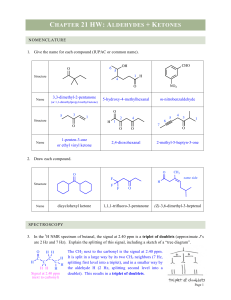
Test - Angelfire
... 4. A calculator is essential for the Chemistry 12 Provincial Examination. The calculator must be a hand-held device designed primarily for mathematical computations involving logarithmic and trigonometric functions and may also include graphing functions. Computers, calculators with a QWERTY keyboar ...
... 4. A calculator is essential for the Chemistry 12 Provincial Examination. The calculator must be a hand-held device designed primarily for mathematical computations involving logarithmic and trigonometric functions and may also include graphing functions. Computers, calculators with a QWERTY keyboar ...
Gas Laws
... 1. Ideal combustion, resulting in only carbon dioxide and water, rarely happens. In general, at the very least, some carbon monoxide is also produced. The real combustion of methane is more closely represented, then, by the unbalanced equation CH4 + O2 H2O + CO2 + CO. If 0.100 mol CH4 is allowed t ...
... 1. Ideal combustion, resulting in only carbon dioxide and water, rarely happens. In general, at the very least, some carbon monoxide is also produced. The real combustion of methane is more closely represented, then, by the unbalanced equation CH4 + O2 H2O + CO2 + CO. If 0.100 mol CH4 is allowed t ...
Processes for making sugar and/or sugar alcohol dehydration
... catalyst and to substantially separate the one or more dehy dration products from the residual sugars, so that the residual sugars can be put to further productive use as indicated here after. ...
... catalyst and to substantially separate the one or more dehy dration products from the residual sugars, so that the residual sugars can be put to further productive use as indicated here after. ...
Ch-6-Alcohols and phenols - Home
... • Phenol is a crystalline solid, with a higher boiling point than alcohols, and it is moderately soluble in water. • It is slightly acidic: the phenol molecule has weak tendencies to lose the H+ ion from the hydroxyl group, resulting in the highly water-soluble phenolate anion C6H5O−, called phenoxi ...
... • Phenol is a crystalline solid, with a higher boiling point than alcohols, and it is moderately soluble in water. • It is slightly acidic: the phenol molecule has weak tendencies to lose the H+ ion from the hydroxyl group, resulting in the highly water-soluble phenolate anion C6H5O−, called phenoxi ...
p-BLOCK ELEMENTS - einstein classes
... Fluoboric acid is a strong acid. The [BF4]— ion is tetrahedral, and fluoborates resemble perchlorates ClO4— and sulphates in crystal structure and solubility (KClO4 and KBF4 are both not very soluble in water). Trihalides of Boron The boron halides are covalent. BF3 is gaseous, BCl3 liquid and BI3 i ...
... Fluoboric acid is a strong acid. The [BF4]— ion is tetrahedral, and fluoborates resemble perchlorates ClO4— and sulphates in crystal structure and solubility (KClO4 and KBF4 are both not very soluble in water). Trihalides of Boron The boron halides are covalent. BF3 is gaseous, BCl3 liquid and BI3 i ...
Chemistry
... need for students to develop skills that will be of long term value in an increasingly technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the teaching and learning ...
... need for students to develop skills that will be of long term value in an increasingly technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the teaching and learning ...
Deans Community High School Intermediate 2 Revision Notes www
... collide in the air very frequently, but do not normally undergo a chemical reaction as there is insufficient energy in the collisions. Factors affecting reaction rate - Surface area If a large piece of material (1 particle) has a size of 2cm x 2cm x 2cm, the surface area of the reactant will be 24cm ...
... collide in the air very frequently, but do not normally undergo a chemical reaction as there is insufficient energy in the collisions. Factors affecting reaction rate - Surface area If a large piece of material (1 particle) has a size of 2cm x 2cm x 2cm, the surface area of the reactant will be 24cm ...
