CHE-310 Organic Chemistry I_
... For alkyl halides, alcohols and ethers, be able to name compounds correctly (nomenclature). Where necessay, be able to specify congiguration in the name. Know the two new mechanisms that we have learned in these chapters: SN2, SN1. Know which mechanisms go with which reactions under which conditions ...
... For alkyl halides, alcohols and ethers, be able to name compounds correctly (nomenclature). Where necessay, be able to specify congiguration in the name. Know the two new mechanisms that we have learned in these chapters: SN2, SN1. Know which mechanisms go with which reactions under which conditions ...
Experimental skills and abilities
... 1 The evaporation process should be done very slowly. This is because sugar can easily char as it solidifies around the sides of the evaporating basin during the evaporating process. Also the crystallisation will require a lot longer for crystals to form from the concentrated solution and may need ...
... 1 The evaporation process should be done very slowly. This is because sugar can easily char as it solidifies around the sides of the evaporating basin during the evaporating process. Also the crystallisation will require a lot longer for crystals to form from the concentrated solution and may need ...
«Классы и номенклатура неорганических соединений»
... Specify, which from the processes must be care at the higher pressure A. * 3H2(g) + N2(g)↔ 2NH3(g) B. Fe(s) + H2O(g) ↔ FeO(s) + H2(g) C. N2(g) + O2(g) ↔ 2NO(g) D. C(s) + О2(g) ↔ CO2(g) E. H2(g) + Cl2(g) ↔ 2HCl(g) 2. Specify the unit for the rate of chemical reaction? A. *mol ∙ L–1 s–1 B. mol ∙ ml/s– ...
... Specify, which from the processes must be care at the higher pressure A. * 3H2(g) + N2(g)↔ 2NH3(g) B. Fe(s) + H2O(g) ↔ FeO(s) + H2(g) C. N2(g) + O2(g) ↔ 2NO(g) D. C(s) + О2(g) ↔ CO2(g) E. H2(g) + Cl2(g) ↔ 2HCl(g) 2. Specify the unit for the rate of chemical reaction? A. *mol ∙ L–1 s–1 B. mol ∙ ml/s– ...
Net ionic equation
... Reactions of acids and bases •Neutralization: acid + base are mixed: HNO3(aq) + KOH(aq) ??? •Salt = ionic compound cation from base anion from acid. •Neutralization of acid with metal hydroxide produces water and a salt. •Acids + carbonates = CO2 and H2O ...
... Reactions of acids and bases •Neutralization: acid + base are mixed: HNO3(aq) + KOH(aq) ??? •Salt = ionic compound cation from base anion from acid. •Neutralization of acid with metal hydroxide produces water and a salt. •Acids + carbonates = CO2 and H2O ...
Kinetics of Oxidation of Benzyl Alcohol with Dilute Nitric Acid
... oxidation of benzyl alcohol using molecular oxygen requires a heterogeneous catalyst, high temperature (210 °C), and longer reaction times (>5 h). Although the reported selectivity is quite good (75%-95%), the conversion levels are too low (maximum of 40%).14 Practically, in all the previously cited ...
... oxidation of benzyl alcohol using molecular oxygen requires a heterogeneous catalyst, high temperature (210 °C), and longer reaction times (>5 h). Although the reported selectivity is quite good (75%-95%), the conversion levels are too low (maximum of 40%).14 Practically, in all the previously cited ...
Preparation of d, l-Phenylalanine by Amidocarbonylation of Benzyl
... alcohol was not described before; the expected product is phenylacetic acid. It seems that in both cases the acetamide catalyzes the hydroformylation reaction in some way. The effect seems general for amides. It was found that in the presence of a stoichiometric amount of DMF rather than acetamide, ...
... alcohol was not described before; the expected product is phenylacetic acid. It seems that in both cases the acetamide catalyzes the hydroformylation reaction in some way. The effect seems general for amides. It was found that in the presence of a stoichiometric amount of DMF rather than acetamide, ...
