic Properties of the Elements ...
Atomic Structure
... The atoms of any one element are different from those of any other element. 3. Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. ...
... The atoms of any one element are different from those of any other element. 3. Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. ...
ATOMS / ATOMIC STRUCTURE NOTES Atom
... Atom-the smallest possible piece of an element Proton-the positively charged particle found in the nucleus of an atom Neutron-the neutral (no charge) particle found in the nucleus of an atom Electron- the negatively charged particle found on the outside of the nucleus of an atom Nucleus-the dense co ...
... Atom-the smallest possible piece of an element Proton-the positively charged particle found in the nucleus of an atom Neutron-the neutral (no charge) particle found in the nucleus of an atom Electron- the negatively charged particle found on the outside of the nucleus of an atom Nucleus-the dense co ...
All That Matters - Teach-n-Learn-Chem
... particles as such. However, in all three instances the new information led to an explosion of new thinking and ideas about the nature of matter. How can proper experimental methods prepare a researcher for the event of making an unexpected discovery? ...
... particles as such. However, in all three instances the new information led to an explosion of new thinking and ideas about the nature of matter. How can proper experimental methods prepare a researcher for the event of making an unexpected discovery? ...
Understanding Atomic Structure of an Element
... – Protons: a positively charged particle located inside the nucleus – Neutrons: contain no electrical charge and is located in the nucleus ...
... – Protons: a positively charged particle located inside the nucleus – Neutrons: contain no electrical charge and is located in the nucleus ...
Slide 1 - Effingham County Schools
... chemical reactions. This lead to the discovery of several basic laws. •The law of conservation of mass (matter (mass) is neither created nor destroyed during ordinary physical or chemical changes). •Antoine Lavoisier, a French chemist, verified this law by experimentation in 1789. ...
... chemical reactions. This lead to the discovery of several basic laws. •The law of conservation of mass (matter (mass) is neither created nor destroyed during ordinary physical or chemical changes). •Antoine Lavoisier, a French chemist, verified this law by experimentation in 1789. ...
File - 8th Grade Physical Science
... Look at a copy of the periodic table. The elements which have the same number of electrons in their outer orbitals (valence electrons), are in the same “family” and have similar characteristics, depending on the number. Group 1: 1 valence electron – Alkali Metals Group 2: 2 valence electrons – A ...
... Look at a copy of the periodic table. The elements which have the same number of electrons in their outer orbitals (valence electrons), are in the same “family” and have similar characteristics, depending on the number. Group 1: 1 valence electron – Alkali Metals Group 2: 2 valence electrons – A ...
The discovery of the electron
... Element name Atomic mass Different periodic tables provide different amounts of info and in different orders ...
... Element name Atomic mass Different periodic tables provide different amounts of info and in different orders ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.