Chemistry IGCSE
... Often it does not matter if a substance is not pure. We wash in tap water; without thinking too much about what is in it, but sometimes purity is very important. If you are making a new medical drug, or a flavouring for food, you must make sure it contains nothing that could harm people. An unwanted ...
... Often it does not matter if a substance is not pure. We wash in tap water; without thinking too much about what is in it, but sometimes purity is very important. If you are making a new medical drug, or a flavouring for food, you must make sure it contains nothing that could harm people. An unwanted ...
www.fahadsacademy.com
... 1. Ionic compounds are hard crystalline solids with flat sides and regular shapes because the ions are arrnged in straight rows in strong ionic bonds. 2. Ionic compounds have very high melting points and boiling points. 3. The strong forces holding ionic compounds prevents them to evaporate easily. ...
... 1. Ionic compounds are hard crystalline solids with flat sides and regular shapes because the ions are arrnged in straight rows in strong ionic bonds. 2. Ionic compounds have very high melting points and boiling points. 3. The strong forces holding ionic compounds prevents them to evaporate easily. ...
Chemical Reactions
... Identify the type of reaction for each of the following synthesis or decomposition reactions, and write the balanced equation: Nitrogen and oxygen react to form nitrogen ...
... Identify the type of reaction for each of the following synthesis or decomposition reactions, and write the balanced equation: Nitrogen and oxygen react to form nitrogen ...
21 More About Amines • Heterocyclic Compounds
... Amines are also exceedingly important compounds to organic chemists, far too important to leave until the end of a course in organic chemistry. We have, therefore, already studied many aspects of amines and their chemistry. For example, we have seen that the nitrogen in amines is sp 3 hybridized and ...
... Amines are also exceedingly important compounds to organic chemists, far too important to leave until the end of a course in organic chemistry. We have, therefore, already studied many aspects of amines and their chemistry. For example, we have seen that the nitrogen in amines is sp 3 hybridized and ...
105 ACID - DW Brooks
... Operational Definitions. Acids and bases are chemical species that exhibit distinctive sets of observable properties. Acids taste sour (like vinegar and lemon), cause blue litmus to turn red, liberate hydrogen gas when they react with certain metals (like iron, zinc, and aluminum), and neutralize ba ...
... Operational Definitions. Acids and bases are chemical species that exhibit distinctive sets of observable properties. Acids taste sour (like vinegar and lemon), cause blue litmus to turn red, liberate hydrogen gas when they react with certain metals (like iron, zinc, and aluminum), and neutralize ba ...
Answers - U of L Class Index
... Why would BeCl2 be a molecular compound when all of the other alkaline earth metals react with chlorine gas to give ionic compounds? [2 marks] The Be2+ cation is very small. Therefore, its charge density is much higher than any other M2+ cation formed from an alkaline earth metal. This makes Be2+ a ...
... Why would BeCl2 be a molecular compound when all of the other alkaline earth metals react with chlorine gas to give ionic compounds? [2 marks] The Be2+ cation is very small. Therefore, its charge density is much higher than any other M2+ cation formed from an alkaline earth metal. This makes Be2+ a ...
chem A exercise package C
... process of overlapping atoms is called covalent bonding. The substance that results from covalent bonding is called a covalent substance. The process of overlapping atoms will keep occurring for a particular atom until it has a stable number of electrons in its original boundary. In general, covalen ...
... process of overlapping atoms is called covalent bonding. The substance that results from covalent bonding is called a covalent substance. The process of overlapping atoms will keep occurring for a particular atom until it has a stable number of electrons in its original boundary. In general, covalen ...
Kinetics and Mechanism of Uncatalyzed and Ag (I) Catalyzed
... sulphuric acid medium and sulphato complexes, such as CeSO42+, Ce(SO4)2 and Ce(SO4)32- have been established and quantified [26]. However, cerium (IV) in perchloric acid medium does not indicate complex formation, although Ce4+, Ce(OH)3+, (Ce-O-Ce)6+ and (HOCe-O-CeOH)4+ species of cerium (IV) are we ...
... sulphuric acid medium and sulphato complexes, such as CeSO42+, Ce(SO4)2 and Ce(SO4)32- have been established and quantified [26]. However, cerium (IV) in perchloric acid medium does not indicate complex formation, although Ce4+, Ce(OH)3+, (Ce-O-Ce)6+ and (HOCe-O-CeOH)4+ species of cerium (IV) are we ...
