PHYSICOCHEMICAL PROPERTIES OF ORGANIC MEDICINAL
... Principles of Drug Action 1, Spring 2005, Alcohols The alcohol and phenol moieties are common functional groups found in drug compounds. The primary physicochemical property of importance in the drug chemistry of the alcohols is the O-H dipole which can imparts water solubility by donating and acce ...
... Principles of Drug Action 1, Spring 2005, Alcohols The alcohol and phenol moieties are common functional groups found in drug compounds. The primary physicochemical property of importance in the drug chemistry of the alcohols is the O-H dipole which can imparts water solubility by donating and acce ...
acid
... To this point we have written chemical equations in terms of neutral species (molecules, or formula units). However, in aqueous solution it is often more appropriate to write equations in terms of ions. There are three ways we normally write chemical equations: 1) Molecular equation. All reactants a ...
... To this point we have written chemical equations in terms of neutral species (molecules, or formula units). However, in aqueous solution it is often more appropriate to write equations in terms of ions. There are three ways we normally write chemical equations: 1) Molecular equation. All reactants a ...
N5 Chemistry 2014
... 6. A student reacted acidified potassium permanganate solution with oxalic acid, C2H2O4. 2MnO4–(aq) + 5C2H2O4(aq) + 6H+(aq) ...
... 6. A student reacted acidified potassium permanganate solution with oxalic acid, C2H2O4. 2MnO4–(aq) + 5C2H2O4(aq) + 6H+(aq) ...
Chemistry of alcohols (powerpoint)
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
5. Formulae, equations and amounts of substance
... •Results should be clearly recorded in a table •Result should be recorded in full (i.e. both initial and final readings) •Record titre volumes to 2dp (0.05 cm3) ...
... •Results should be clearly recorded in a table •Result should be recorded in full (i.e. both initial and final readings) •Record titre volumes to 2dp (0.05 cm3) ...
Chemistry of CHLORINE
... chlorine gas. Chemical equation 2Fe(s) + 3Cl2 (g) → 2FeCl3(g) c) (i) Why is the brown solid collected at the point as shown in method I and II. -Heated iron (III) Chloride crystals sublime to gas and solidify on the cooler parts. (ii) Name another metal that can be used in place of iron to react wit ...
... chlorine gas. Chemical equation 2Fe(s) + 3Cl2 (g) → 2FeCl3(g) c) (i) Why is the brown solid collected at the point as shown in method I and II. -Heated iron (III) Chloride crystals sublime to gas and solidify on the cooler parts. (ii) Name another metal that can be used in place of iron to react wit ...
Boronic acids facilitate rapid oxime condensations at neutral pH
... the range of 4.5–5, is consistent with normal oxime formation35 except there is a distortion in the sigmoidal shape at higher pH (i.e. 7.2 & 8). The asymmetry in the pH dependence curve is inverted in comparison to a typical oxime condensation (acetone with hydroxylamine),35 which shows a more prono ...
... the range of 4.5–5, is consistent with normal oxime formation35 except there is a distortion in the sigmoidal shape at higher pH (i.e. 7.2 & 8). The asymmetry in the pH dependence curve is inverted in comparison to a typical oxime condensation (acetone with hydroxylamine),35 which shows a more prono ...
acetyl coenzyme A - Fakultas Farmasi Unand
... Dimethylallyl pyrophosphate has a leaving group (pyrophosphate) at an allylic carbon; it is reactive toward nucleophilic substitution at this position. Dr. Wolf's CHM 424 ...
... Dimethylallyl pyrophosphate has a leaving group (pyrophosphate) at an allylic carbon; it is reactive toward nucleophilic substitution at this position. Dr. Wolf's CHM 424 ...
Document
... diacteyl at an alkaline pH, and the addition of -naphthol which forms a red-colored complex with diacetyl. • The production of acetoin and butylene glycol by glucose fermentation is an important biochemical property used for the identification of Klebsiella, Enterobacter, and ...
... diacteyl at an alkaline pH, and the addition of -naphthol which forms a red-colored complex with diacetyl. • The production of acetoin and butylene glycol by glucose fermentation is an important biochemical property used for the identification of Klebsiella, Enterobacter, and ...
Learning Outcomes Leaving Certificate Chemistry
... define relative atomic mass (Ar) using the C12 scale define isotope describe the composition of isotopes using hydrogen and carbon as examples describe how a mass spectrometer can be used to determine relative atomic mass describe the principles on which the Mass Spectrometer is based explain the fu ...
... define relative atomic mass (Ar) using the C12 scale define isotope describe the composition of isotopes using hydrogen and carbon as examples describe how a mass spectrometer can be used to determine relative atomic mass describe the principles on which the Mass Spectrometer is based explain the fu ...
1 - Academics
... 18. The best definition for and explanation of electronegativity is: a) Electronegativity is the measure of the tendency of a combined atom to attract a shared pair of electrons to itself; elements in the upper right hand side of the periodic table tend to be more electronegative because they have f ...
... 18. The best definition for and explanation of electronegativity is: a) Electronegativity is the measure of the tendency of a combined atom to attract a shared pair of electrons to itself; elements in the upper right hand side of the periodic table tend to be more electronegative because they have f ...