Active Learning Questions
... 31. Of the hydrogen halides, only HF is a weak acid. Give a possible explanation. 32. Explain why the following are done, both of which are related to acid–base chemistry. a. Power plants burning coal with high sulfur content use scrubbers to help eliminate sulfur emissions. b. A gardener mixes lime ...
... 31. Of the hydrogen halides, only HF is a weak acid. Give a possible explanation. 32. Explain why the following are done, both of which are related to acid–base chemistry. a. Power plants burning coal with high sulfur content use scrubbers to help eliminate sulfur emissions. b. A gardener mixes lime ...
MOLECULAR REPRESENTATIONS AND INFRARED
... C=O: 1630-1780 cm-1, strong absorption If you need to use other frequencies to identify other functional groups (and sometimes you will), a table of IR frequencies will be provided. ...
... C=O: 1630-1780 cm-1, strong absorption If you need to use other frequencies to identify other functional groups (and sometimes you will), a table of IR frequencies will be provided. ...
1. Write the balanced equation for the combustion of butane (C4H10
... 7. Molten iron and carbon monoxide are produced in a blast furnace by the reaction of iron(III) oxide and coke (pure carbon). If 25.0 kilograms of pure Fe2O3 is used, how many kilograms of iron can be produced? The reaction is: Fe2O3 + 3 C ---> 2 Fe + 3 CO 8. The average human requires 120.0 grams o ...
... 7. Molten iron and carbon monoxide are produced in a blast furnace by the reaction of iron(III) oxide and coke (pure carbon). If 25.0 kilograms of pure Fe2O3 is used, how many kilograms of iron can be produced? The reaction is: Fe2O3 + 3 C ---> 2 Fe + 3 CO 8. The average human requires 120.0 grams o ...
THE STUDY OF INTERMEDIARY METABOLISM OF
... by this procedure (60, 61). The method introduces deuterium into fatty acids only at the a-carbon atom. Another catalyst is active platinum (62, 63, 64, 65). A number of deutero-fatty acids, otherwise not easily obtainable, such as palmitic acid, lauric acid, capric, caprylic acid (61), and iso-capr ...
... by this procedure (60, 61). The method introduces deuterium into fatty acids only at the a-carbon atom. Another catalyst is active platinum (62, 63, 64, 65). A number of deutero-fatty acids, otherwise not easily obtainable, such as palmitic acid, lauric acid, capric, caprylic acid (61), and iso-capr ...
Ordinary Level - State Examination Commission
... of which is the element carbon. Coal often contains small quantities of sulfur in the form of iron sulfide (FeS). Name the two oxides of carbon that can be produced when coal is burned. Which one of these oxides is acidic? Figure 17 ...
... of which is the element carbon. Coal often contains small quantities of sulfur in the form of iron sulfide (FeS). Name the two oxides of carbon that can be produced when coal is burned. Which one of these oxides is acidic? Figure 17 ...
Cardiovascular - PHARMACEUTICAL REVIEW
... cherry red cyanosis seen in CO toxicity); carboxy Hb is incapable of carrying O2 to tissues. ...
... cherry red cyanosis seen in CO toxicity); carboxy Hb is incapable of carrying O2 to tissues. ...
Document
... Balancing the Equation for a Redox Reaction in Acidic Solution. The reaction described below is used to determine the sulfite ion concentration present in wastewater from a papermaking plant. Write the balanced equation for this reaction in acidic solution.. ...
... Balancing the Equation for a Redox Reaction in Acidic Solution. The reaction described below is used to determine the sulfite ion concentration present in wastewater from a papermaking plant. Write the balanced equation for this reaction in acidic solution.. ...
Lab 1
... Primary substances, called elements, build all the materials about you. Some look similar, but others look unlike anything else. In this experiment, you will describe the physical properties of elements in a laboratory display and determine the location of elements on a blank periodic table. A. Phys ...