Shifting Equilibrium
... Formation of a Gas Reactions that form a gas as a product are one example of reactions that go to completion. When a strong acid is added to an aqueous solution of baking soda, or sodium bicarbonate, carbon dioxide is formed. The net ionic equation shows that ions are removed. H3O+ + HCO 3 → 2 ...
... Formation of a Gas Reactions that form a gas as a product are one example of reactions that go to completion. When a strong acid is added to an aqueous solution of baking soda, or sodium bicarbonate, carbon dioxide is formed. The net ionic equation shows that ions are removed. H3O+ + HCO 3 → 2 ...
November 2016 (v1) QP - Paper 4 CIE Chemistry A-level
... What do these Kstab values tell us about the relative positions of equilibria 1 and 2? ...
... What do these Kstab values tell us about the relative positions of equilibria 1 and 2? ...
November 2016 (v3) QP - Paper 4 CIE Chemistry A-level
... What do these Kstab values tell us about the relative positions of equilibria 1 and 2? ...
... What do these Kstab values tell us about the relative positions of equilibria 1 and 2? ...
Alcohols
... Named by adding -thiol to alkane name. The -SH group is called mercapto. Complex with heavy metals: Hg, As, Au. More acidic than alcohols, react with NaOH to form thiolate ion. ...
... Named by adding -thiol to alkane name. The -SH group is called mercapto. Complex with heavy metals: Hg, As, Au. More acidic than alcohols, react with NaOH to form thiolate ion. ...
Experiment 11 CHEMICAL REACTIONS
... Get a “gas collecting bottle” from the reagent bench and take it and your cover glass to the hood where the instructor will ignite a sample of phosphorus in a “def lagrating spoon” and lower it briefly into your gas bottle. As soon as the spoon is removed, rapidly place the cover glass over the mout ...
... Get a “gas collecting bottle” from the reagent bench and take it and your cover glass to the hood where the instructor will ignite a sample of phosphorus in a “def lagrating spoon” and lower it briefly into your gas bottle. As soon as the spoon is removed, rapidly place the cover glass over the mout ...
B.Sc. (CHEMISTRY) - Dr B. R. Ambedkar University
... Distillation of nitrobenzene and aniline using air condenser. Crystallization: Concept of induction of crystallization. Phthalic acid from hot water (using fluted filter paper and steamless funnel). Acetanilide from boiling water. Naphthalene from ethanol. Benzoic acid from water. Decolorisation and ...
... Distillation of nitrobenzene and aniline using air condenser. Crystallization: Concept of induction of crystallization. Phthalic acid from hot water (using fluted filter paper and steamless funnel). Acetanilide from boiling water. Naphthalene from ethanol. Benzoic acid from water. Decolorisation and ...
Chemistry English
... Atomic theory: if the matter were divided a sufficient number of times, it could eventually be reduced to the indivisible, indestructible particles called atom. The atomic theory was presented by the British chemist John Dalton (1766-1844) in the early 1800s. It is one of the greatest advances in th ...
... Atomic theory: if the matter were divided a sufficient number of times, it could eventually be reduced to the indivisible, indestructible particles called atom. The atomic theory was presented by the British chemist John Dalton (1766-1844) in the early 1800s. It is one of the greatest advances in th ...
Class_X–Science__term_I
... 4) The chemical reactions can be classified into different types such as— a) Combination reactions:-The reactions in which two or more substances combine to form a new substance are called combination reaction. For example, 2Mg(s) + O2 (g) 2 MgO (s) b) Decomposition reaction - The reaction in whic ...
... 4) The chemical reactions can be classified into different types such as— a) Combination reactions:-The reactions in which two or more substances combine to form a new substance are called combination reaction. For example, 2Mg(s) + O2 (g) 2 MgO (s) b) Decomposition reaction - The reaction in whic ...
SOLUBILITY RULES FOR IONIC COMPOUNDS IN WATER
... (a) An acidified solution of hydrogen peroxide is added to a solution of sodium iodide. (b) Chlorine gas is passed over powdered aluminum. (c) Solutons of mercury (I) nitrate and potassium sulfate are mixed. (d) A strip of magnesium metal is added to a solution of silver nitrate. (e) Solutions of le ...
