
Pauling Scale of Electronegativities for the Various Elements
... All O2_, OH_, and S2_ compounds are insoluble except: Na+, K+, NH2+, Ca2+, Sr2+, and Ba2+. Metal sulfides are the least soluble followed by H2S; hydroxides are only slightly more soluble than sulfides. ...
... All O2_, OH_, and S2_ compounds are insoluble except: Na+, K+, NH2+, Ca2+, Sr2+, and Ba2+. Metal sulfides are the least soluble followed by H2S; hydroxides are only slightly more soluble than sulfides. ...
CHM203 - National Open University of Nigeria
... strength of a hydrogen bond ranges from 10 to 40 kJ mol-1. Hydrogen bonding has an important influence on physical properties such as melting point, boiling point and solubility of substances. This will be illustrated using examples in the following subsections. The dipole-dipole, induced dipole-ind ...
... strength of a hydrogen bond ranges from 10 to 40 kJ mol-1. Hydrogen bonding has an important influence on physical properties such as melting point, boiling point and solubility of substances. This will be illustrated using examples in the following subsections. The dipole-dipole, induced dipole-ind ...
CLASSES AND NOMENCLATURE OF INORGANIC COMPOUNDS
... Specify the factor which does not shift the 2 times: equilibrium to the right: A 8 times A increase the temperature of the system B 2 times B increase the concentration of H2S. C 4 times C decrease the concentration of SO2. D 16 times D increase of the pressure E 6 times E decrease of the temperatur ...
... Specify the factor which does not shift the 2 times: equilibrium to the right: A 8 times A increase the temperature of the system B 2 times B increase the concentration of H2S. C 4 times C decrease the concentration of SO2. D 16 times D increase of the pressure E 6 times E decrease of the temperatur ...
SAMPLE QUESTION PAPER SIR.S.M.TAHIR CHEMISTRY Mob: 9557076999
... (i) Account for the following observations. (a) Aldehydes are more reactive than ketone towards nucleophiles. (b) The aldehydes and ketones have lower boiling points than the corresponding acids. (c) The aldehydes and ketones undergo a number of addition reactions. (ii) Give chemical tests to distin ...
... (i) Account for the following observations. (a) Aldehydes are more reactive than ketone towards nucleophiles. (b) The aldehydes and ketones have lower boiling points than the corresponding acids. (c) The aldehydes and ketones undergo a number of addition reactions. (ii) Give chemical tests to distin ...
Chemical Reactions (Part One)
... The ripening of all fruit and vegetables involves similar chemical reactions. The speed of ripening is affected by the temperature and by the presence of a chemical called ethene, C2H4. Food scientists can tell producers and supermarkets the best conditions for slowing down or speeding up the ripeni ...
... The ripening of all fruit and vegetables involves similar chemical reactions. The speed of ripening is affected by the temperature and by the presence of a chemical called ethene, C2H4. Food scientists can tell producers and supermarkets the best conditions for slowing down or speeding up the ripeni ...
Basso08_preprint - University of Strathclyde
... product formation of Z-L-Asp-L-Phe-OMe. This helped to explain both final yields and the enzyme kinetics in this system.57 In such systems salts can be used also when thermodynamics are unfavourable and precipitation is not achieved. The addition of specific counter-ions that form poorly soluble sal ...
... product formation of Z-L-Asp-L-Phe-OMe. This helped to explain both final yields and the enzyme kinetics in this system.57 In such systems salts can be used also when thermodynamics are unfavourable and precipitation is not achieved. The addition of specific counter-ions that form poorly soluble sal ...
KEY_Reaction Types WS
... task is deciding what type of reaction is taking place. In this chapter we study three types: ...
... task is deciding what type of reaction is taking place. In this chapter we study three types: ...
Balancing and Predicting Chemical Reactions:
... • Complete the experiment – observations, reaction predictions, etc. at each station before you continue to another station • When finished, begin working on WSs ...
