Fragrant Esters Esters are prepared in a reaction between a
... can prepare esters that produce different scents. Sometimes only small changes are needed to get a completely different fragrance. For example experiment 1 (see below) yields an ester that smells like apple. A slightly longer chain produces an orange scent. Having a branched chain leads to banana oi ...
... can prepare esters that produce different scents. Sometimes only small changes are needed to get a completely different fragrance. For example experiment 1 (see below) yields an ester that smells like apple. A slightly longer chain produces an orange scent. Having a branched chain leads to banana oi ...
Experiment #3: Asymmetric Synthesis – Use of a Chiral Manganese
... For this extremely useful compound, Eric Jacobsen was awarded the 1994 Fluka Prize for Reagent of the Year. The Fluka Prize is awarded annually for a new compound that has been shown to be “a reagent of prime importance, useful in organic chemistry, biochemistry, or analytical chemistry.” Jacobsen’s ...
... For this extremely useful compound, Eric Jacobsen was awarded the 1994 Fluka Prize for Reagent of the Year. The Fluka Prize is awarded annually for a new compound that has been shown to be “a reagent of prime importance, useful in organic chemistry, biochemistry, or analytical chemistry.” Jacobsen’s ...
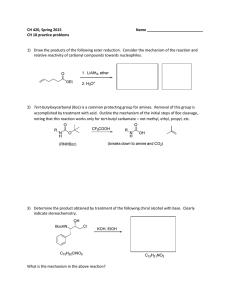
CH 420, Spring 2015 Name ___________________________ CH 18 practice problems
... noting that this reaction works only for tert-butyl carbamate – not methyl, ethyl, propyl, etc. ...
... noting that this reaction works only for tert-butyl carbamate – not methyl, ethyl, propyl, etc. ...
C h e m g u i d e ... ALCOHOLS: MANUFACTURE
... b) How is the ethanol separated from the reaction mixture? c) Give the following conditions for the reaction: (i) the catalyst; (ii) the pressure; (iii) the temperature. d) If you reacted other alkenes with steam under similar conditions, there may appear to be more than one possible alcohol that co ...
... b) How is the ethanol separated from the reaction mixture? c) Give the following conditions for the reaction: (i) the catalyst; (ii) the pressure; (iii) the temperature. d) If you reacted other alkenes with steam under similar conditions, there may appear to be more than one possible alcohol that co ...
review sheet plus practice problems
... What is the name for this alkyl halide / alcohol / ether? Is an alcohol 1°, 2°, or 3°? What are the products of free radical halogenation of an alkane (ex: Cl2/uv light)? Give the chain mechanism for free radical halogenation. What is the selectivity for brominations vs. fluorinations? Why are allyl ...
... What is the name for this alkyl halide / alcohol / ether? Is an alcohol 1°, 2°, or 3°? What are the products of free radical halogenation of an alkane (ex: Cl2/uv light)? Give the chain mechanism for free radical halogenation. What is the selectivity for brominations vs. fluorinations? Why are allyl ...
Answers to “Tragic Flaws” 1. Can`t form an ether using an alcohol
... using POCl3 with pyridine – no control over which way the double bond will form – both sides would form a trisubstituted alkene. Multiple products! 13. Grignard additions form alcohols. Not ketones. Missing an oxidation in the sequence. 14. Tragic flaws all over the place. Need ketone FIRST to do co ...
... using POCl3 with pyridine – no control over which way the double bond will form – both sides would form a trisubstituted alkene. Multiple products! 13. Grignard additions form alcohols. Not ketones. Missing an oxidation in the sequence. 14. Tragic flaws all over the place. Need ketone FIRST to do co ...
... were higher with electron-rich arenes (R1 = Me, OMe) compared to electron-poor ones (R1 = Hal, CF3). The EWG on the alkene is required since only the uncyclized olefins 4 were isolated with electron-neutral alkenes (R4 = alkyl, aryl). The methodology was extended to secondary and primary alcohols (5 ...
Exam 3 Review
... The third exam will be on Wed, July 29, covering material in Chapter 17 – 19 of the text. The test will focus on chemistry of alcohols, ketones, aldehydes, and organic synthesis. The following textbook sections will be covered on the exam: 17.1 to 17.9 17.12 (NMR of alcohols only) (skip sections 17. ...
... The third exam will be on Wed, July 29, covering material in Chapter 17 – 19 of the text. The test will focus on chemistry of alcohols, ketones, aldehydes, and organic synthesis. The following textbook sections will be covered on the exam: 17.1 to 17.9 17.12 (NMR of alcohols only) (skip sections 17. ...