General Chemistry Unit 11
... In a single replacement reaction a single uncombined element replaces another in a compound. Two reactants yield two products. For example when zinc combines with hydrochloric acid, the zinc replaces hydrogen. The chemical equation for this single replacement reaction looks like: reactant + reactant ...
... In a single replacement reaction a single uncombined element replaces another in a compound. Two reactants yield two products. For example when zinc combines with hydrochloric acid, the zinc replaces hydrogen. The chemical equation for this single replacement reaction looks like: reactant + reactant ...
chapter4-bur.2917051..
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
Net ionic equation
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
Quiz Samples
... What property of light is measured in terms of Hz: a)b)c) Calculate what wavelength is expected for light of photons produced due to n = 4 to n = 2 transition in a hydrogen atom (R = 3.25x1015 Hz, c = 3.00x1010 cm/sec): a)b)c) What wavelength is expected for light composed of photons due to n=5 to n ...
... What property of light is measured in terms of Hz: a)b)c) Calculate what wavelength is expected for light of photons produced due to n = 4 to n = 2 transition in a hydrogen atom (R = 3.25x1015 Hz, c = 3.00x1010 cm/sec): a)b)c) What wavelength is expected for light composed of photons due to n=5 to n ...
Chapter 18.2
... The main steps in balancing redox equations: 1. Using oxidation numbers, identify what is oxidized (what loses electrons) and what is reduced (what gains electrons). 2. What are the products after the oxidation and reduction take place? 3. Is the redox reaction done under acidic or basic conditions? ...
... The main steps in balancing redox equations: 1. Using oxidation numbers, identify what is oxidized (what loses electrons) and what is reduced (what gains electrons). 2. What are the products after the oxidation and reduction take place? 3. Is the redox reaction done under acidic or basic conditions? ...
Aldehydes And Ketones
... • the parent alkane is the longest chain that contains the carbonyl group • for ketones, change the suffix -e to -one • number the chain to give C=O the smaller number • the IUPAC retains the common names acetone, acetophenone, and benzophenone O ...
... • the parent alkane is the longest chain that contains the carbonyl group • for ketones, change the suffix -e to -one • number the chain to give C=O the smaller number • the IUPAC retains the common names acetone, acetophenone, and benzophenone O ...
8872 Chemistry H1 syllabus for 2016
... recognition of the need for students to develop skills that will be of long term value in an increasingly technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the tea ...
... recognition of the need for students to develop skills that will be of long term value in an increasingly technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the tea ...
Atomic Structure
... called .................. Atoms of magnesium are neutral because they contain the same number of electrons and .................. ...
... called .................. Atoms of magnesium are neutral because they contain the same number of electrons and .................. ...
Organic Chemistry Fifth Edition
... property—they are more soluble in nonpolar solvents than in water. Some of the most important of them—the ones in this chapter—are related in that they have acetic acid (acetate) as their biosynthetic origin. In many biosynthetic pathways a substance called acetyl coenzyme A serves as the source of ...
... property—they are more soluble in nonpolar solvents than in water. Some of the most important of them—the ones in this chapter—are related in that they have acetic acid (acetate) as their biosynthetic origin. In many biosynthetic pathways a substance called acetyl coenzyme A serves as the source of ...
Chapter 16, Amines
... ¾Amines are polar, hydrogen bond with water, and 1Eand 2E with each other, but more weakly than alcohols ¾Amines have lower boiling points than alcohols of similar molecular weight, they are also soluble in water if the substituent group part is not too large and non-polar. C1-C7 = miscible to sligh ...
... ¾Amines are polar, hydrogen bond with water, and 1Eand 2E with each other, but more weakly than alcohols ¾Amines have lower boiling points than alcohols of similar molecular weight, they are also soluble in water if the substituent group part is not too large and non-polar. C1-C7 = miscible to sligh ...
Research on Hydrogenation of FAME to Fatty Alcohols
... acid methyl ester was above 99% with the condition of less than 4.0h-1 space velocity. While in terms of purpose products, it was more than 90%, and increased slightly with space velocity increased. Compared with 0.15~0.4h-1 space velocity which calculated according to tradition process of hydrogeno ...
