Q4) How the following conversions can be carried out?
... a result, it is easier to lose a proton. Also, the o-nitrophenoxide ion formed after the loss of protons is stabilized by resonance. Hence, ortho nitrophenol is a stronger acid. On the other hand, methoxy group is an electron-releasing group. Thus, it increases the electron density in the O−H bond a ...
... a result, it is easier to lose a proton. Also, the o-nitrophenoxide ion formed after the loss of protons is stabilized by resonance. Hence, ortho nitrophenol is a stronger acid. On the other hand, methoxy group is an electron-releasing group. Thus, it increases the electron density in the O−H bond a ...
Chemistry 1B General Chemistry Exp 1 Spring 2017
... The laboratory notebook will be collected periodically without prior announcement to ensure you are practicing correct scientific procedures for collecting experimental data. A portion of your laboratory grade will be based on your neatness, completeness, and attention to detail. Failure to bring yo ...
... The laboratory notebook will be collected periodically without prior announcement to ensure you are practicing correct scientific procedures for collecting experimental data. A portion of your laboratory grade will be based on your neatness, completeness, and attention to detail. Failure to bring yo ...
CHEMISTRY (862)
... (ii) The concept of energy barrier (it is suggested that some experiments may be devised which may deal with the above mentioned factors). The concept of threshold energy, activation energy, energy of the reactant, energy of the products and the energy of the activated complex. Exothermic and endoth ...
... (ii) The concept of energy barrier (it is suggested that some experiments may be devised which may deal with the above mentioned factors). The concept of threshold energy, activation energy, energy of the reactant, energy of the products and the energy of the activated complex. Exothermic and endoth ...
I- Introduction
... 4- It should have a high formula weight (to minimize weighing error). 5- It should posses the properties required for a titration (soluble and react rapidly ....). ...
... 4- It should have a high formula weight (to minimize weighing error). 5- It should posses the properties required for a titration (soluble and react rapidly ....). ...
Chapters E-18 review - Bakersfield College
... 10. How can we convert thé first compound into thé second compound? Détermine thé type of chemical reaction, catalyst (if it is necessary) and other necessary conditions: a) Benzène to Chlorobenzene ...
... 10. How can we convert thé first compound into thé second compound? Détermine thé type of chemical reaction, catalyst (if it is necessary) and other necessary conditions: a) Benzène to Chlorobenzene ...
All chemical equations must be balanced, that is, they must have the
... These reactions usually involve an element reacting with an ionic compound. The element can only replace the part of the compound that forms the same type of ion that the element forms : a metallic element will replace the positive part of the compound and a non-metal will replace the negative part. ...
... These reactions usually involve an element reacting with an ionic compound. The element can only replace the part of the compound that forms the same type of ion that the element forms : a metallic element will replace the positive part of the compound and a non-metal will replace the negative part. ...
Fragmentation pathway for glutamine identification: Loss of 73 da
... the McLafferty rearrangement except that it does not involve a radical cation; thus in this paper, it is referred to as a McLafferty-type rearrangement. A similar rearrangement was also documented for metal adducts of peptide°esters°[24].°ii)°the°side°chain°carbonyl°carbon could be attacked by eithe ...
... the McLafferty rearrangement except that it does not involve a radical cation; thus in this paper, it is referred to as a McLafferty-type rearrangement. A similar rearrangement was also documented for metal adducts of peptide°esters°[24].°ii)°the°side°chain°carbonyl°carbon could be attacked by eithe ...
Balanced Chemical Equation
... • Predict the products, if any, note oxidation #’s, and balance the reaction: 1. __Fe(s) + __CuSO4(aq) __FeSO4(aq) + __Cu(s) 2. __Br2(l) + __MgCl2(aq) No Reaction 3. __Fe(s) + __Na3PO4(aq) No Reaction 4. __Al(s) + _3CuCl(aq) __AlCl3(aq) + 3_Cu(s) ...
... • Predict the products, if any, note oxidation #’s, and balance the reaction: 1. __Fe(s) + __CuSO4(aq) __FeSO4(aq) + __Cu(s) 2. __Br2(l) + __MgCl2(aq) No Reaction 3. __Fe(s) + __Na3PO4(aq) No Reaction 4. __Al(s) + _3CuCl(aq) __AlCl3(aq) + 3_Cu(s) ...