Oxidation of Alcohols
... using an oxidising agent, notated by [o]. • A suitable oxidising agent is a solution containing acidified dichromate ions (H+ and Cr O 2-). • These ions come from a mixture of K Cr O and sulphuric acid. • During the reaction there will be a colour change of orange to green. ...
... using an oxidising agent, notated by [o]. • A suitable oxidising agent is a solution containing acidified dichromate ions (H+ and Cr O 2-). • These ions come from a mixture of K Cr O and sulphuric acid. • During the reaction there will be a colour change of orange to green. ...
Group Activity 3 [10 PTS]
... 1. Write the condensed structural formula of each of the following alcohols a. 1-propanol ...
... 1. Write the condensed structural formula of each of the following alcohols a. 1-propanol ...
... around the ring. Yields are excellent (>75%) in almost all cases, except for R3 = CO2Et (50%) and R3 = H (42%). The stability of halogen substituents (R1 = Cl, Br) to the reaction conditions, providing a handle for further functionalization, is worthy of note. Two examples of spirocyclic dihydrobenz ...
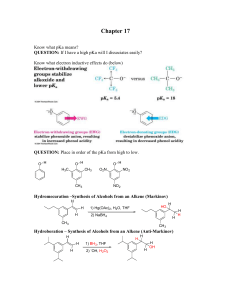
Chapter 17 - Ellis Benjamin
... Chapter 17 Know what pKa means? QUESTION: If I have a high pKa will I dissociates easily? Know what electron inductive effects do (below) ...
... Chapter 17 Know what pKa means? QUESTION: If I have a high pKa will I dissociates easily? Know what electron inductive effects do (below) ...
2010-09-16 Alcohols
... look so long and so regretfully upon the closed door that we do not see the one which has opened for us.” – Alexander Graham Bell ...
... look so long and so regretfully upon the closed door that we do not see the one which has opened for us.” – Alexander Graham Bell ...
packet 1, 2325 key
... products A-C. The IRs are shown for products A-C. The value of the molecular ion peak is given along with the intensity ratio (relative to the parent peak) of the M+2 peak. Indicate the structure of each product. ...
... products A-C. The IRs are shown for products A-C. The value of the molecular ion peak is given along with the intensity ratio (relative to the parent peak) of the M+2 peak. Indicate the structure of each product. ...
Alcohol, Aldehydes and Acids
... Ethanol Ethanol, when fermented from sugar, is the alcohol in beverages. It can also be made from ethene by the addition of water for nonbeverage use, like an additive to gassoline to make "gasahol." 2-Propanol (better known as isopropyl alcohol) is in (with some water) rubbing alcohol. It is also ...
... Ethanol Ethanol, when fermented from sugar, is the alcohol in beverages. It can also be made from ethene by the addition of water for nonbeverage use, like an additive to gassoline to make "gasahol." 2-Propanol (better known as isopropyl alcohol) is in (with some water) rubbing alcohol. It is also ...
Homework #7, Graded Answers
... e.) 2-methyl-2-pentanol is subjected to oxidation No reaction tertiary alcohols do not oxidize 31.) Each of the following conversions requires more than one step. Show the reagents you would use and draw structural formulas for intermediate compounds formed in each conversion. O ...
... e.) 2-methyl-2-pentanol is subjected to oxidation No reaction tertiary alcohols do not oxidize 31.) Each of the following conversions requires more than one step. Show the reagents you would use and draw structural formulas for intermediate compounds formed in each conversion. O ...
Abdul Majeed Seayad Project Synopsis (96 - ACE
... borrowing strategy: Alcohols as feedstock, for useful transformations without functional group readily available nontoxic feedstock would electrophiles in C-N and C-C bond interconversions is a challenge in organic and process chemistry. In a greatly impact the way in which APIs are forming reaction ...
... borrowing strategy: Alcohols as feedstock, for useful transformations without functional group readily available nontoxic feedstock would electrophiles in C-N and C-C bond interconversions is a challenge in organic and process chemistry. In a greatly impact the way in which APIs are forming reaction ...
DEHYDRATION - ALKENE TEST EXERCISES
... DEHYDRATION - ALKENE TEST EXERCISES 1. Give a detailed mechanism for the acid-catalyzed dehydration of cyclohexanol to cyclohexene. ...
... DEHYDRATION - ALKENE TEST EXERCISES 1. Give a detailed mechanism for the acid-catalyzed dehydration of cyclohexanol to cyclohexene. ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.









![Group Activity 3 [10 PTS]](http://s1.studyres.com/store/data/010780770_1-3445600a9b56e890a0f283c789afe8fb-300x300.png)