... • Complete the experiment – observations, reaction predictions, etc. at each station before you continue to another station • When finished, begin working on WSs ...
Document
... 15. The biological significance of chirality (stereoisomerism) is that A. because proteins (enzymes) are made of chiral subunits they usually react with only one stereoisomer of a molecule. B. because proteins (enzymes) are not made of chiral subunits they react with all stereoisomers of a molecule. ...
... 15. The biological significance of chirality (stereoisomerism) is that A. because proteins (enzymes) are made of chiral subunits they usually react with only one stereoisomer of a molecule. B. because proteins (enzymes) are not made of chiral subunits they react with all stereoisomers of a molecule. ...
Chapter 12
... Disulfide bond A covalent bond between two sulfur atoms of two different amino acids in a protein molecule. Salt bridge An attraction between a negatively charged side chain and a positively charged side chain in a protein molecule. Triglyceride A compound with three hydrocarbon groups attached to a ...
... Disulfide bond A covalent bond between two sulfur atoms of two different amino acids in a protein molecule. Salt bridge An attraction between a negatively charged side chain and a positively charged side chain in a protein molecule. Triglyceride A compound with three hydrocarbon groups attached to a ...
File
... __________ take place) - If this reaction takes place in a closed container, both reactants and products can be found - This is not considered to be a quantitative reaction - can be explained by considering the reverse reaction: CaCO3(s) CaO(s) + CO2(g) - The equilibrium equation is CaCO3(s) CaO ...
... __________ take place) - If this reaction takes place in a closed container, both reactants and products can be found - This is not considered to be a quantitative reaction - can be explained by considering the reverse reaction: CaCO3(s) CaO(s) + CO2(g) - The equilibrium equation is CaCO3(s) CaO ...
Chlorine
... usually converted back to mercury by reaction with water, producing hydrogen and sodium ( or potassium ) hydroxide at a commercially useful concentration ( 50 % by weight ) . The mercury is then recycled to the primary cell . The mercury process is the least energy - efficient of the three main tech ...
... usually converted back to mercury by reaction with water, producing hydrogen and sodium ( or potassium ) hydroxide at a commercially useful concentration ( 50 % by weight ) . The mercury is then recycled to the primary cell . The mercury process is the least energy - efficient of the three main tech ...
File
... What is percentage yield and why do we use it? In chemical reactions we rarely, if ever, get the amount/quantity of products we calculate from a (balanced) chemical equation. The reasons for this can be: • at the end of the reaction there may be reactant left unconverted to product (see excess) • ...
... What is percentage yield and why do we use it? In chemical reactions we rarely, if ever, get the amount/quantity of products we calculate from a (balanced) chemical equation. The reasons for this can be: • at the end of the reaction there may be reactant left unconverted to product (see excess) • ...
Chem 12 Prov Exam PLO Review
... particles convert all their PE into KE C. particles are in an excited state and are catalyzed D. particles have sufficient KE and proper molecular orientation ...
... particles convert all their PE into KE C. particles are in an excited state and are catalyzed D. particles have sufficient KE and proper molecular orientation ...
- Career Point Kota
... (a) The complex [Co(NH3)5(SCN)] exhibit the linkage isomerism. (b) In both [NiCl4]2– & [Ni(CN)4]2– the nickel is in +2 o.s. and having configuration 3d8 and it contain 2 unpaired eΘ but CN is a strong ligand compare to Cl so it repel the eΘ density of metal ion because of which eΘ get paired in case ...
... (a) The complex [Co(NH3)5(SCN)] exhibit the linkage isomerism. (b) In both [NiCl4]2– & [Ni(CN)4]2– the nickel is in +2 o.s. and having configuration 3d8 and it contain 2 unpaired eΘ but CN is a strong ligand compare to Cl so it repel the eΘ density of metal ion because of which eΘ get paired in case ...
