Electrone transport chain and oxidative phosphorylation
... form. Rather, they are predominantly found in a ring (cyclic) form. [Note: Pyranose refers to a sixmembered ring consisting of five carbons and one oxygen, whereas furanose denotes a five-membered ring with four carbons and one oxygen.] ...
... form. Rather, they are predominantly found in a ring (cyclic) form. [Note: Pyranose refers to a sixmembered ring consisting of five carbons and one oxygen, whereas furanose denotes a five-membered ring with four carbons and one oxygen.] ...
Insulin and Glucagon
... coming post-absorptive period. Little glucose is normally converted to fat. Note that over-eating carbohydrates (sucrose and fructose) can and does lead to fat production and storage. The kidneys take up about 9-10% of the consumed glucose as lactate which is excreted from red blood cells. RBCs lack ...
... coming post-absorptive period. Little glucose is normally converted to fat. Note that over-eating carbohydrates (sucrose and fructose) can and does lead to fat production and storage. The kidneys take up about 9-10% of the consumed glucose as lactate which is excreted from red blood cells. RBCs lack ...
$doc.title
... How to organisms get energy? • Cells use a high energy molecule to fuel cellular processes Adenosine Triphosphate (ATP) ...
... How to organisms get energy? • Cells use a high energy molecule to fuel cellular processes Adenosine Triphosphate (ATP) ...
Laboratory 3: Biological Molecules
... Laboratory 3: Biological Molecules Today we will be testing a meal to determine which biological components (sugar, starch, or protein) they contain. A. Carbohydrates Carbohydrates contain mostly carbon, hydrogen and oxygen and serve as a source of quick energy for life. The word saccharide comes fr ...
... Laboratory 3: Biological Molecules Today we will be testing a meal to determine which biological components (sugar, starch, or protein) they contain. A. Carbohydrates Carbohydrates contain mostly carbon, hydrogen and oxygen and serve as a source of quick energy for life. The word saccharide comes fr ...
Code Questions Answers 1. Write the reactions of glycolysis
... During the oxidation of glyceralhyde-3-phophate the reducing equivalents are transferred to the acceptor NAD+ (nicotinamide adenine dinucleotide). The reduced NADH under aerobic conditions enter into mitochondria and produces 3 molecules of ATP through its passage into electron transport chain or re ...
... During the oxidation of glyceralhyde-3-phophate the reducing equivalents are transferred to the acceptor NAD+ (nicotinamide adenine dinucleotide). The reduced NADH under aerobic conditions enter into mitochondria and produces 3 molecules of ATP through its passage into electron transport chain or re ...
1-2 (Weigent)
... carboxylase to malonyl CoA. Malonyl CoA is then converted to long chain fatty acids (in the liver, aka liver ketogenesis or Carnitine shuffle). The long chain fatty acids are then converted to triglycerides from which you will then get LDL’s which now are secreted in the blood and transported to the ...
... carboxylase to malonyl CoA. Malonyl CoA is then converted to long chain fatty acids (in the liver, aka liver ketogenesis or Carnitine shuffle). The long chain fatty acids are then converted to triglycerides from which you will then get LDL’s which now are secreted in the blood and transported to the ...
Role of Calcium-Sensing Receptor (CaSR) in pancreatic islet
... in Nuf mice. (Collaboration with MRC Harwell) Negative CaSR allosteric modulators or peptides targeted to the extracellular domain of the CaSR, will be administered by oral gavage or subcutaneously implanted osmotic minipumps to wild-type and affected Nuf mice over a 5-day period and their effects o ...
... in Nuf mice. (Collaboration with MRC Harwell) Negative CaSR allosteric modulators or peptides targeted to the extracellular domain of the CaSR, will be administered by oral gavage or subcutaneously implanted osmotic minipumps to wild-type and affected Nuf mice over a 5-day period and their effects o ...
Respiration - Indian River Research and Education Center
... • First Law of Thermodynamics: –Energy can not be created or destroyed –Thus, total energy at the beginning of a reaction must equal energy at the end ...
... • First Law of Thermodynamics: –Energy can not be created or destroyed –Thus, total energy at the beginning of a reaction must equal energy at the end ...
