Electrophilic Aromatic Substitution and Substituted Benzenes
... substitution occurs, resulting in replacement of H by Br on the aromatic ring to form ortho and para isomers. • With Br2 and light or heat (radical conditions), substitution of H by Br occurs at the benzylic carbon of the alkyl group. ...
... substitution occurs, resulting in replacement of H by Br on the aromatic ring to form ortho and para isomers. • With Br2 and light or heat (radical conditions), substitution of H by Br occurs at the benzylic carbon of the alkyl group. ...
Document
... substitution occurs, resulting in replacement of H by Br on the aromatic ring to form ortho and para isomers. • With Br2 and light or heat (radical conditions), substitution of H by Br occurs at the benzylic carbon of the alkyl group. ...
... substitution occurs, resulting in replacement of H by Br on the aromatic ring to form ortho and para isomers. • With Br2 and light or heat (radical conditions), substitution of H by Br occurs at the benzylic carbon of the alkyl group. ...
Week 7 - Acid-base, redox
... Classifying, Writing, and Balancing Redox Reactions We previously classified, wrote, and balanced precipitation, acid-base, and gas-forming reactions. Redox reactions have electron transfer, and that is what sets them apart from the other reaction types. With redox, one atom loses one or more electr ...
... Classifying, Writing, and Balancing Redox Reactions We previously classified, wrote, and balanced precipitation, acid-base, and gas-forming reactions. Redox reactions have electron transfer, and that is what sets them apart from the other reaction types. With redox, one atom loses one or more electr ...
Tech Tip 0013 - Hydrates and Salts
... Confusion is not uncommon when reporting the final values calculated with using such standards. The analyst must first determine if the end result will be reported as the hydrate or salt, or as the compound without the hydrate or salt. This determines how the initial calibration curve is generated. ...
... Confusion is not uncommon when reporting the final values calculated with using such standards. The analyst must first determine if the end result will be reported as the hydrate or salt, or as the compound without the hydrate or salt. This determines how the initial calibration curve is generated. ...
C3 Revision Question Booklet
... To gain full marks in this question you should write your ideas in good English. Put them into a sensible order and use the correct scientific words. ...
... To gain full marks in this question you should write your ideas in good English. Put them into a sensible order and use the correct scientific words. ...
Dr David`s Chemistry Test Answers
... are used up and converted to insoluble silver chloride. 3. The reaction of nitrogen with hydrogen to give ammonia is reversible. The ammonia has a tendency to decompose back into nitrogen and hydrogen particularly at elevated temperatures. The formation of sulphur trioxide from sulphur dioxide and o ...
... are used up and converted to insoluble silver chloride. 3. The reaction of nitrogen with hydrogen to give ammonia is reversible. The ammonia has a tendency to decompose back into nitrogen and hydrogen particularly at elevated temperatures. The formation of sulphur trioxide from sulphur dioxide and o ...
Lewis Acids and Bases Hard and Soft Acid/Base Theory
... Hard and Soft Acid/Base Theory Definitions of Lewis Acid-Base Theory. Lewis acid/base theory (sometimes called donor-acceptor theory) is a broad, widely applicable approach to the classification of chemical substances and the analysis of chemical reactions. According to this theory, a base is define ...
... Hard and Soft Acid/Base Theory Definitions of Lewis Acid-Base Theory. Lewis acid/base theory (sometimes called donor-acceptor theory) is a broad, widely applicable approach to the classification of chemical substances and the analysis of chemical reactions. According to this theory, a base is define ...
Chemical reaction model:
... diffusion of oxygen molecules. The radical migration was through a hydrogen abstraction mechanism, and was governed by a diffusion coefficient of 3.0 x 10-18 cm2/sec at 20 C. They also commented that the apparent stability of peroxy radicals could be because of their non-mobility, preventing the pe ...
