AP Chemistry
... principles of the behavior of gases. a. A metal cylinder with a volume of 5.25 L contains 3.22 g of He(g) and 11.56 g of N2(g) at 15.0oC. (1) Calculate the total pressure. ...
... principles of the behavior of gases. a. A metal cylinder with a volume of 5.25 L contains 3.22 g of He(g) and 11.56 g of N2(g) at 15.0oC. (1) Calculate the total pressure. ...
Class Notes
... a. The carbanion is added first, at one point in time, under strongly anionic conditions o The first three steps all occur under these anionic conditions b. Acid is only added much later, in a second laboratory step. This gives a cationic environment. c. Why don’t you just protonate after the first ...
... a. The carbanion is added first, at one point in time, under strongly anionic conditions o The first three steps all occur under these anionic conditions b. Acid is only added much later, in a second laboratory step. This gives a cationic environment. c. Why don’t you just protonate after the first ...
Introduction and literature survey
... probable reactions are postulated under commercial operating conditions. These include reactions of methanol with di- and trimethylamines as well as decompositions of the amines. Optimum space velocities were found for a given temperature and pressure at which maximum conversion was obtained. The ex ...
... probable reactions are postulated under commercial operating conditions. These include reactions of methanol with di- and trimethylamines as well as decompositions of the amines. Optimum space velocities were found for a given temperature and pressure at which maximum conversion was obtained. The ex ...
Net Ionic Prep Session NMSI INSTRUCTOR
... 5. Strong bases: hydroxides [and oxides] of IA and IIA* metals—write these bases dissociated. WRITE ALL WEAK ACIDS AND BASES AS MOLECULES—be on the look out for BF3 and its cousins BCl3, etc. They are classic Lewis acids and when reacting with ammonia (a classic weak Lewis base), the product is F3BN ...
... 5. Strong bases: hydroxides [and oxides] of IA and IIA* metals—write these bases dissociated. WRITE ALL WEAK ACIDS AND BASES AS MOLECULES—be on the look out for BF3 and its cousins BCl3, etc. They are classic Lewis acids and when reacting with ammonia (a classic weak Lewis base), the product is F3BN ...
Otto F. Meyerhof - Nobel Lecture
... research on the part of Parnas and Verzar. In what relation the lactic acid stands to muscle performance, where it comes from and what becomes of it when it disappears in the presence of oxygen, was completely obscure. In fact, there were several different, irreconcilable interpretations current, al ...
... research on the part of Parnas and Verzar. In what relation the lactic acid stands to muscle performance, where it comes from and what becomes of it when it disappears in the presence of oxygen, was completely obscure. In fact, there were several different, irreconcilable interpretations current, al ...
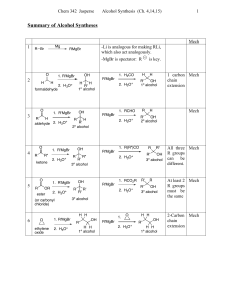
Alcohols
... Stabilized by anhydrous ether Iodides most reactive May be formed from any halide primary secondary tertiary vinyl aryl ...
... Stabilized by anhydrous ether Iodides most reactive May be formed from any halide primary secondary tertiary vinyl aryl ...
Pictures and Graphs
... dotted line. Sketch what would happen to both concentrations each if NO 2 was added until it’s concentration reached 0.03 M. ...
... dotted line. Sketch what would happen to both concentrations each if NO 2 was added until it’s concentration reached 0.03 M. ...
The Effects of Ozone on Compounds in Epicuticular Waxes in Plant
... Ozone is a significant stressor of more than 25% of the world’s forests and continues to remain a major threat to a large population of forests across the world.15 The effects of ozone on plant leaves can be detected through changes in plant gene expression, which in turn can lead to modifications i ...
... Ozone is a significant stressor of more than 25% of the world’s forests and continues to remain a major threat to a large population of forests across the world.15 The effects of ozone on plant leaves can be detected through changes in plant gene expression, which in turn can lead to modifications i ...
OCR Gateway Science
... (d) Explain why you would not use universal indicator for this titration. (e) Phenolphthalein indicator was used to find the end point of the titration. Why is this a suitable indicator and what is the colour change at the end point? The diagram shows the change in pH when an acid is added to an alk ...
... (d) Explain why you would not use universal indicator for this titration. (e) Phenolphthalein indicator was used to find the end point of the titration. Why is this a suitable indicator and what is the colour change at the end point? The diagram shows the change in pH when an acid is added to an alk ...
AP 2005 Chemistry Free-Response Questions
... Your responses to the rest of the questions in this part of the examination will be graded on the basis of the accuracy and relevance of the information cited. Explanations should be clear and well organized. Examples and equations may be included in your responses where appropriate. Specific answer ...
... Your responses to the rest of the questions in this part of the examination will be graded on the basis of the accuracy and relevance of the information cited. Explanations should be clear and well organized. Examples and equations may be included in your responses where appropriate. Specific answer ...
File
... All metals (on the left side of the periodic table) form cations and nonmetals (on the left side of the periodic table) form anions primarily. In order to determine the formula of the compound they create you must make sure their ions sum to zero. For example, table salt is sodium chloride. Using th ...
... All metals (on the left side of the periodic table) form cations and nonmetals (on the left side of the periodic table) form anions primarily. In order to determine the formula of the compound they create you must make sure their ions sum to zero. For example, table salt is sodium chloride. Using th ...
The p-Block Elements The p-Block Elements
... Ammonia is present in small quantities in air and soil where it is formed by the decay of nitrogenous organic matter e.g., urea. NH2 CONH2 + 2H2O → ( NH4 )2CO3 U 2NH3 + H2O + CO2 On a small scale ammonia is obtained from ammonium salts which decompose when treated with caustic soda or lime. 2NH4Cl + ...