Cellular respiration
... 2. What is another name for the Aerobic System? 3. Describe the conditions under which an athlete would be relying mostly on the Aerobic System to produced ATP. 4. What must proteins and fats be converted into in order to be used as fuel for the Aerobic System? ...
... 2. What is another name for the Aerobic System? 3. Describe the conditions under which an athlete would be relying mostly on the Aerobic System to produced ATP. 4. What must proteins and fats be converted into in order to be used as fuel for the Aerobic System? ...
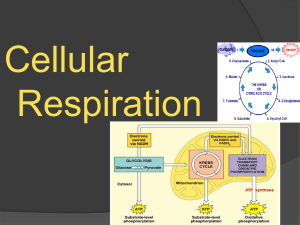
Cellular Respiration
... Energy of products is less than reactants Free energy is released! Cellular respiration The energy from glucose is released and harnessed into ATP at a controlled rate! ...
... Energy of products is less than reactants Free energy is released! Cellular respiration The energy from glucose is released and harnessed into ATP at a controlled rate! ...
PowerPoint 簡報
... tissues and in other organisms that carry out lactic acid fermentation. Acetaldehyde is the product of this reaction. ...
... tissues and in other organisms that carry out lactic acid fermentation. Acetaldehyde is the product of this reaction. ...
Biochemistry - english for biology
... fructose molecule joined together. Another important disaccharide is lactose, consisting of a glucose molecule and a galactose molecule. As most humans age, the production of lactase, the enzyme that hydrolyzes lactose back into glucose and galactose, typically decreases. This results in lactase def ...
... fructose molecule joined together. Another important disaccharide is lactose, consisting of a glucose molecule and a galactose molecule. As most humans age, the production of lactase, the enzyme that hydrolyzes lactose back into glucose and galactose, typically decreases. This results in lactase def ...
THE METABOLISM OF KETONE BODIES
... following a meal containing carbohydrates. • The elevated level of glucose stimulates the secretion of the hormone insulin from the pancreas, which increases the flow of glucose into muscle and adipose tissue for the synthesis of glycogen. • As blood glucose levels drop, the secretion of insulin dec ...
... following a meal containing carbohydrates. • The elevated level of glucose stimulates the secretion of the hormone insulin from the pancreas, which increases the flow of glucose into muscle and adipose tissue for the synthesis of glycogen. • As blood glucose levels drop, the secretion of insulin dec ...
Metabolic Disorders/ Cardiovascular Disease PPAR
... novel potential drug for the treatment of clinical conditions in metabolic and metabolic-related cardiovascular disease. The company has identified novel pan-active molecules that target three separately validated isoforms of PPAR - alpha, delta and gamma - utilizing its proprietary ScaffoldBased Dr ...
... novel potential drug for the treatment of clinical conditions in metabolic and metabolic-related cardiovascular disease. The company has identified novel pan-active molecules that target three separately validated isoforms of PPAR - alpha, delta and gamma - utilizing its proprietary ScaffoldBased Dr ...
Mass-Action Ratios!
... above its "resting" or Keq state, equilibrium can be regained by shifting the reactions to the right (glucose is shipped out, or glycolysis is continued). Similarly, if Glu-1-P builds up, it is too restrictive to simply say the phosphoglucomutase reaction will shift right, when equilibrium can be re ...
... above its "resting" or Keq state, equilibrium can be regained by shifting the reactions to the right (glucose is shipped out, or glycolysis is continued). Similarly, if Glu-1-P builds up, it is too restrictive to simply say the phosphoglucomutase reaction will shift right, when equilibrium can be re ...
Energy Production - University of Massachusetts Amherst
... Amino acids, which combine to form proteins, consist of a carbon backbone and an amine group. If the amine group is removed (deamination) the resulting molecule may enter into the energy producing pathways ...
... Amino acids, which combine to form proteins, consist of a carbon backbone and an amine group. If the amine group is removed (deamination) the resulting molecule may enter into the energy producing pathways ...
Pentose Phosphate Pathway
... Glyceraldehyde‐3‐P and fructose‐6‐P may be converted to glucose‐6‐P, via enzymes of gluconeogenesis, for reentry to Pentose Phosphate Pathway, maximizing formation of NADPH, which is need for reductive biosynthesis. ...