... diffusion of oxygen molecules. The radical migration was through a hydrogen abstraction mechanism, and was governed by a diffusion coefficient of 3.0 x 10-18 cm2/sec at 20 C. They also commented that the apparent stability of peroxy radicals could be because of their non-mobility, preventing the pe ...
pH - OCCC.edu
... Weak Acids To calculate the pH of a weak acid solution: Write the ionization equilibrium for the acid. Write the equilibrium constant expression and its numerical value. Set up a table showing initial concentration, change, equilibrium concentration. Substitute equilibrium concentrations ...
... Weak Acids To calculate the pH of a weak acid solution: Write the ionization equilibrium for the acid. Write the equilibrium constant expression and its numerical value. Set up a table showing initial concentration, change, equilibrium concentration. Substitute equilibrium concentrations ...
Revised Syllabus - M. Sc. First Year - Chemistry
... a) Types of Aliphatic electrophilic substitution reaction: SE1, SE2, SEi. b) Electrophilic substitution accompanied by double bond shift. c) Effect of substrate, leaving group and solvent polarity on the reactivity. ...
... a) Types of Aliphatic electrophilic substitution reaction: SE1, SE2, SEi. b) Electrophilic substitution accompanied by double bond shift. c) Effect of substrate, leaving group and solvent polarity on the reactivity. ...
Lecture 4
... a. The oxidation number of oxygen is usually –2 in both ionic and molecular compounds. The major exception is in compounds called peroxides, which contain O22- ion, giving each oxygen an oxidation number of –1. b. The Oxidation number of hydrogen is +1 when bonded to nonmetals and –1 when bonded to ...
... a. The oxidation number of oxygen is usually –2 in both ionic and molecular compounds. The major exception is in compounds called peroxides, which contain O22- ion, giving each oxygen an oxidation number of –1. b. The Oxidation number of hydrogen is +1 when bonded to nonmetals and –1 when bonded to ...
Solution
... A 0.5662-g sample of an ionic compound containing chloride ions and an unknown metal is dissolved in water and treated with an excess of AgNO3. If 1.0882 g of AgCl precipitate forms, what is the percent by mass of Cl in the original compound? ...
... A 0.5662-g sample of an ionic compound containing chloride ions and an unknown metal is dissolved in water and treated with an excess of AgNO3. If 1.0882 g of AgCl precipitate forms, what is the percent by mass of Cl in the original compound? ...
Descriptive Chemistry of Elements p
... [B(OH)4] (aq) + H3O+(aq) Thus the acidity of H3BO3 is due to abstraction of OH ion from a water molecule (producing a H+ ion) and not due to H+ donation by H3BO3. H3BO3 is a monobasic/monoprotic acid not a triprotic acid. H3BO3 3 H+ + BO3 ...
... [B(OH)4] (aq) + H3O+(aq) Thus the acidity of H3BO3 is due to abstraction of OH ion from a water molecule (producing a H+ ion) and not due to H+ donation by H3BO3. H3BO3 is a monobasic/monoprotic acid not a triprotic acid. H3BO3 3 H+ + BO3 ...
anhydride cured-epoxy matrices
... A competing reaction is epoxy groups reacting with a secondary alcohol forming an (-hydroxy) ether linkage. ...
... A competing reaction is epoxy groups reacting with a secondary alcohol forming an (-hydroxy) ether linkage. ...
AMINES
... Ans) This is because FeCl2 formed gets hydrolysed to release HCl in the reaction. Thus, only a small amount of HCl is required to initiate the reaction. Q2) Ammonlysis of R-X is not a preferred method for preparing amines Ans) This method yields a mixture of primary, secondary, tertiary a mines and ...
... Ans) This is because FeCl2 formed gets hydrolysed to release HCl in the reaction. Thus, only a small amount of HCl is required to initiate the reaction. Q2) Ammonlysis of R-X is not a preferred method for preparing amines Ans) This method yields a mixture of primary, secondary, tertiary a mines and ...