Chapter 13 Organic Chemistry
... while the highly branched alkane C8H18 causes little knocking and is assigned an octane rating of 100. A gasoline with an octane rating of 87 causes the same knocking as a mixture that is 87% in the branched alkane and 13% of the straight chain alkane. Alkenes are organic compounds that contain carb ...
... while the highly branched alkane C8H18 causes little knocking and is assigned an octane rating of 100. A gasoline with an octane rating of 87 causes the same knocking as a mixture that is 87% in the branched alkane and 13% of the straight chain alkane. Alkenes are organic compounds that contain carb ...
+ H 2 O
... Often called a neutralization titration Because the acid neutralizes the base Often called volumetric analysis since titration is made to determine concentrations It involves delivery of a measured volume of solution of known concentration (titrant) Titrant is added to the unknown (analyte) ...
... Often called a neutralization titration Because the acid neutralizes the base Often called volumetric analysis since titration is made to determine concentrations It involves delivery of a measured volume of solution of known concentration (titrant) Titrant is added to the unknown (analyte) ...
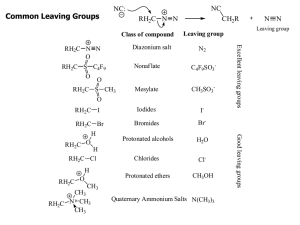
Common Leaving Groups
... •As a general rule, elimination reactions can always compete with substitution reactions. We can, however, alter the reaction conditions to favour one process over another. •To favour E1 over SN1 for alcohols, use an acid with a non-nucleophilic conjugate base (H2SO4, H3PO4). To favour SN1 over E1, ...
... •As a general rule, elimination reactions can always compete with substitution reactions. We can, however, alter the reaction conditions to favour one process over another. •To favour E1 over SN1 for alcohols, use an acid with a non-nucleophilic conjugate base (H2SO4, H3PO4). To favour SN1 over E1, ...
10 | Carbon: More Than Just Another Element
... more complex structures. This will become increasingly evident in this brief tour of organic chemistry. A carbon atom can reach an octet of electrons in various ways (Figure 10.1): • By forming four single bonds. A carbon atom can bond to four other atoms, which can be either atoms of other elements ...
... more complex structures. This will become increasingly evident in this brief tour of organic chemistry. A carbon atom can reach an octet of electrons in various ways (Figure 10.1): • By forming four single bonds. A carbon atom can bond to four other atoms, which can be either atoms of other elements ...
Document
... The empirical formula of a compound is CH2 Which molecular formula is correctly paired with a structural formula for this compound? ...
... The empirical formula of a compound is CH2 Which molecular formula is correctly paired with a structural formula for this compound? ...
[SESSION-2014-2015] SUBJECT - SCIENCE PATNA REGION
... 10. What is rancidity? Write the common methods to prevent it. 11. What is corrosion? State the conditions necessary for rusting of iron. How rusting is harmful? 12. Name the type of reactions in the following cases: a. Garbage producing foul smell b. Burning of natural gas. c. Carbon dioxide gas pa ...
... 10. What is rancidity? Write the common methods to prevent it. 11. What is corrosion? State the conditions necessary for rusting of iron. How rusting is harmful? 12. Name the type of reactions in the following cases: a. Garbage producing foul smell b. Burning of natural gas. c. Carbon dioxide gas pa ...
Unit - 7.pmd
... pπ -p π multiple bonds with itself and with other elements having small size and high electronegativity (e.g., C, O). Heavier elements of this group do not form pπ -pπ bonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diat ...
... pπ -p π multiple bonds with itself and with other elements having small size and high electronegativity (e.g., C, O). Heavier elements of this group do not form pπ -pπ bonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diat ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.




















![[SESSION-2014-2015] SUBJECT - SCIENCE PATNA REGION](http://s1.studyres.com/store/data/008930072_1-5a35e1ae8e3204ea88999f1418a93013-300x300.png)