... Ammonia is present in small quantities in air and soil where it is formed by the decay of nitrogenous organic matter e.g., urea. NH2 CONH2 + 2H2O → ( NH4 )2CO3 U 2NH3 + H2O + CO2 On a small scale ammonia is obtained from ammonium salts which decompose when treated with caustic soda or lime. 2NH4Cl + ...
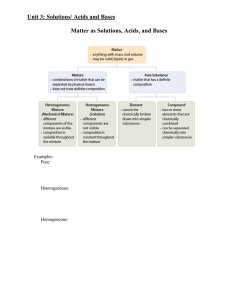
Outline for Unit 1 Solutions, Acid/Base, and Gases
... With strong electrolytes, we said that the solute ionizes in order to dissolve in water. This means that the compound has separated into ions. Different compounds have different ratios of ions within them. A substance such as NaOH has one Na ion and one hydroxide ion. However, Ba(OH)2 would separate ...
... With strong electrolytes, we said that the solute ionizes in order to dissolve in water. This means that the compound has separated into ions. Different compounds have different ratios of ions within them. A substance such as NaOH has one Na ion and one hydroxide ion. However, Ba(OH)2 would separate ...
Unit 4 Chemistry of Carbon
... OH muscles from heavy activity or H C C O H down the sugar maltose in dough heavy drinking. when OH into acetic acid, giving sour sugars.breakdown. H dough. Acetic acid: found in vinegar. O C ...
... OH muscles from heavy activity or H C C O H down the sugar maltose in dough heavy drinking. when OH into acetic acid, giving sour sugars.breakdown. H dough. Acetic acid: found in vinegar. O C ...
Chapter 4 Acids and bases
... leveling effect that brings all strong acids down to the acidity of H3O+. To distinguish the acidity strengths of HBr and HI, one has to use a less basic solvent. Similar situation exists for strong bases. In water, strong bases behave as solutions of OH-. The strengths of strong bases can only be d ...
... leveling effect that brings all strong acids down to the acidity of H3O+. To distinguish the acidity strengths of HBr and HI, one has to use a less basic solvent. Similar situation exists for strong bases. In water, strong bases behave as solutions of OH-. The strengths of strong bases can only be d ...
corrected Amino acids and Protein
... Since N- methylene derivative thus formed containing a free COOH group can be titrated against standard alkali. This reaction is used for the estimation of amino acids and known as Sorenson formal titration method. B) Reaction due to –COOH group: i) Formation of salt with bases: Amino acids forms s ...
... Since N- methylene derivative thus formed containing a free COOH group can be titrated against standard alkali. This reaction is used for the estimation of amino acids and known as Sorenson formal titration method. B) Reaction due to –COOH group: i) Formation of salt with bases: Amino acids forms s ...
Step 2 - The Grange School Blogs
... The electron is negatively charged The proton is positively charged ...
... The electron is negatively charged The proton is positively charged ...
1 - Cathedral High School
... 3.2.1 Describe and explain the periodic trends in atomic radii, ionic radii, ionization energies, electronegativity and melting points for the alkali metals (Li Cs), halogens (F I) and period 3 elements (Na Ar). Cross reference with topics 2, 4 and 5. Data for all these properties are listed i ...
... 3.2.1 Describe and explain the periodic trends in atomic radii, ionic radii, ionization energies, electronegativity and melting points for the alkali metals (Li Cs), halogens (F I) and period 3 elements (Na Ar). Cross reference with topics 2, 4 and 5. Data for all these properties are listed i ...
uplift luna ap chemistry
... CnH2n+1OH; Do not be fooled—this looks like a hydroxide ion, but is not! It does not make this hydrocarbon an alkaline or basic compound. Do not name these as a hydroxide! C2H6 is ethane while C2H5OH is ethanol. NAMING BINARY IONIC COMPOUNDS How do I know it is ionic? The chemical formula will begin ...
... CnH2n+1OH; Do not be fooled—this looks like a hydroxide ion, but is not! It does not make this hydrocarbon an alkaline or basic compound. Do not name these as a hydroxide! C2H6 is ethane while C2H5OH is ethanol. NAMING BINARY IONIC COMPOUNDS How do I know it is ionic? The chemical formula will begin ...
Biodiesel Production and Fuel Quality_JVG
... hydroxide as the reaction catalyst and phosphoric acid for neutralization so that the salt formed is potassium phosphate, which can be used for fertilizer. After acidulation and separation of the free fatty acids, the me thanol in the glycerol is removed by a vacuum flash process, or another type of ...
... hydroxide as the reaction catalyst and phosphoric acid for neutralization so that the salt formed is potassium phosphate, which can be used for fertilizer. After acidulation and separation of the free fatty acids, the me thanol in the glycerol is removed by a vacuum flash process, or another type of ...
Summer Work
... 6. How many kilojoules of heat energy are required to heat all the aluminum (C of Al =0.902J/g•°C) in a roll of aluminum foil (500.0 g) from room temperature (25.0 °C) to the temperature of a hot oven (250.0 °C)? ...
... 6. How many kilojoules of heat energy are required to heat all the aluminum (C of Al =0.902J/g•°C) in a roll of aluminum foil (500.0 g) from room temperature (25.0 °C) to the temperature of a hot oven (250.0 °C)? ...
Entering and leaving group effects in Oh ligand substitutions
... Ligand deprotonation generates a conjugate base of the starting complex containing an amido ligand, NH2–. ...
... Ligand deprotonation generates a conjugate base of the starting complex containing an amido ligand, NH2–. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.