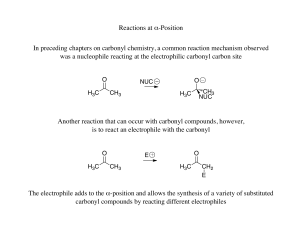
Reactions at α-Position In preceding chapters on carbonyl chemistry
... While reacting two different ketones with alkoxide base is impractical due to the variety of products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
... While reacting two different ketones with alkoxide base is impractical due to the variety of products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
SCH4U Exam Review
... SCH4U Exam Review Unit 1 – Energy Changes and Rates of Reactions 1. The molar heat of vapourization of acetone, C3H6O is 30.3 kJ/mol at its boiling point. How much heat would be liberated by the condensation of 1.00 g of acetone? ANS: 0.522 kJ 2. Aluminum reacts readily with chlorine gas to produce ...
... SCH4U Exam Review Unit 1 – Energy Changes and Rates of Reactions 1. The molar heat of vapourization of acetone, C3H6O is 30.3 kJ/mol at its boiling point. How much heat would be liberated by the condensation of 1.00 g of acetone? ANS: 0.522 kJ 2. Aluminum reacts readily with chlorine gas to produce ...
5. Formulae, equations and amounts of substance
... •Results should be clearly recorded in a table •Result should be recorded in full (i.e. both initial and final readings) •Record titre volumes to 2dp (0.05 cm3) ...
... •Results should be clearly recorded in a table •Result should be recorded in full (i.e. both initial and final readings) •Record titre volumes to 2dp (0.05 cm3) ...
Amount of substance
... This equilibrium, at a temperature of 585 K and a total pressure of 540 kPa, occurs in a vessel of volume 1.80 dm3. At equilibrium, the vessel contains 0.0500 mol of SO2(g), 0.0800 mol of O2(g) and 0.0700 mol of SO3(g). At equilibrium in the same vessel of volume 1.80 dm3 under altered conditions, t ...
... This equilibrium, at a temperature of 585 K and a total pressure of 540 kPa, occurs in a vessel of volume 1.80 dm3. At equilibrium, the vessel contains 0.0500 mol of SO2(g), 0.0800 mol of O2(g) and 0.0700 mol of SO3(g). At equilibrium in the same vessel of volume 1.80 dm3 under altered conditions, t ...
synthetic approaches for quinoline and isoquinoline
... naturally occurring alkaloids including papaverine and morphine. The isoquinoline ring in these natural compound derives from the aromatic amino acid tyrosine. 6,7,8,9,10,11 ...
... naturally occurring alkaloids including papaverine and morphine. The isoquinoline ring in these natural compound derives from the aromatic amino acid tyrosine. 6,7,8,9,10,11 ...
Stoichiometry - Norbraten
... (a) What is the maximum mass of iron, in grams, that can be obtained from 454.0 g of iron(III) oxide? (b) What mass of CO is required to reduce the iron(III) oxide to iron metal? ...
... (a) What is the maximum mass of iron, in grams, that can be obtained from 454.0 g of iron(III) oxide? (b) What mass of CO is required to reduce the iron(III) oxide to iron metal? ...
View/Open - AURA - Alfred University
... that triflic acid is not responsible for the progress of the reaction, suggests that PEDOT can mediate the Ritter reaction under mild conditions and it may now be possible to use unprotected, acid-sensitive functional groups. In an attempt to determine the scope of the PEDOT-mediated Ritter reactio ...
... that triflic acid is not responsible for the progress of the reaction, suggests that PEDOT can mediate the Ritter reaction under mild conditions and it may now be possible to use unprotected, acid-sensitive functional groups. In an attempt to determine the scope of the PEDOT-mediated Ritter reactio ...
Chemistry in Society Homework Booklet
... following balanced equation. N2 + 3H2 2NH3 The table shows the percentages of ammonia present at equilibrium under different conditions of temperature T and pressure P when hydrogen and nitrogen gases were mixed in a 3:1 molar ratio. Is this an endothermic or exothermic reaction? Give a reason for y ...
... following balanced equation. N2 + 3H2 2NH3 The table shows the percentages of ammonia present at equilibrium under different conditions of temperature T and pressure P when hydrogen and nitrogen gases were mixed in a 3:1 molar ratio. Is this an endothermic or exothermic reaction? Give a reason for y ...
17 - Wiley
... negative charge at different locations around the benzene ring instead of on the oxygen atom: ...
... negative charge at different locations around the benzene ring instead of on the oxygen atom: ...
Determination of Active Ingredients in Commercial Bleach and Vinegar
... Acetic acid, CH3COOH, is the acid component of vinegar. Its concentration varies slightly in different samples but must be at least 4% by mass (acetic acid in water) to meet the minimum commercial standards. Concentration of acetic acid may even exceed 5% in some vinegar products. Caramel flavoring ...
... Acetic acid, CH3COOH, is the acid component of vinegar. Its concentration varies slightly in different samples but must be at least 4% by mass (acetic acid in water) to meet the minimum commercial standards. Concentration of acetic acid may even exceed 5% in some vinegar products. Caramel flavoring ...
AP Chemistry Review Preparing for the AP
... Know the bond angles on a Bent Geometry, Trigonal Planer, Trigonal Pyrimidal, and Tetrahedral. Also know why bond angles shrink as lone pairs are added (b/c if increased repulsion amongst the electrons causing the bond angles to squeeze) It would be a safe bet to assume that when a metal by itself i ...
... Know the bond angles on a Bent Geometry, Trigonal Planer, Trigonal Pyrimidal, and Tetrahedral. Also know why bond angles shrink as lone pairs are added (b/c if increased repulsion amongst the electrons causing the bond angles to squeeze) It would be a safe bet to assume that when a metal by itself i ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.