Unit 8 Packet - Page 1 of 18 Honors Chemistry
... balance polyatomic ions on each side of the equation as one unit balance H and O last (they often appear in more than one compound) 4. Check; if coefficients are not the lowest possible; reduce down. *****Remember NEVER change subscripts***** Examples: 1. water breaks down into hydrogen and oxygen ...
... balance polyatomic ions on each side of the equation as one unit balance H and O last (they often appear in more than one compound) 4. Check; if coefficients are not the lowest possible; reduce down. *****Remember NEVER change subscripts***** Examples: 1. water breaks down into hydrogen and oxygen ...
Multiple Choice Questions
... Ans: Ethanol can form hydrogen bonds with water molecules, but ethane cannot. When ethanol dissolves, the decrease in the system's entropy that results from formation of ordered arrays of water around the CH3CH2– group is partly compensated by the favorable interactions (hydrogen bonds) of the hydro ...
... Ans: Ethanol can form hydrogen bonds with water molecules, but ethane cannot. When ethanol dissolves, the decrease in the system's entropy that results from formation of ordered arrays of water around the CH3CH2– group is partly compensated by the favorable interactions (hydrogen bonds) of the hydro ...
Alcohols, Phenols and Ethers
... Increasing order of acidity is ethanol < water < phenol. The phenoxide ion obtained after the removal of a proton is stabilised by resonance whereas the ethoxide ion obtained after the removal of a proton is destabilised by ‘+I’ effect of —C2H5 group. Therefore phenol is stronger acid than ethanol. ...
... Increasing order of acidity is ethanol < water < phenol. The phenoxide ion obtained after the removal of a proton is stabilised by resonance whereas the ethoxide ion obtained after the removal of a proton is destabilised by ‘+I’ effect of —C2H5 group. Therefore phenol is stronger acid than ethanol. ...
Chapter 13, sections 13.5 - Properties of Aldehydes and Ketones
... which contains Cu2+, reacts with aldehydes that have an adjacent OH group. • An aldehyde is oxidized to a carboxylic acid, while Cu2+ is reduced to give red Cu2O(s). ...
... which contains Cu2+, reacts with aldehydes that have an adjacent OH group. • An aldehyde is oxidized to a carboxylic acid, while Cu2+ is reduced to give red Cu2O(s). ...
Hydrothermal Reactions from Sodium Hydrogen Carbonate to Phenol
... fluids, which supports the occurrence of an abiogenic source of hydrocarbons.10 Usually, formation of abiogenic hydrocarbons by the reduction of carbon dioxide was thought to occur under hydrothermal conditions involving FischerTropsch reactions and the serpentinization of ultramafic rocks.11,12 Gen ...
... fluids, which supports the occurrence of an abiogenic source of hydrocarbons.10 Usually, formation of abiogenic hydrocarbons by the reduction of carbon dioxide was thought to occur under hydrothermal conditions involving FischerTropsch reactions and the serpentinization of ultramafic rocks.11,12 Gen ...
Proximity Effects on Reaction Rates
... • Groups that are more electron-poor are more electrophilic and therefore react more quickly with nucleophiles ...
... • Groups that are more electron-poor are more electrophilic and therefore react more quickly with nucleophiles ...
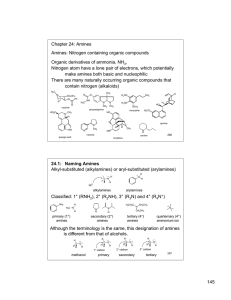
145 Chapter 24: Amines Amines: Nitrogen containing organic
... Synthesis of primary amines from the reaction of alkyl halides or tosylates with “ammonia equivalents” Azide ion is a very strong nucleophile and react with 1° or 2° alkyl halides or tosylates via an SN2 reaction. The resulting azide can be reduced to a 1° amine. H N N N ...
... Synthesis of primary amines from the reaction of alkyl halides or tosylates with “ammonia equivalents” Azide ion is a very strong nucleophile and react with 1° or 2° alkyl halides or tosylates via an SN2 reaction. The resulting azide can be reduced to a 1° amine. H N N N ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.