Basic Concepts
... already at equilibrium and, if it is not, how to predict whether its composition will change with time (whether the reaction will proceed to the right or to the left) 1. If Q = K, the system is at equilibrium, no further change in the composition of the system will occur unless the conditions are ch ...
... already at equilibrium and, if it is not, how to predict whether its composition will change with time (whether the reaction will proceed to the right or to the left) 1. If Q = K, the system is at equilibrium, no further change in the composition of the system will occur unless the conditions are ch ...
Basic Concepts - Department of Chemistry
... Calculating an Equilibrium Constant from Equilibrium Concentrations • can be calculated when equilibrium concentrations, or partial pressures are substituted into the equilibrium constant expression for the reaction. • When equilibrium concentrations are not given the equilibrium concentrations can ...
... Calculating an Equilibrium Constant from Equilibrium Concentrations • can be calculated when equilibrium concentrations, or partial pressures are substituted into the equilibrium constant expression for the reaction. • When equilibrium concentrations are not given the equilibrium concentrations can ...
Week 1 NEPHAR 201- Analytical Chemistry II_Introduction_5
... Weak A solute that does not fully dissociate into ions in solution. These substances only partially ionize in solution. Ex. H2CO3, NH3, AgCl ...
... Weak A solute that does not fully dissociate into ions in solution. These substances only partially ionize in solution. Ex. H2CO3, NH3, AgCl ...
Chemistry – V – BSC – 503
... 1. To determine normality of xN HCl by pH metry. 2. To determine normality and dissociation constant of weak acid (xN CH3COOH) by pH metry. 3. To determine normality and dissociation constant of dibasic acid (xN oxalic acid/malonic acid/maleic acid) using 0.1N NaOH Solution. Colourimetry 1. Find out ...
... 1. To determine normality of xN HCl by pH metry. 2. To determine normality and dissociation constant of weak acid (xN CH3COOH) by pH metry. 3. To determine normality and dissociation constant of dibasic acid (xN oxalic acid/malonic acid/maleic acid) using 0.1N NaOH Solution. Colourimetry 1. Find out ...
2010 Chemistry Written examination 2
... a. The most common method for the industrial production of hydrogen is the steam reforming process, which requires high temperature, high pressure and a Ni catalyst. The equation for this reaction is CH4(g) + H2O(g) ...
... a. The most common method for the industrial production of hydrogen is the steam reforming process, which requires high temperature, high pressure and a Ni catalyst. The equation for this reaction is CH4(g) + H2O(g) ...
PRACTICE – Naming and Writing Ionic Compounds
... Na2S2O3(aq) + 4Cl2(g) + 5H2O(aq) 2NaHSO4(aq) + 8HCl(aq) a. How many moles of Na2S2O3 are needed to react with 0.12mol of Cl2? ...
... Na2S2O3(aq) + 4Cl2(g) + 5H2O(aq) 2NaHSO4(aq) + 8HCl(aq) a. How many moles of Na2S2O3 are needed to react with 0.12mol of Cl2? ...
PRACTICE FINAL EXAM CHEMISTRY 152 This
... a) 1.0 M H2SO4 / 1.0 M NaHSO4 b) 1.0 M H2 SO3 / 1.0 M Na2SO3 c) 1.0 M Na2SO3/ 1.0 M NaHSO3 d) 1.0 M HNO2 / 1.0 M NaF e) 1.0 M HNO3 / 1.0 M NaNO3 ...
... a) 1.0 M H2SO4 / 1.0 M NaHSO4 b) 1.0 M H2 SO3 / 1.0 M Na2SO3 c) 1.0 M Na2SO3/ 1.0 M NaHSO3 d) 1.0 M HNO2 / 1.0 M NaF e) 1.0 M HNO3 / 1.0 M NaNO3 ...
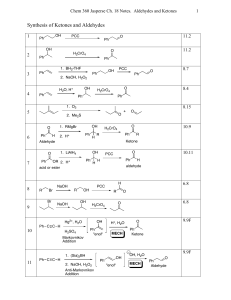
Synthesis of Ketones and Aldehydes
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
Chapter 10 Chemical Reactions
... Combustion: Hydrocarbons (compounds with only C and H atoms) burn in the presence of O2 gas to produce steam (water vapor) and carbon dioxide gas. Example: C2H4(g) + 3 O2(g) 2 CO2(g) + 2 H2O (g) Objectives: ...
... Combustion: Hydrocarbons (compounds with only C and H atoms) burn in the presence of O2 gas to produce steam (water vapor) and carbon dioxide gas. Example: C2H4(g) + 3 O2(g) 2 CO2(g) + 2 H2O (g) Objectives: ...
Endothermic reactions
... called exergonic (ek sur GAH nihk) reactions. In these reactions, less energy is required to break the original bonds than is released when new bonds are formed. As a result, some form of energy, such as light or heat, is given off by the reaction. The familiar glow from the reaction inside a glow s ...
... called exergonic (ek sur GAH nihk) reactions. In these reactions, less energy is required to break the original bonds than is released when new bonds are formed. As a result, some form of energy, such as light or heat, is given off by the reaction. The familiar glow from the reaction inside a glow s ...
AP Chemistry: Aqueous Reactions and Solution Stoichiometry
... Take a known volume of the HCl solution (e.g., 20.0 mL) and measure the number of milliliters of NaOH solution required to react completely with the HCl solution. The point at which stoichiometrically equivalent quantities of NaOH and HCl are brought together is known as the equivalence point of the ...