... Glyceraldehyde‐3‐P and fructose‐6‐P may be converted to glucose‐6‐P, via enzymes of gluconeogenesis, for reentry to Pentose Phosphate Pathway, maximizing formation of NADPH, which is need for reductive biosynthesis. ...
Chapter 7 Active Reading Guide
... 23. Oxidative phosphorylation involves two components: the electron transport chain and ATP synthesis. Referring to Figure 7.12, notice that each member of the electron transport chain is lower in free __________ than the preceding member of the chain, but higher in ____________________. The molecul ...
... 23. Oxidative phosphorylation involves two components: the electron transport chain and ATP synthesis. Referring to Figure 7.12, notice that each member of the electron transport chain is lower in free __________ than the preceding member of the chain, but higher in ____________________. The molecul ...
Biology 5.3 Cellular Respiration - Chemistry
... chain, continuing reactions create a large amount of ATP from the materials from stage 2. ...
... chain, continuing reactions create a large amount of ATP from the materials from stage 2. ...
INTRODUCTORY BIOCHEMISTRY Bio. 28 First Midterm
... 32. [6] For an enzyme to be allosterically regulated, it must (choose one or more): a) have a variable Vmax. b) have multiple subunits. c) have multiple substrate binding sites. d) have multiple regulatory domains. e) have two or more binding sites for substrate(s) and/or regulatory effector(s) ...
... 32. [6] For an enzyme to be allosterically regulated, it must (choose one or more): a) have a variable Vmax. b) have multiple subunits. c) have multiple substrate binding sites. d) have multiple regulatory domains. e) have two or more binding sites for substrate(s) and/or regulatory effector(s) ...
The importance of gluconeogenesis as an important
... high levels of AMP, and activated by high levels of ATP & low levels of AMP. It is allosterically inhibited by Fructose 2,6-bisphosphate (reciprocal regulation with glycolysis) (Fructose 1,6 BisphosphateFructose 6-P ) ...
... high levels of AMP, and activated by high levels of ATP & low levels of AMP. It is allosterically inhibited by Fructose 2,6-bisphosphate (reciprocal regulation with glycolysis) (Fructose 1,6 BisphosphateFructose 6-P ) ...
Oxidative degradation of glucose File
... is transferred to ADP by the enzyme pyruvate kinase to generate, at this stage, two molecules of ATP per molecule of glucose oxidized and enolpyruvate is formed. • Enolpyruvate formed is converted spontaneousny to the keto form pyruvate. This is an irreversible step ...
... is transferred to ADP by the enzyme pyruvate kinase to generate, at this stage, two molecules of ATP per molecule of glucose oxidized and enolpyruvate is formed. • Enolpyruvate formed is converted spontaneousny to the keto form pyruvate. This is an irreversible step ...
Glycolysis
... 1) Inherited enzyme deficiencies of glycolysis - Pyruvate kinase deficiency; it genetic deficiency of this enzyme in the erythrocytes lead to hemolytic anemia (excess destruction of RBC) - The normal RBC lacks the mitochondria and it is completely depend on the glycolysis as source of energy. - The ...
... 1) Inherited enzyme deficiencies of glycolysis - Pyruvate kinase deficiency; it genetic deficiency of this enzyme in the erythrocytes lead to hemolytic anemia (excess destruction of RBC) - The normal RBC lacks the mitochondria and it is completely depend on the glycolysis as source of energy. - The ...
Glucose

Glucose is a sugar with the molecular formula C6H12O6. The name ""glucose"" (/ˈɡluːkoʊs/) comes from the Greek word γλευκος, meaning ""sweet wine, must"". The suffix ""-ose"" is a chemical classifier, denoting a carbohydrate. It is also known as dextrose or grape sugar. With 6 carbon atoms, it is classed as a hexose, a sub-category of monosaccharides. α-D-glucose is one of the 16 aldose stereoisomers. The D-isomer (D-glucose) occurs widely in nature, but the L-isomer (L-glucose) does not. Glucose is made during photosynthesis from water and carbon dioxide, using energy from sunlight. The reverse of the photosynthesis reaction, which releases this energy, is a very important source of power for cellular respiration. Glucose is stored as a polymer, in plants as starch and in animals as glycogen.