... acid methyl ester was above 99% with the condition of less than 4.0h-1 space velocity. While in terms of purpose products, it was more than 90%, and increased slightly with space velocity increased. Compared with 0.15~0.4h-1 space velocity which calculated according to tradition process of hydrogeno ...
Studies toward the Stereoselective Synthesis of the
... Mycotoxins are toxic secondary metabolites produced by fungi and they are the causative agents of various diseases in man and his domestic animals. Human beings and animals get the diseases, commonly called mycotoxicoses through the ingestion of foods or feeds contaminated by these toxic fungal meta ...
... Mycotoxins are toxic secondary metabolites produced by fungi and they are the causative agents of various diseases in man and his domestic animals. Human beings and animals get the diseases, commonly called mycotoxicoses through the ingestion of foods or feeds contaminated by these toxic fungal meta ...
South Pasadena • AP Chemistry
... 8. Draw a diagram for activation energy that indicates that the reaction is endothermic and one that indicates that the reaction is exothermic. Label the x-axis, y-axis, reactants, products, and what amount of activation energy is needed for each reaction. 9. What does the Ksp value indicate about a ...
... 8. Draw a diagram for activation energy that indicates that the reaction is endothermic and one that indicates that the reaction is exothermic. Label the x-axis, y-axis, reactants, products, and what amount of activation energy is needed for each reaction. 9. What does the Ksp value indicate about a ...
2 - mrstorie
... Increases across, decreases down. Across – more proton attraction, down – more shielding and further away from nucleus 11. What are the trends observed for electronegativty, electron affinity, and metallic character? EN - Increases across, decreases down. EA – Increases across, decreases down. MC – ...
... Increases across, decreases down. Across – more proton attraction, down – more shielding and further away from nucleus 11. What are the trends observed for electronegativty, electron affinity, and metallic character? EN - Increases across, decreases down. EA – Increases across, decreases down. MC – ...
the Language of Chemistry
... I have presented the various scientific concepts as vital, compelling and meaningful which might otherwise seem dull. Each part of the book has been carefully planned to make it student friendly and present Chemistry in an interesting, understandable and enjoyable manner. I have tried to stress the ...
... I have presented the various scientific concepts as vital, compelling and meaningful which might otherwise seem dull. Each part of the book has been carefully planned to make it student friendly and present Chemistry in an interesting, understandable and enjoyable manner. I have tried to stress the ...
Methane reacts with 1 mole of chlorine in presence of
... 3. What is meant by term homologous series ?What is the general formula of Alkenes 4. Classify the following as Alkane and alkyne,, CH4, C2H4 , C2H2 , C2H6 , C3H4 5.Explain why carbon generally form compound by covalent bonding? 6. Give example of each a..open chain b. branched chain hydrocarbon 7. ...
... 3. What is meant by term homologous series ?What is the general formula of Alkenes 4. Classify the following as Alkane and alkyne,, CH4, C2H4 , C2H2 , C2H6 , C3H4 5.Explain why carbon generally form compound by covalent bonding? 6. Give example of each a..open chain b. branched chain hydrocarbon 7. ...
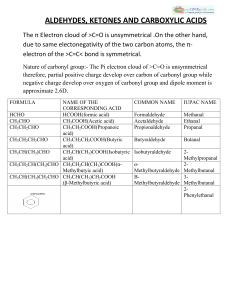
ALDEHYDES, KETONES AND CARBOXYLIC ACIDS
... :-Aldehydes, Ketones and Carboxylic acids are important classes of organic compounds containing carbonyl groups. :-They are highly polar molecules. :-They boil at higher temperatures than the corresponding hydrocarbons and weakly polar compounds such as ethers. :-Lower members are soluble in water b ...
... :-Aldehydes, Ketones and Carboxylic acids are important classes of organic compounds containing carbonyl groups. :-They are highly polar molecules. :-They boil at higher temperatures than the corresponding hydrocarbons and weakly polar compounds such as ethers. :-Lower members are soluble in water b ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.