... Primary substances, called elements, build all the materials about you. Some look similar, but others look unlike anything else. In this experiment, you will describe the physical properties of elements in a laboratory display and determine the location of elements on a blank periodic table. A. Phys ...
Gas Laws
... 1. Ideal combustion, resulting in only carbon dioxide and water, rarely happens. In general, at the very least, some carbon monoxide is also produced. The real combustion of methane is more closely represented, then, by the unbalanced equation CH4 + O2 H2O + CO2 + CO. If 0.100 mol CH4 is allowed t ...
... 1. Ideal combustion, resulting in only carbon dioxide and water, rarely happens. In general, at the very least, some carbon monoxide is also produced. The real combustion of methane is more closely represented, then, by the unbalanced equation CH4 + O2 H2O + CO2 + CO. If 0.100 mol CH4 is allowed t ...
Topic 14 - Fertilisers
... Ammonia has a characteristic pungent smell. Ammonia is very soluble in water as shown by the fountain experiment. ...
... Ammonia has a characteristic pungent smell. Ammonia is very soluble in water as shown by the fountain experiment. ...
Chemistry 11 Review
... 8. Which of the following reactions is most likely to have the greatest rate at room temperature? a) Ag+(aq) + I- (aq) AgI(s) b) H2 (g) + Cl2 (g) 2HCl (g) c) C3H8 (g) + 5O2 (g) 3CO2 (g) + 4H2O (g) ...
... 8. Which of the following reactions is most likely to have the greatest rate at room temperature? a) Ag+(aq) + I- (aq) AgI(s) b) H2 (g) + Cl2 (g) 2HCl (g) c) C3H8 (g) + 5O2 (g) 3CO2 (g) + 4H2O (g) ...
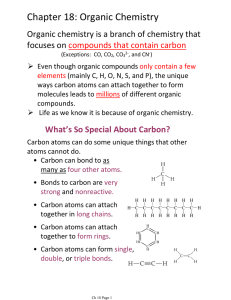
Chapter 18: Organic Chemistry
... 1. Select the longest carbon chain and name it as the normal straight-chain alkane (this is the "parent chain") 2. Number the parent chain starting at the end of the chain nearest the first alkyl substituent. 3. Use the numbers obtained by the application of rule #2 to designate the locations of the ...
... 1. Select the longest carbon chain and name it as the normal straight-chain alkane (this is the "parent chain") 2. Number the parent chain starting at the end of the chain nearest the first alkyl substituent. 3. Use the numbers obtained by the application of rule #2 to designate the locations of the ...
Chemistry workbook
... aqueous zinc sulphate is mixed with a solution of ammonium phosphate to produce ammonium sulfate solution and zinc phosphate precipitate ...
... aqueous zinc sulphate is mixed with a solution of ammonium phosphate to produce ammonium sulfate solution and zinc phosphate precipitate ...
General and Organic Chemistry Review Primer
... the number of protons and neutrons. Calculating an element’s mass number is complicated by the existence of isotopes, atoms of an element with the same number of protons but different numbers of neutrons. Many naturally occurring elements exist as a mixture of isotopes. For example, carbon has three ...
... the number of protons and neutrons. Calculating an element’s mass number is complicated by the existence of isotopes, atoms of an element with the same number of protons but different numbers of neutrons. Many naturally occurring elements exist as a mixture of isotopes. For example, carbon has three ...
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
... Mechanism of Formation of Imines Primary amine adds to C=O Proton is lost from N and adds to O to yield a neutral ...
... Mechanism of Formation of Imines Primary amine adds to C=O Proton is lost from N and adds to O to yield a neutral ...
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
... Mechanism of Formation of Imines Primary amine adds to C=O Proton is lost from N and adds to O to yield a neutral ...
... Mechanism of Formation of Imines Primary amine adds to C=O Proton is lost from N and adds to O to yield a neutral ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.