Mass Mass - White Plains Public Schools
... Knowing the molar relationships between the substances involved allow us to calculate the mass relationships. This quantitative study of chemical reactions is called stoichiometry. Mass-Mass problems generally involve the following steps: 1. Changing the mass given to the number of moles with the f ...
... Knowing the molar relationships between the substances involved allow us to calculate the mass relationships. This quantitative study of chemical reactions is called stoichiometry. Mass-Mass problems generally involve the following steps: 1. Changing the mass given to the number of moles with the f ...
Carbohydrates: Occurrence, Structures and Chemistry
... which is often used as a synonym for ‘monosaccharide’, but may also be applied to simple compounds containing more than one monosaccharide unit. Indeed, in everyday usage ‘sugar’ signifies table sugar, which is sucrose (German ‘Saccharose’; French ‘sucrose’ or ‘saccharose’), a disaccharide composed ...
... which is often used as a synonym for ‘monosaccharide’, but may also be applied to simple compounds containing more than one monosaccharide unit. Indeed, in everyday usage ‘sugar’ signifies table sugar, which is sucrose (German ‘Saccharose’; French ‘sucrose’ or ‘saccharose’), a disaccharide composed ...
Structured questions
... A student tried to investigate the pH value of rainwater collected in an area with an incinerator and a lot of factories. a) Could he use methyl orange to measure the pH value of rainwater? Explain briefly. b) Name a device he can use to measure the pH value of rainwater accurately. c) He found that ...
... A student tried to investigate the pH value of rainwater collected in an area with an incinerator and a lot of factories. a) Could he use methyl orange to measure the pH value of rainwater? Explain briefly. b) Name a device he can use to measure the pH value of rainwater accurately. c) He found that ...
Lecture - Ch 17
... Compounds: Grignard Reagents • Grignard reagents do not give addition products with carboxylic acids – Acidic carboxyl hydrogen reacts with the basic Grignard reagent to yield a hydrocarbon and the magnesium salt of the acid ...
... Compounds: Grignard Reagents • Grignard reagents do not give addition products with carboxylic acids – Acidic carboxyl hydrogen reacts with the basic Grignard reagent to yield a hydrocarbon and the magnesium salt of the acid ...
Chapter 1 Structure and Bonding
... ROH plus Strong Mineral Acid 1) Remember that ROH plus HBr gives RBr because nucleophile is present 2) Protonation by mineral acid gives good leaving group (H2O) but does not give an interfering nucleophile ...
... ROH plus Strong Mineral Acid 1) Remember that ROH plus HBr gives RBr because nucleophile is present 2) Protonation by mineral acid gives good leaving group (H2O) but does not give an interfering nucleophile ...
AP Chemistry - cloudfront.net
... 9.52 Draw a Lewis structure for (a) SiF4; (b) SeCL2; (c) COF2 (C central); (d) PH4 +; (e) C2F4. 9.54 Draw Lewis structure of all the important resonance forms of each of the following: (a) NO2; (b) NO2F (N central); (c) HNO3 (HONO2). 9.56 Draw a Lewis structure in the following g: (a) IF5; (b) AlH4- ...
... 9.52 Draw a Lewis structure for (a) SiF4; (b) SeCL2; (c) COF2 (C central); (d) PH4 +; (e) C2F4. 9.54 Draw Lewis structure of all the important resonance forms of each of the following: (a) NO2; (b) NO2F (N central); (c) HNO3 (HONO2). 9.56 Draw a Lewis structure in the following g: (a) IF5; (b) AlH4- ...
Effect of an industrial chemical waste on the uptake
... The binding or complexing of metal ions by humic substances (HS), as widely distributed natural ligands in soil and natural waters, is an important factor determining metal toxicity, bioavailability and transport.2–4 Humic and fulvic acids, as fractions of humic substances, are the most important HS ...