CHAPTER 4: AQUEOUS REACTIONS AND SOLUTION
... called an electrolyte because it will allow electric current to flow through it. Example: NaCl A substance that does not form ions in solution is called a nonelectrolyte. Example: C12H22O11 ...
... called an electrolyte because it will allow electric current to flow through it. Example: NaCl A substance that does not form ions in solution is called a nonelectrolyte. Example: C12H22O11 ...
3.1 - Weathering Define mechanical and chemical weathering
... http://www.northland.cc.mn.us/biology/Biology1111/animations/diss olve.html ...
... http://www.northland.cc.mn.us/biology/Biology1111/animations/diss olve.html ...
STOICHIOMETRY:
... calculations about the masses, volumes or concentrations of reactants and products involved in a chemical reaction. The reason we balance chemical reactions is not just to solve tricky chemistry problems. We understand that it is necessary to satisfy the Law of Conservation of Mass; a reaction that ...
... calculations about the masses, volumes or concentrations of reactants and products involved in a chemical reaction. The reason we balance chemical reactions is not just to solve tricky chemistry problems. We understand that it is necessary to satisfy the Law of Conservation of Mass; a reaction that ...
Kjeldahl Method for Determination of Nitrogen
... This significantly increases the rate of organic decomposition in the digestion mixture, shortening the length of time required for digestion. There are several precautions to keep in mind concerning salt addition. First, it is possible to raise the solution temperature of the digestion mixture too ...
... This significantly increases the rate of organic decomposition in the digestion mixture, shortening the length of time required for digestion. There are several precautions to keep in mind concerning salt addition. First, it is possible to raise the solution temperature of the digestion mixture too ...
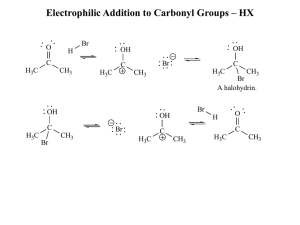
Electrophilic Addition to Carbonyl Groups – HX
... • If an aldol reaction is worked up under acidic conditions, an E2 reaction will follow, giving a double bond conjugated to the carbonyl group: ...
... • If an aldol reaction is worked up under acidic conditions, an E2 reaction will follow, giving a double bond conjugated to the carbonyl group: ...
AP Chem Summer Assignment KEY
... B. Sample problem: What ionic compound would form when calcium ions combine with bromide ions? Steps to the Criss Cross Method: 1. Write the ions with their charges, cations are always first. Ca2+ Br12. Cross over the charges by using the absolute value of each ion’s charge as the subscript for the ...
... B. Sample problem: What ionic compound would form when calcium ions combine with bromide ions? Steps to the Criss Cross Method: 1. Write the ions with their charges, cations are always first. Ca2+ Br12. Cross over the charges by using the absolute value of each ion’s charge as the subscript for the ...
Module 3 -- Lesson 4
... alcohol to produce an ester and water. An ester is a compound with a pleasant odor that can be synthesized in the laboratory by reacting an alcohol and an organic acid (chemistry 30S students do this as part of their course at the CLC). Esters account for the distinctive odors of many fruits such as ...
... alcohol to produce an ester and water. An ester is a compound with a pleasant odor that can be synthesized in the laboratory by reacting an alcohol and an organic acid (chemistry 30S students do this as part of their course at the CLC). Esters account for the distinctive odors of many fruits such as ...
Equations - Pearson Schools and FE Colleges
... A word equation has the names of the reactants on the left, an arrow in the middle, and the names of the products on the right. ...
... A word equation has the names of the reactants on the left, an arrow in the middle, and the names of the products on the right. ...
Types of Reactions Lab
... HCl: Corrosive! Harmful if ingested (burning of mouth, throat, etc.). Swallowing may be fatal. Skin & eye contact may cause severe burns depending on concentration. Wash skin/eyes for 15 minutes if contact occurs. Ethanol: Flammable! Low flash point and high evaporation rates require extreme caution ...
