Honors Chemistry Unit 4 Student Packet: Honors Chemistry Problem
... 8. Chemists sometimes obtain calcium by reacting calcium oxide with aluminum. If the sample of calcium oxide is 85.0% pure, what mass of calcium would be produced form 500.g of the calcium oxide sample? 9. An ore containing 80.0 % zinc sulfide is heated with oxygen gas to produce zinc oxide and sulf ...
... 8. Chemists sometimes obtain calcium by reacting calcium oxide with aluminum. If the sample of calcium oxide is 85.0% pure, what mass of calcium would be produced form 500.g of the calcium oxide sample? 9. An ore containing 80.0 % zinc sulfide is heated with oxygen gas to produce zinc oxide and sulf ...
Unique Solutions
... a hydrogen gas and iron chloride are produced b chlorine gas and iron hydroxide are produced c no reaction takes place d iron salt and water are produced You are given potassium chromate solution. In order to obtain an orange precipitate what should we mix in it? ...
... a hydrogen gas and iron chloride are produced b chlorine gas and iron hydroxide are produced c no reaction takes place d iron salt and water are produced You are given potassium chromate solution. In order to obtain an orange precipitate what should we mix in it? ...
Organic_1_6.1ed_2012_02nd_module_functional_groups_and_IR
... C=O: 1630-1780 cm-1, strong absorption If you need to use other frequencies to identify other functional groups (and sometimes you will), a table of IR frequencies will be provided. ...
... C=O: 1630-1780 cm-1, strong absorption If you need to use other frequencies to identify other functional groups (and sometimes you will), a table of IR frequencies will be provided. ...
Chapter 4: Aqueous Solutions (Chs 4 and 5 in Jespersen, Ch4 in
... Chemical equilibrium - chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates ...
... Chemical equilibrium - chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates ...
CHEMISTRY SEC 06 SYLLABUS
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
Chemistry and the material world
... Systems that are not at equilibrium can be described by a similar ...
... Systems that are not at equilibrium can be described by a similar ...
Chapter 6: Chemical Equilibrium
... 9. The reaction, Q + 2 SO3(g) 2 SO2(g) + O2(g) is endothermic. Predict what will happen if the temperature is increased. a. Kc remains the same b. Kc decreases c. the pressure decreases d. more SO3(g) is produced * e. Kc increases T increase, reaction will shift to right side and Kc increase 10. Con ...
... 9. The reaction, Q + 2 SO3(g) 2 SO2(g) + O2(g) is endothermic. Predict what will happen if the temperature is increased. a. Kc remains the same b. Kc decreases c. the pressure decreases d. more SO3(g) is produced * e. Kc increases T increase, reaction will shift to right side and Kc increase 10. Con ...
AP Chemistry Summer Assignment
... basics down. Over the summer, you are responsible for doing the worksheets in this review packet. They are due on the first day of school and will be your first grade! You will have a test on this material at the end of the first week of school. The material in this packet should be mostly review fr ...
... basics down. Over the summer, you are responsible for doing the worksheets in this review packet. They are due on the first day of school and will be your first grade! You will have a test on this material at the end of the first week of school. The material in this packet should be mostly review fr ...
Saturday Study Session 1 1st Class Reactions
... • Solids, liquids, and gases can NOT be broken into ions • SOLUTION – if it says a solution, then it CAN be broken into ions if it is soluble in water. • Only ionic compounds can become separate ions in a solution. • The 6 strong acids (HCl, HBr, HI, HNO3, HClO4, H2SO4) and the strong bases (group 1 ...
... • Solids, liquids, and gases can NOT be broken into ions • SOLUTION – if it says a solution, then it CAN be broken into ions if it is soluble in water. • Only ionic compounds can become separate ions in a solution. • The 6 strong acids (HCl, HBr, HI, HNO3, HClO4, H2SO4) and the strong bases (group 1 ...
answers to part a of the national high school
... This question concerns the stoichiometry of the double displacement reaction between solutions of a metal nitrate (of formula M(NO3)x, say) and potassium iodide to give potassium nitrate and a precipitate of the metal iodide. It may be represented by the following equation: ...
... This question concerns the stoichiometry of the double displacement reaction between solutions of a metal nitrate (of formula M(NO3)x, say) and potassium iodide to give potassium nitrate and a precipitate of the metal iodide. It may be represented by the following equation: ...
Types of Chemical Reactions
... In a single replacement reaction, a single uncombined element replaces another element within a compound; in other words, one element trades places with another element in a compound. These reactions come in the general form of: A + BC → AC + B One example of a single displacement reaction is when z ...
... In a single replacement reaction, a single uncombined element replaces another element within a compound; in other words, one element trades places with another element in a compound. These reactions come in the general form of: A + BC → AC + B One example of a single displacement reaction is when z ...
Acids and Bases Unit
... This is a guessing question so be prepared for different responses. Most likely, that the light bulb will light up After testing the conductivity of the strong acid and before testing the weak acid in solution: o Did anything happen to the light bulb? Is that what we expected? If done correct ...
... This is a guessing question so be prepared for different responses. Most likely, that the light bulb will light up After testing the conductivity of the strong acid and before testing the weak acid in solution: o Did anything happen to the light bulb? Is that what we expected? If done correct ...