... The binding or complexing of metal ions by humic substances (HS), as widely distributed natural ligands in soil and natural waters, is an important factor determining metal toxicity, bioavailability and transport.2–4 Humic and fulvic acids, as fractions of humic substances, are the most important HS ...
aq - HCC Learning Web
... • Aqueous solutions of lead(II) nitrate and potassium iodide produce a yellow precipitate of lead(II) iodide and an aqueous solution of potassium nitrate Pb(NO3)2(aq) + KI(aq) PbI2(s) + KNO3(aq) • Aqueous solutions of calcium nitrate and sodium carbonate react to give a white precipitate of calciu ...
... • Aqueous solutions of lead(II) nitrate and potassium iodide produce a yellow precipitate of lead(II) iodide and an aqueous solution of potassium nitrate Pb(NO3)2(aq) + KI(aq) PbI2(s) + KNO3(aq) • Aqueous solutions of calcium nitrate and sodium carbonate react to give a white precipitate of calciu ...
Chapter 1 questions
... An oxide of sulfur contains 40.0% by mass of sulfur. Calculate the empirical formula of the oxide. Q8. Analysis by mass has indicated the following percentage composition by mass of certain compounds. Calculate the empirical formula of each: a) carbon 75.0%, hydrogen 25.0% b) magnesium 60.3%, oxygen ...
... An oxide of sulfur contains 40.0% by mass of sulfur. Calculate the empirical formula of the oxide. Q8. Analysis by mass has indicated the following percentage composition by mass of certain compounds. Calculate the empirical formula of each: a) carbon 75.0%, hydrogen 25.0% b) magnesium 60.3%, oxygen ...
Alcohols - Chem1-tsu
... alcohols generally involve the reactions of -OH group. They can undergo substitution as well as elimination reaction. Reactions of alcohols are classified into three types. 1) Reactions involving cleavage of -OH bond. Reaction with metals Alcohols and phenols react with metals such as sodium, potass ...
... alcohols generally involve the reactions of -OH group. They can undergo substitution as well as elimination reaction. Reactions of alcohols are classified into three types. 1) Reactions involving cleavage of -OH bond. Reaction with metals Alcohols and phenols react with metals such as sodium, potass ...
GROUP 13 ELEMENTS -THE BORON FAMILY -
... 31. It has the electron configuration [Ar]2s2 2p1 and +3 oxidation state. The melting point is 29.8º C and therefore melts by increasing room temperature by a little. Gallium is important because it forms gallium arsenide (GaAs), which can convert light directly into electricity. Also due to thermit ...
... 31. It has the electron configuration [Ar]2s2 2p1 and +3 oxidation state. The melting point is 29.8º C and therefore melts by increasing room temperature by a little. Gallium is important because it forms gallium arsenide (GaAs), which can convert light directly into electricity. Also due to thermit ...
Basic Chemical Concepts I
... A 25.00 mL sample of HCl(aq) was added to a 0.1000 g sample of CaCO3. All the CaCO3 reacted, leaving some unreacted HCl(aq). CaCO3(s) + 2 HCl(aq) → CaCl2(aq) + H2O + CO2(g) The unreacted HCl(aq) required 43.82 mL of 0.01185 M Ba(OH)2(aq) to complete the following reaction. 2 HCl(aq) + Ba(OH)2(aq) → ...
... A 25.00 mL sample of HCl(aq) was added to a 0.1000 g sample of CaCO3. All the CaCO3 reacted, leaving some unreacted HCl(aq). CaCO3(s) + 2 HCl(aq) → CaCl2(aq) + H2O + CO2(g) The unreacted HCl(aq) required 43.82 mL of 0.01185 M Ba(OH)2(aq) to complete the following reaction. 2 HCl(aq) + Ba(OH)2(aq) → ...
Practical and selective aerobic oxidation of alcohols to
... reaction occurred in a two-phase flow along the length of the reactor, staged injection of oxygen was used to maintain a consistent amount of gas along the reaction bed. Although characteristics of the flow system were very well-defined, the generality of the system was not demonstrated, as only ben ...