... (a) An acidified solution of hydrogen peroxide is added to a solution of sodium iodide. (b) Chlorine gas is passed over powdered aluminum. (c) Solutons of mercury (I) nitrate and potassium sulfate are mixed. (d) A strip of magnesium metal is added to a solution of silver nitrate. (e) Solutions of le ...
1994 AP Chemistry Multiple Choice
... 54. All of the following statements concerning the characteristics of the halogens are true EXCEPT: (A) The first ionization energies (potentials) decrease as the atomic numbers of the halogens increase. (B) Fluorine is the best oxidizing agent. (C) Fluorine atoms have the smallest radii. (D) Iodine ...
... 54. All of the following statements concerning the characteristics of the halogens are true EXCEPT: (A) The first ionization energies (potentials) decrease as the atomic numbers of the halogens increase. (B) Fluorine is the best oxidizing agent. (C) Fluorine atoms have the smallest radii. (D) Iodine ...
Combining the Benefits of Homogeneous and Heterogeneous
... such as acetonitrile, dioxane, and THF that can be used for homogeneously catalyzed reactions. Modest pressures of a soluble gas, generally CO2, achieve facile post-reaction heterogeneous separation of products from the catalyst. Examples shown here are rhodiumcatalyzed hydroformylation of 1-octene ...
... such as acetonitrile, dioxane, and THF that can be used for homogeneously catalyzed reactions. Modest pressures of a soluble gas, generally CO2, achieve facile post-reaction heterogeneous separation of products from the catalyst. Examples shown here are rhodiumcatalyzed hydroformylation of 1-octene ...
Chemistry Unit 3 Holiday Homework Questions
... 15. 25.0 Ml of arsenic(III) chloride solution is reacted with an excess of 0.200 M NaOH solution, forming 3.15 g of precipitate of arsenic hydroxide. AsCl3(aq) + 3NaOH(aq) As(OH)3(s) + 3NaCl(aq) (a) Find the number of moles of arsenic hydroxide. Ans: 0.025mol (b) Find the number of moles of sodium ...
... 15. 25.0 Ml of arsenic(III) chloride solution is reacted with an excess of 0.200 M NaOH solution, forming 3.15 g of precipitate of arsenic hydroxide. AsCl3(aq) + 3NaOH(aq) As(OH)3(s) + 3NaCl(aq) (a) Find the number of moles of arsenic hydroxide. Ans: 0.025mol (b) Find the number of moles of sodium ...
Chapter 6 Chemical reactions Classification And Mass Relationships
... • An atom in its neutral state has an oxidation number of zero. • A monoatomic ion has an oxidation number equal to its charge. • In a molecular compound the atom usually has the same oxidation number it would have it were a monoatomic ion. • The sum of oxidation numbers in a neutral compound is ze ...
... • An atom in its neutral state has an oxidation number of zero. • A monoatomic ion has an oxidation number equal to its charge. • In a molecular compound the atom usually has the same oxidation number it would have it were a monoatomic ion. • The sum of oxidation numbers in a neutral compound is ze ...
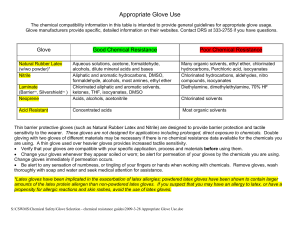
Appropriate Glove Use
... Aliphatic and aromatic hydrocarbons, DMSO, formaldehyde, alcohols, most amines, ethyl ether Chlorinated aliphatic and aromatic solvents, ketones, THF, isocyanates, DMSO Acids, alcohols, acetonitrile ...
... Aliphatic and aromatic hydrocarbons, DMSO, formaldehyde, alcohols, most amines, ethyl ether Chlorinated aliphatic and aromatic solvents, ketones, THF, isocyanates, DMSO Acids, alcohols, acetonitrile ...
Amines
... Amines have an sp3 hybridized nitrogen In principle, tertiary amines with three different R groups should be chiral (i.e., have a stereocenter). However, rapid pyramidal inversion of the amine nitrogen prevents isolation of the enantiomers except where the nitrogen is part of a ring or has other geo ...
... Amines have an sp3 hybridized nitrogen In principle, tertiary amines with three different R groups should be chiral (i.e., have a stereocenter). However, rapid pyramidal inversion of the amine nitrogen prevents isolation of the enantiomers except where the nitrogen is part of a ring or has other geo ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.