Alcohols - Miller, Jonathan
... Esters are fragrant smelling liquids used in perfumes and fruit flavourings (often larger molecular masses) formed when an alcohol reacts with a carboxylic acid in the presence of an acid catalyst (small amount of conc. H2SO4). It is an equilibrium reaction. ...
... Esters are fragrant smelling liquids used in perfumes and fruit flavourings (often larger molecular masses) formed when an alcohol reacts with a carboxylic acid in the presence of an acid catalyst (small amount of conc. H2SO4). It is an equilibrium reaction. ...
Dess-Martin Oxidation
... • Dess-Martin periodinane (DMP) is a chemical reagent used to oxidize primary alcohols to aldehydes and secondary alcohols to ketones ...
... • Dess-Martin periodinane (DMP) is a chemical reagent used to oxidize primary alcohols to aldehydes and secondary alcohols to ketones ...
Test Review
... You should be able to explain the structure and properties of alcohols, phenols, ethers, and thiols. ...
... You should be able to explain the structure and properties of alcohols, phenols, ethers, and thiols. ...
exam qs on alcohols, alkenes, esters
... give two products which are structurally isomeric. Draw structural formulae for these two structural isomers and explain why the formation of two structural isomers is possible in this case. Alcohol selected ...
... give two products which are structurally isomeric. Draw structural formulae for these two structural isomers and explain why the formation of two structural isomers is possible in this case. Alcohol selected ...
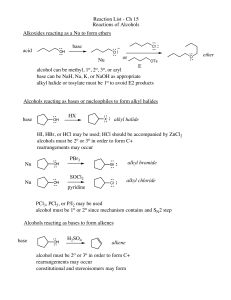
Reaction List - Ch 15 Reactions of Alcohols Alkoxides reacting as a
... Reaction List - Ch 15 Reactions of Alcohols Alkoxides reacting as a Nu to form ethers base ...
... Reaction List - Ch 15 Reactions of Alcohols Alkoxides reacting as a Nu to form ethers base ...
Review Chapter 19
... Protection of alcohols Alcohols are a common organic functional group. The prevalence of this group is in no small part due to its myriad of reaction pathways. Alcohols are weak acids, weak bases, nucleophiles and electrophiles. Sometimes it is necessary to cover over this react ...
... Protection of alcohols Alcohols are a common organic functional group. The prevalence of this group is in no small part due to its myriad of reaction pathways. Alcohols are weak acids, weak bases, nucleophiles and electrophiles. Sometimes it is necessary to cover over this react ...
Formation of C-C Bonds via Catalytic Hydrogenation and Transfer
... Abstract: Carbon-carbon bond formation lies at the heart of chemical synthesis. Research in the Krische laboratory reveals reductive C-C bond formation can be achieved under the conditions of catalytic hydrogenation. This concept is extended further via “C-C bond forming transfer hydrogenations”, wh ...
... Abstract: Carbon-carbon bond formation lies at the heart of chemical synthesis. Research in the Krische laboratory reveals reductive C-C bond formation can be achieved under the conditions of catalytic hydrogenation. This concept is extended further via “C-C bond forming transfer hydrogenations”, wh ...
L-13
... the reaction and led to the production of allylated product 3a in 80% yield (entry 2).[6] Strong Lewis acids such as AlCl3 or BF3・OEt2 were not effective for the allylation (entries 3 and 4), probably because these catalysts are not stable under protic conditions. Sc(OTf)3 only gave a low yield of 3 ...
... the reaction and led to the production of allylated product 3a in 80% yield (entry 2).[6] Strong Lewis acids such as AlCl3 or BF3・OEt2 were not effective for the allylation (entries 3 and 4), probably because these catalysts are not stable under protic conditions. Sc(OTf)3 only gave a low yield of 3 ...
C h e m g u i d e ... ALDEHYDES AND KETONES: REDUCTION
... things get more complicated, it is easier to show the structures more fully.) ...
... things get more complicated, it is easier to show the structures more fully.) ...
Topic: Functional Grp # 2: Alcohol
... • Alcohols: higher bp than corresponding alkane • The bigger the molecule the higher the • ‘Like dissolves Like’ – Alcohols tend to be very soluble in water ...
... • Alcohols: higher bp than corresponding alkane • The bigger the molecule the higher the • ‘Like dissolves Like’ – Alcohols tend to be very soluble in water ...