... reaction occurred in a two-phase flow along the length of the reactor, staged injection of oxygen was used to maintain a consistent amount of gas along the reaction bed. Although characteristics of the flow system were very well-defined, the generality of the system was not demonstrated, as only ben ...
Word - icho39.chem.msu.ru
... 1. In 1875 the French chemist Paul-Emile Lecoq de Boisbaudran studied the spectra of zinc ore and discovered the traces of a new element, which he called “gallium” from the Latin word "Gallia" meaning "France" and perhaps also from the Latin word "gallus" (the cock, a translation of Lecoq). In the s ...
... 1. In 1875 the French chemist Paul-Emile Lecoq de Boisbaudran studied the spectra of zinc ore and discovered the traces of a new element, which he called “gallium” from the Latin word "Gallia" meaning "France" and perhaps also from the Latin word "gallus" (the cock, a translation of Lecoq). In the s ...
Topic 4 - Lloyd Crosby
... c. Examples of acid/base with gas formers (1) A solution of potassium hydrogen carbonate is mixed with an excess of sulfuric acid: KHCO3 (aq) + H2SO4 (aq) KHCO3 (aq) + H2SO4 (aq) KCl (aq) + CO2 (g) + H2O (l) Balanced net ionic equation HCO3 (aq) + H+ (aq) CO2 (g) + H2O (l) Evidence for reacti ...
... c. Examples of acid/base with gas formers (1) A solution of potassium hydrogen carbonate is mixed with an excess of sulfuric acid: KHCO3 (aq) + H2SO4 (aq) KHCO3 (aq) + H2SO4 (aq) KCl (aq) + CO2 (g) + H2O (l) Balanced net ionic equation HCO3 (aq) + H+ (aq) CO2 (g) + H2O (l) Evidence for reacti ...
Basic Chemical Concepts I
... A 25.00 mL sample of HCl(aq) was added to a 0.1000 g sample of CaCO3. All the CaCO3 reacted, leaving some unreacted HCl(aq). CaCO3(s) + 2 HCl(aq) → CaCl2(aq) + H2O + CO2(g) The unreacted HCl(aq) required 43.82 mL of 0.01185 M Ba(OH)2(aq) to complete the following reaction. 2 HCl(aq) + Ba(OH)2(aq) → ...
... A 25.00 mL sample of HCl(aq) was added to a 0.1000 g sample of CaCO3. All the CaCO3 reacted, leaving some unreacted HCl(aq). CaCO3(s) + 2 HCl(aq) → CaCl2(aq) + H2O + CO2(g) The unreacted HCl(aq) required 43.82 mL of 0.01185 M Ba(OH)2(aq) to complete the following reaction. 2 HCl(aq) + Ba(OH)2(aq) → ...
Syllabus of Medical / Dental Colleges Entrance Test 2016
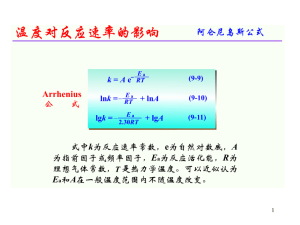
... equation; order of reaction; rate constant; half-life of a reaction; rate-determining step b) Explain qualitatively, in terms of collisions, the effect of concentration changes on the rate of a reaction c) Explain that, in the presence of a catalyst, a reaction has a different mechanism, i.e. one of ...
... equation; order of reaction; rate constant; half-life of a reaction; rate-determining step b) Explain qualitatively, in terms of collisions, the effect of concentration changes on the rate of a reaction c) Explain that, in the presence of a catalyst, a reaction has a different mechanism, i.e. one of ...
13: Carbonyl Compounds: Ketones, Aldehydes, Carboxylic Acids
... aldehydes must have at least one H bonded to the C=O (Figure 13.8). Figure 13.8 As a result, the C=O group of aldehydes is always at the end of an alkane chain. In contrast, the C=O group of a ketone can never be at the end of an alkane chain and must always be part of the interior of that chain. We ...