... HCl: Corrosive! Harmful if ingested (burning of mouth, throat, etc.). Swallowing may be fatal. Skin & eye contact may cause severe burns depending on concentration. Wash skin/eyes for 15 minutes if contact occurs. Ethanol: Flammable! Low flash point and high evaporation rates require extreme caution ...
Theoretical Competition - Austrian Chemistry Olympiad
... it will disintegrate immediately to give a Li-isotope and an α-particle. Both of them have kinetic energy from the nuclear reaction and a range of about 10 µm, which corresponds approximately to the diameter of a cell. They therefore attack only cancer cells. ...
... it will disintegrate immediately to give a Li-isotope and an α-particle. Both of them have kinetic energy from the nuclear reaction and a range of about 10 µm, which corresponds approximately to the diameter of a cell. They therefore attack only cancer cells. ...
Chapter 4
... 3) The oxidation state of oxygen in compounds is -2, except in peroxides, such as H2O2 where it is -1. 4) The oxidation state of hydrogen in compounds is +1, except in metal hydrides, like NaH, where it is -1. ...
... 3) The oxidation state of oxygen in compounds is -2, except in peroxides, such as H2O2 where it is -1. 4) The oxidation state of hydrogen in compounds is +1, except in metal hydrides, like NaH, where it is -1. ...
2nd Semester final review
... c. How many mL are needed to dissolve 100 g of CH3OH (molar mass 32 g = 1 mole) into a 3.6 M solution 868 mL 37. What is the difference between a solute, a solvent, and a solution? Solute is the substance being dissolved (often solid) Solvent is the substance doing dissolving (usually liquid) Soluti ...
... c. How many mL are needed to dissolve 100 g of CH3OH (molar mass 32 g = 1 mole) into a 3.6 M solution 868 mL 37. What is the difference between a solute, a solvent, and a solution? Solute is the substance being dissolved (often solid) Solvent is the substance doing dissolving (usually liquid) Soluti ...
The Hydroxylation of Aromatic Nitro Compounds by Alkalies
... should diminish the yield; but no such diminution occurs. The only remaining product is water; and this is now believed to render the potassium hydroxide incapable of further reaction by coating the surface. Wohl's statement that the hydroxylation proceeds in the absence of air Is true. but then the ...
... should diminish the yield; but no such diminution occurs. The only remaining product is water; and this is now believed to render the potassium hydroxide incapable of further reaction by coating the surface. Wohl's statement that the hydroxylation proceeds in the absence of air Is true. but then the ...
A Plausible Simultaneous Synthesis of Amino Acids and Simple
... experimental and analytical details of this work are provided in the Supporting Information). The formation of dipeptides in a mildly basic medium (pH 8–10) created by ammonia in the spark-discharge experiment contrasts with previous reports that indicate that acidic conditions are necessary to prom ...
... experimental and analytical details of this work are provided in the Supporting Information). The formation of dipeptides in a mildly basic medium (pH 8–10) created by ammonia in the spark-discharge experiment contrasts with previous reports that indicate that acidic conditions are necessary to prom ...
Print out Reviews # 1 through # 17
... 6. Round off each of the following measurements to 3 significant figures. (A) 98.473 L (B) 12.17 oC (C) 0.00076321 kg (D) 7.485 x 104 mm (E) 57.048 m (F) 1764.9 mL 7. Distinguish between accuracy and precision of a measurement. 8. A technician experimentally determined the boiling point of octane as ...
... 6. Round off each of the following measurements to 3 significant figures. (A) 98.473 L (B) 12.17 oC (C) 0.00076321 kg (D) 7.485 x 104 mm (E) 57.048 m (F) 1764.9 mL 7. Distinguish between accuracy and precision of a measurement. 8. A technician experimentally determined the boiling point of octane as ...
General, Organic, and Biological Chemistry
... 46) Isotopes are atoms of the same element that have A) different atomic numbers. B) the same atomic numbers but different numbers of protons. C) the same atomic numbers but different numbers of electrons. D) the same atomic number but different numbers of neutrons. E) the same atomic mass but diff ...
... 46) Isotopes are atoms of the same element that have A) different atomic numbers. B) the same atomic numbers but different numbers of protons. C) the same atomic numbers but different numbers of electrons. D) the same atomic number but different numbers of neutrons. E) the same atomic mass but diff ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.