Page 1
... d. Determine the number of moles for 3.58 x 1023 formula units ZnCl2? (particles to moles) 3.58 x 1023 formula units ZnCl2 1 mol ZnCl2 = 0.59 mol ZnCl2 6.02 x 1023 formula units ZnCl2 ...
... d. Determine the number of moles for 3.58 x 1023 formula units ZnCl2? (particles to moles) 3.58 x 1023 formula units ZnCl2 1 mol ZnCl2 = 0.59 mol ZnCl2 6.02 x 1023 formula units ZnCl2 ...
7. Alkenes: Reactions and Synthesis
... • Potassium permanganate (KMnO4) can produce carboxylic acids and carbon dioxide if H’s are present on C=C O ...
... • Potassium permanganate (KMnO4) can produce carboxylic acids and carbon dioxide if H’s are present on C=C O ...
Esterification and Esters
... Use of Azeotropes to Remove Water. With the aliphatic alcohols and esters of medium volatility, a variety of azeotropes is encountered on distillation (see DISTILLATION, AZEOTROPIC AND EXTRACTIVE). Removal of these azeotropes from the esterification reaction mixture drives the equilibrium in favor o ...
... Use of Azeotropes to Remove Water. With the aliphatic alcohols and esters of medium volatility, a variety of azeotropes is encountered on distillation (see DISTILLATION, AZEOTROPIC AND EXTRACTIVE). Removal of these azeotropes from the esterification reaction mixture drives the equilibrium in favor o ...
Chapter 3: Calculations with Chemical Formulas
... Acid–Base Neutralization: A process in which an acid reacts with a base to yield water plus an ionic compound called a salt. ...
... Acid–Base Neutralization: A process in which an acid reacts with a base to yield water plus an ionic compound called a salt. ...
Ketones and Aldehydes
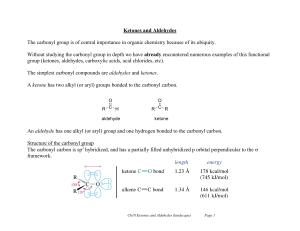
... Ketones and Aldehydes The carbonyl group is of central importance in organic chemistry because of its ubiquity. Without studying the carbonyl group in depth we have already encountered numerous examples of this functional group (ketones, aldehydes, carboxylic acids, acid chlorides, etc). The simples ...
... Ketones and Aldehydes The carbonyl group is of central importance in organic chemistry because of its ubiquity. Without studying the carbonyl group in depth we have already encountered numerous examples of this functional group (ketones, aldehydes, carboxylic acids, acid chlorides, etc). The simples ...
Chapter 4 - profpaz.com
... Identifying the oxidation-reduction nature of reactions between metals and non-metals is straight forward because of ion formation. However, redox reactions that occur between two non-metals are more difficult to characterize since no ions are formed. ...
... Identifying the oxidation-reduction nature of reactions between metals and non-metals is straight forward because of ion formation. However, redox reactions that occur between two non-metals are more difficult to characterize since no ions are formed. ...
Exam - Vcaa
... A forensic chemist tests mud from a crime scene to determine whether the mud contains zinc. Which one of the following analytical techniques would be best suited to this task? ...
... A forensic chemist tests mud from a crime scene to determine whether the mud contains zinc. Which one of the following analytical techniques would be best suited to this task? ...
Ethers and Epoxides
... • Diethyl ether is used industrially as a solvent • Tetrahydrofuran (THF) is a solvent that is a cyclic ether • Epoxides contain a C-O-C unit which make-up a ...
... • Diethyl ether is used industrially as a solvent • Tetrahydrofuran (THF) is a solvent that is a cyclic ether • Epoxides contain a C-O-C unit which make-up a ...
2010 Exam
... Select the letter of the correct response from those provided. EITHER shade the letter on your computer scorable card OR place the letter in the blank provided on your Multiple Choice Answer Sheet, whichever format is being used by your school for this exam. Do ALL questions in this section. ...
... Select the letter of the correct response from those provided. EITHER shade the letter on your computer scorable card OR place the letter in the blank provided on your Multiple Choice Answer Sheet, whichever format is being used by your school for this exam. Do ALL questions in this section. ...
Handout VI
... Trivially, aldehydes are usually named after the acids they form on oxidation. The –ic in the name of the acid is then replaced by aldehyde to get the name of the aldehyde. ...
... Trivially, aldehydes are usually named after the acids they form on oxidation. The –ic in the name of the acid is then replaced by aldehyde to get the name of the aldehyde. ...
Chemistry 12 - hrsbstaff.ednet.ns.ca
... 40. What effect does a catalyst have? A. increases the reaction rate by decreasing the heat of reaction B. increases the reaction rate by increasing the activation energy of the reverse reaction C. increases the reaction rate by lowering the activation energy of the forward reaction only D. increase ...
... 40. What effect does a catalyst have? A. increases the reaction rate by decreasing the heat of reaction B. increases the reaction rate by increasing the activation energy of the reverse reaction C. increases the reaction rate by lowering the activation energy of the forward reaction only D. increase ...
SCH 3U - mquagliaoths
... 34) Lead (II) sulfate will be a precipitate if you add sulfate ions to the solution. Similarly, silver chloride will precipitate out if chloride ions are added to the mixture. So, add one of these ions in, then filter off the precipitate. Then add the other ion and isolate the second precipitate. 43 ...
... 34) Lead (II) sulfate will be a precipitate if you add sulfate ions to the solution. Similarly, silver chloride will precipitate out if chloride ions are added to the mixture. So, add one of these ions in, then filter off the precipitate. Then add the other ion and isolate the second precipitate. 43 ...