... Take a known volume of the HCl solution (e.g., 20.0 mL) and measure the number of milliliters of NaOH solution required to react completely with the HCl solution. The point at which stoichiometrically equivalent quantities of NaOH and HCl are brought together is known as the equivalence point of the ...
getting started 3.1 hydrocarbons
... stronger intermolecular attractions that require more energy (higher temperatures) to overcome. Very large nonpolar molecules are affected by London dispersion forces that may cause their boiling points to be comparable to those of smaller polar molecules. 4. Functional groups can contain carbon–car ...
... stronger intermolecular attractions that require more energy (higher temperatures) to overcome. Very large nonpolar molecules are affected by London dispersion forces that may cause their boiling points to be comparable to those of smaller polar molecules. 4. Functional groups can contain carbon–car ...
Synthesis of Ketones and Aldehydes
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
04_01_03.html
... Overview of Chapter This chapter introduces chemical reactions and their mechanisms by focusing on two reactions that yield alkyl halides. (1) alcohol + hydrogen halide ROH + HX RX + H2O (2) alkane + halogen RH + X2 RX + HX Both are substitution reactions ...
... Overview of Chapter This chapter introduces chemical reactions and their mechanisms by focusing on two reactions that yield alkyl halides. (1) alcohol + hydrogen halide ROH + HX RX + H2O (2) alkane + halogen RH + X2 RX + HX Both are substitution reactions ...
Tin-Catalyzed Esterification and Transesterification Reactions: A
... since they are readily saponified by the homogeneous alkali catalyst used to transesterify the TG, leading to a loss of catalyst as well as increased products, purification costs [19]. The main approach to improve the processing of oils which contain FFAs in large amount is firstly to promote a step ...
... since they are readily saponified by the homogeneous alkali catalyst used to transesterify the TG, leading to a loss of catalyst as well as increased products, purification costs [19]. The main approach to improve the processing of oils which contain FFAs in large amount is firstly to promote a step ...
CH 4 Notes
... Common bases are NH3 (ammonia), Drano, milk of magnesia. Compounds that do not contain OH1- ions can also be bases. Proton transfer between NH3 (a weak base) and water (a weak acid) is an example of an acid-base reaction. Since there is a mixture of NH3, H2O, NH41+, and OH1- in solution, we writ ...
... Common bases are NH3 (ammonia), Drano, milk of magnesia. Compounds that do not contain OH1- ions can also be bases. Proton transfer between NH3 (a weak base) and water (a weak acid) is an example of an acid-base reaction. Since there is a mixture of NH3, H2O, NH41+, and OH1- in solution, we writ ...
Journal of Molecular Catalysis A, 302
... [27]. The second role of the acid is to hydrogen bond with the fluorine atoms on the methyl group of the arene, a process that is unique to fluorine [28]. The interaction of the proton with the fluorine atoms facilitates the replacement of fluorine by oxygen from the vanadate species, leading to the for ...
... [27]. The second role of the acid is to hydrogen bond with the fluorine atoms on the methyl group of the arene, a process that is unique to fluorine [28]. The interaction of the proton with the fluorine atoms facilitates the replacement of fluorine by oxygen from the vanadate species, leading to the for ...
Chapter 4 Quantities of Reactants and Products 4.1 Chemical
... 4.7 Percent Composition and Empirical Formulas (p. 150) In a combustion analysis of a compound containing carbon and hydrogen, the compound reacts with oxygen and all of the carbon in the compound is converted to carbon dioxide and the hydrogen in the compound is converted to water. 2 C4H10(g) + 13 ...
... 4.7 Percent Composition and Empirical Formulas (p. 150) In a combustion analysis of a compound containing carbon and hydrogen, the compound reacts with oxygen and all of the carbon in the compound is converted to carbon dioxide and the hydrogen in the compound is converted to water. 2 C4H10(g) + 13 ...
Chapter 8 - Chemical Equations
... Step 2 – You will compare the type of element by itself to the similar type of element in the compound. In this case, aluminum is a metal, so I will compare it with the metal in the compound (which is Pb). Step 3 – RULE: The element that is by itself must be HIGHER on (closer to the top of) the Acti ...
... Step 2 – You will compare the type of element by itself to the similar type of element in the compound. In this case, aluminum is a metal, so I will compare it with the metal in the compound (which is Pb). Step 3 – RULE: The element that is by itself must be HIGHER on (closer to the top of) the Acti ...
20 More About Oxidation–Reduction Reactions
... The carbonyl group of an amide is reduced to a methylene group (CH 2) by lithium aluminum hydride (Section 18.5). Primary, secondary, and tertiary amines are formed, depending on the number of substituents bonded to the nitrogen of the amide. To obtain the amine in its neutral basic form, acid is no ...
... The carbonyl group of an amide is reduced to a methylene group (CH 2) by lithium aluminum hydride (Section 18.5). Primary, secondary, and tertiary amines are formed, depending on the number of substituents bonded to the nitrogen of the amide. To obtain the amine in its neutral basic form, acid is no ...
Experiment 9
... great so that the forces of interionic interaction manifest themselves appreciably even at low concentration of an electrolyte. As a result, the ions are not completely free. This is why the state of ions in a solution is described, in addition to their concentration, by their activity, i.e. the con ...
... great so that the forces of interionic interaction manifest themselves appreciably even at low concentration of an electrolyte. As a result, the ions are not completely free. This is why the state of ions in a solution is described, in addition to their concentration, by their activity, i.e. the con ...
82KB - NZQA
... A: Pb2+(aq) + SO42–(aq) → PbSO4(s) OR Pb(NO3)2(aq) + K2SO4(aq) → PbSO4(s) + 2KNO3(aq) ...
... A: Pb2+(aq) + SO42–(aq) → PbSO4(s) OR Pb(NO3)2(aq) + K2SO4(aq) → PbSO4(s) + 2KNO3(aq) ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.