Discussion Sheet 11
... Problem 3. Use problems 1 and 2 as a flow chart to propose two step transformations with alcohols as synthetic intermediates.: ...
... Problem 3. Use problems 1 and 2 as a flow chart to propose two step transformations with alcohols as synthetic intermediates.: ...
Enantiodivergent conversion of chiral secondary alcohols into
... •Summary (comparison to other methods) ...
... •Summary (comparison to other methods) ...
Functional Groups
... Functional Groups A specific arrangement of atoms in an organic compound that is capable of characteristic chemical reactions. In other words, a substituent group other than an alkyl group. Most organic chemistry is functionalgroup chemistry. We will do one functional group. ...
... Functional Groups A specific arrangement of atoms in an organic compound that is capable of characteristic chemical reactions. In other words, a substituent group other than an alkyl group. Most organic chemistry is functionalgroup chemistry. We will do one functional group. ...
F:\CH 361 2014\Prelab 6 2011 CH361 modified for 2014.wpd
... organic chemistry textbook and the rest of experiment II in your lab manual. This prelab has two pages. ...
... organic chemistry textbook and the rest of experiment II in your lab manual. This prelab has two pages. ...
Chem 30CL - Lecture 1c - UCLA Chemistry and Biochemistry
... How do Chemists control Chirality? • Chiral pool: optically active compounds that can be isolated from natural sources (i.e., amino acids, monosaccharides, terpenes, etc.) and can be used as reactants or as part of a chiral catalyst or a chiral auxiliary • The TADDOL, DIOP and the Chiraphos ligand ...
... How do Chemists control Chirality? • Chiral pool: optically active compounds that can be isolated from natural sources (i.e., amino acids, monosaccharides, terpenes, etc.) and can be used as reactants or as part of a chiral catalyst or a chiral auxiliary • The TADDOL, DIOP and the Chiraphos ligand ...
alcohols-II-12-ques
... RCH2OH + PDC [(C5H5NH+)2 Cr2O72–] A) The alcohol is oxidized to an acid, and the Cr(VI) is reduced. B) The alcohol is oxidized to an aldehyde, and the Cr(VI) is reduced. C) The alcohol is reduced to an aldehyde, and the Cr(III) is oxidized. D) The alcohol is oxidized to a ketone, and the Cr(VI) is ...
... RCH2OH + PDC [(C5H5NH+)2 Cr2O72–] A) The alcohol is oxidized to an acid, and the Cr(VI) is reduced. B) The alcohol is oxidized to an aldehyde, and the Cr(VI) is reduced. C) The alcohol is reduced to an aldehyde, and the Cr(III) is oxidized. D) The alcohol is oxidized to a ketone, and the Cr(VI) is ...
Review and New - ChemConnections
... • What is the product of the reaction of 1butanol with PCC in CH2Cl2? ...
... • What is the product of the reaction of 1butanol with PCC in CH2Cl2? ...
Chap Thirteen: Alcohols
... its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and reactivity. Predict the reagents needed to produce a given product from an alcohol in one or ...
... its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and reactivity. Predict the reagents needed to produce a given product from an alcohol in one or ...
CHAPTER 10 Properties and Preparation of Alcohols
... Synthesis of Alcohols (Review) • Nucleophilic substitution (usually SN2) of alkyl halide. • Alkene Addition: – Water in acid solution (suffers from rearrangements). – Oxymercuration–demercuration. – Hydroboration–oxidation. ...
... Synthesis of Alcohols (Review) • Nucleophilic substitution (usually SN2) of alkyl halide. • Alkene Addition: – Water in acid solution (suffers from rearrangements). – Oxymercuration–demercuration. – Hydroboration–oxidation. ...
Outline_CH13_Klein
... its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and reactivity. Predict the reagents needed to produce a given product from an alcohol in one or ...
... its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and reactivity. Predict the reagents needed to produce a given product from an alcohol in one or ...
chemistry 2 - waiukucollegescience
... In order to distinguish between propan-1-ol and propene a student said it was necessary to use bromine water rather than acidified potassium permanganate. Discuss this statement. ...
... In order to distinguish between propan-1-ol and propene a student said it was necessary to use bromine water rather than acidified potassium permanganate. Discuss this statement. ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.