Unit 3 Physical Science: Chemical Reactions
... [calcium sulphate], associated with acid rain, along with others such as NaCl [sodium chloride], NaOH [sodium hydroxide]. The use of Roman numerals should also be covered for compounds such as FeO [iron (II) oxide] and Fe2 O3 [iron (III) oxide]. An activity using aides such as ion clips would be ver ...
... [calcium sulphate], associated with acid rain, along with others such as NaCl [sodium chloride], NaOH [sodium hydroxide]. The use of Roman numerals should also be covered for compounds such as FeO [iron (II) oxide] and Fe2 O3 [iron (III) oxide]. An activity using aides such as ion clips would be ver ...
Types of Chemical Reactions
... Combustion Reactions occur when a substance combines with oxygen releasing a large amount of energy in the form of light and heat, heat, it is a ...
... Combustion Reactions occur when a substance combines with oxygen releasing a large amount of energy in the form of light and heat, heat, it is a ...
Lecture Notes_ch17
... • Identify the parent chain: the longest chain of carbons to which the nitrogen is attached • Replace the “-e” ending of the alkane name with “amine” • Number the parent chain from the end closest to the nitrogen atom • Identify the position of the nitrogen atom with a ...
... • Identify the parent chain: the longest chain of carbons to which the nitrogen is attached • Replace the “-e” ending of the alkane name with “amine” • Number the parent chain from the end closest to the nitrogen atom • Identify the position of the nitrogen atom with a ...
Chemistry notes Important terms *Mass of element in a sample
... *Mass of element in a sample Periods- horizontal rows on a periodic table Groups- vertical rows on a periodic table Ionic compound – electron transfer from a metal to a non-metal Covalent compound- electron sharing between two non-metals Hydrates- have a specific number of water molecules associated ...
... *Mass of element in a sample Periods- horizontal rows on a periodic table Groups- vertical rows on a periodic table Ionic compound – electron transfer from a metal to a non-metal Covalent compound- electron sharing between two non-metals Hydrates- have a specific number of water molecules associated ...
INTRODUCTION TO ORGANIC CHEMISTRY
... Consider all alkyl groups attached to it as branch chains or substituents that have replaced hydrogen atoms of the parent hydrocarbon. If two chains of equal length are found, use the chain that has the larger number of substituents attached to it. The alkane’s name consists of the parent compound’s ...
... Consider all alkyl groups attached to it as branch chains or substituents that have replaced hydrogen atoms of the parent hydrocarbon. If two chains of equal length are found, use the chain that has the larger number of substituents attached to it. The alkane’s name consists of the parent compound’s ...
Alkyl Halides02
... This is also a qualitative test for identifying alcohols, i.e, the Lucas test. HCl with ZnCl 2 catalyst are used. 3 alcohols react quickly, 2 slowly, and 1 don’t react. Note that 1 and 2 alcohols will react with special reagents to produce alkyl halides (i.e., thionyl chloride, SOCl2, or PBr3) ...
... This is also a qualitative test for identifying alcohols, i.e, the Lucas test. HCl with ZnCl 2 catalyst are used. 3 alcohols react quickly, 2 slowly, and 1 don’t react. Note that 1 and 2 alcohols will react with special reagents to produce alkyl halides (i.e., thionyl chloride, SOCl2, or PBr3) ...
organonitrogen compounds i. amines
... of amines and the chemistry of alcohols, which we discussed in Chapter 15. Primary amines (RNH,) and secondary amines (R,NH) are much weaker acids than alcohols (ROH) and form strongly basic anions: ...
... of amines and the chemistry of alcohols, which we discussed in Chapter 15. Primary amines (RNH,) and secondary amines (R,NH) are much weaker acids than alcohols (ROH) and form strongly basic anions: ...
C:\My Documents\My Documents\Teaching\chem130\hunt
... When aluminum metal is dissolved in perchloric acid, aluminum (III) perchlorate and hydrogen gas are formed. In the balanced equation for this reaction, what are the coefficients of hydrogen gas and perchloric acid, respectively? ...
... When aluminum metal is dissolved in perchloric acid, aluminum (III) perchlorate and hydrogen gas are formed. In the balanced equation for this reaction, what are the coefficients of hydrogen gas and perchloric acid, respectively? ...
- Catalyst
... – a solid precipitate forms when aqueous solutions of certain ions are mixed • Acid-Base: proton transfer reactions – acid donates a proton to a base, forming a molecule (water or another weak acid) and an aqueous salt – Acid: proton-donor; Base: proton-acceptor • Oxidation-Reduction: electron trans ...
... – a solid precipitate forms when aqueous solutions of certain ions are mixed • Acid-Base: proton transfer reactions – acid donates a proton to a base, forming a molecule (water or another weak acid) and an aqueous salt – Acid: proton-donor; Base: proton-acceptor • Oxidation-Reduction: electron trans ...
Oxidation catalytic system and oxidation process using the same
... N-hydroxyphthalimide), and a co-catalyst (except phospho vanadomolybdic acid) containing an element selected from the group consisting of Group 2A elements of the Periodic Table of Elements, transition metals (Group 3A to 7A elements, Group 8 elements, Group 1B elements and Group 2B elements of the ...
... N-hydroxyphthalimide), and a co-catalyst (except phospho vanadomolybdic acid) containing an element selected from the group consisting of Group 2A elements of the Periodic Table of Elements, transition metals (Group 3A to 7A elements, Group 8 elements, Group 1B elements and Group 2B elements of the ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.