CHEMISTRY SEC 06 SYLLABUS
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
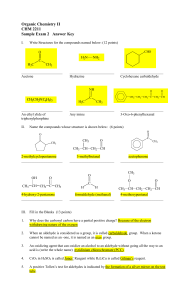
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... When an aldehyde is considered as a group, it is called carbaldehyde group. When a ketone cannot be named as an -one, it is named as an oxo- group. ...
... When an aldehyde is considered as a group, it is called carbaldehyde group. When a ketone cannot be named as an -one, it is named as an oxo- group. ...
CHEMISTRY SEC 06 SYLLABUS
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
CHEMISTRY SEC 06 SYLLABUS
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
2nd Nine Weeks Notes
... 1. The first step in understanding how a given chemical reaction occurs is to determine the form of the rate law. 2. We must determine experimentally the power to which each reactant concentration must be raised in the rate law. a. An exponent of “1” is referred to as first order. b. An exponent of ...
... 1. The first step in understanding how a given chemical reaction occurs is to determine the form of the rate law. 2. We must determine experimentally the power to which each reactant concentration must be raised in the rate law. a. An exponent of “1” is referred to as first order. b. An exponent of ...
Chapter 12 Packet
... http://www.scientificamerican.com/article.cfm?id=how-do-air-bags-work a. Write the balanced decomposition reaction for sodium azide. ...
... http://www.scientificamerican.com/article.cfm?id=how-do-air-bags-work a. Write the balanced decomposition reaction for sodium azide. ...
Lab Grade 250 pts - Western Illinois University
... Quizzes & Exams: Twelve in-class quizzes (10-15 min. duration) will be administered during the term and the “best ten” scores for the quizzes will be considered along ...
... Quizzes & Exams: Twelve in-class quizzes (10-15 min. duration) will be administered during the term and the “best ten” scores for the quizzes will be considered along ...
CHEMISTRY 331 - Western Illinois University
... Quizzes & Exams: Twelve in-class quizzes (10-15 min. duration) will be administered during the term and the “best ten” scores for the quizzes will be considered along with other scores when assigning the final grade for the course. The other two quizzes will be counted as your bonus points for the ...
... Quizzes & Exams: Twelve in-class quizzes (10-15 min. duration) will be administered during the term and the “best ten” scores for the quizzes will be considered along with other scores when assigning the final grade for the course. The other two quizzes will be counted as your bonus points for the ...
Acids, Bases and Salts
... Hydroxides/bases Al(OH)3(s) + 3CH3COOH(aq) → Al(CH3COO)3(aq) + 3H2O(l) Carbonates 3CaCO3(s) + 2H3PO4(aq) → Ca3(PO4)2(aq) + 3CO2(g) + 3H2O(l) Bicarbonates KHCO3(aq) + HNO3(aq) → KNO3(aq) + CO2(g) + H2O(l) Strong acids react with salts of weak acids Na3BO3(aq) + 3HBr(aq) → H3BO3(aq) + 3NaBr(aq) ...
... Hydroxides/bases Al(OH)3(s) + 3CH3COOH(aq) → Al(CH3COO)3(aq) + 3H2O(l) Carbonates 3CaCO3(s) + 2H3PO4(aq) → Ca3(PO4)2(aq) + 3CO2(g) + 3H2O(l) Bicarbonates KHCO3(aq) + HNO3(aq) → KNO3(aq) + CO2(g) + H2O(l) Strong acids react with salts of weak acids Na3BO3(aq) + 3HBr(aq) → H3BO3(aq) + 3NaBr(aq) ...
AP Chemistry
... Assume that chlorine is the central atom of each of the following species above. (a) Draw the Lewis dot structure for each of the above species. (b) List the species in order of decreasing bond angle and justify your answer. (c) Identify the one species which would dimerize and justify your answer. ...
... Assume that chlorine is the central atom of each of the following species above. (a) Draw the Lewis dot structure for each of the above species. (b) List the species in order of decreasing bond angle and justify your answer. (c) Identify the one species which would dimerize and justify your answer. ...
coordination compounds - Ahlcon Public School , Mayur Vihar Ph
... The bond energy of F2 is less than that of Cl2. Why? ...
... The bond energy of F2 is less than that of Cl2. Why? ...
Questions and Solutions
... How can you separate all the components in a mixture containing sand, iron filings, water, gasoline, red water-soluble dye, and blue water-soluble dye? In pure form the dyes are powders…magnet (iron), filter (sand), sep funnel (gas), evaporate (water), ...
... How can you separate all the components in a mixture containing sand, iron filings, water, gasoline, red water-soluble dye, and blue water-soluble dye? In pure form the dyes are powders…magnet (iron), filter (sand), sep funnel (gas), evaporate (water), ...
Balanced Chemical Equation
... • Redox reactions that take place in aqueous media often involve water, hydronium ions, and hydroxide ions as reactants or products. • Although these species are not oxidized or reduced, they do participate in the chemical change in other ways. • These reactions are difficult to balance by inspectio ...
... • Redox reactions that take place in aqueous media often involve water, hydronium ions, and hydroxide ions as reactants or products. • Although these species are not oxidized or reduced, they do participate in the chemical change in other ways. • These reactions are difficult to balance by inspectio ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.