... aldehydes must have at least one H bonded to the C=O (Figure 13.8). Figure 13.8 As a result, the C=O group of aldehydes is always at the end of an alkane chain. In contrast, the C=O group of a ketone can never be at the end of an alkane chain and must always be part of the interior of that chain. We ...
Chapter 7 Lecture
... Writing Chemical Equations for Reactions in Solution: Molecular and Complete Ionic Equations • A molecular equation is an equation showing the complete neutral formulas for every compound in the reaction. • Complete ionic equations show aqueous ionic compounds that normally dissociate in solution a ...
... Writing Chemical Equations for Reactions in Solution: Molecular and Complete Ionic Equations • A molecular equation is an equation showing the complete neutral formulas for every compound in the reaction. • Complete ionic equations show aqueous ionic compounds that normally dissociate in solution a ...
SAT - mvhs-fuhsd.org
... • The masses and atomic numbers must add up to be the same on both sides of the arrow. ...
... • The masses and atomic numbers must add up to be the same on both sides of the arrow. ...
File - Grade 12 Chemistry
... 22. Explain the difference between covalent bonding and polar covalent bonding, using an example of each. 23. What are the three different molecular shapes that can occur with a tetrahedral geometric arrangement of electron groups? 24. The neutralization of nitric acid with potassium hydroxide has a ...
... 22. Explain the difference between covalent bonding and polar covalent bonding, using an example of each. 23. What are the three different molecular shapes that can occur with a tetrahedral geometric arrangement of electron groups? 24. The neutralization of nitric acid with potassium hydroxide has a ...
Chemistry Tests Questions
... 6. An orbital is a region of space about the nucleus of an atom in which there is greatest probability of finding a particular electron. Illustrate the shape of, (i) an s orbital and (ii) a p orbital. 7. In the mass spectrometer a powerful magnetic field is used to deflect .......................... ...
... 6. An orbital is a region of space about the nucleus of an atom in which there is greatest probability of finding a particular electron. Illustrate the shape of, (i) an s orbital and (ii) a p orbital. 7. In the mass spectrometer a powerful magnetic field is used to deflect .......................... ...
Homework,1 Atoms, molecules, and ions
... 6- Calculate the mass percent of nitrogen in HNO3. a) 45.2% b) 25.0% c) 22.2% d) 20.0% e) none of these 7- You are setting up a reaction between two chemicals that react according to the equation 3 A + 4 B products. If you start with 1.00 mole each of both A and B, which chemical will be in exces ...
... 6- Calculate the mass percent of nitrogen in HNO3. a) 45.2% b) 25.0% c) 22.2% d) 20.0% e) none of these 7- You are setting up a reaction between two chemicals that react according to the equation 3 A + 4 B products. If you start with 1.00 mole each of both A and B, which chemical will be in exces ...
AP CHEMISTRY MRS. SPENCER CHAPTER 4 TEST: SOLUTION
... A solution of 0.100 M HCl and a solution of 0.100 M NaOH are prepared. A 40.0 mL sample of one of the solutions is added to a beaker and then titrated with the other solution. A pH electrode is used to obtain the data that are plotted in the titration curve shown above. Identify the solution that wa ...
... A solution of 0.100 M HCl and a solution of 0.100 M NaOH are prepared. A 40.0 mL sample of one of the solutions is added to a beaker and then titrated with the other solution. A pH electrode is used to obtain the data that are plotted in the titration curve shown above. Identify the solution that wa ...
Lecture 7
... Lewis proposed that any particle which could donate a pair of electrons should be called a Lewis base while those particles which accept electron pairs should be called Lewis acids. When a Lewis base donates an electron pair to a Lewis acid a dative covalent bond is formed. ...
... Lewis proposed that any particle which could donate a pair of electrons should be called a Lewis base while those particles which accept electron pairs should be called Lewis acids. When a Lewis base donates an electron pair to a Lewis acid a dative covalent bond is formed. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.