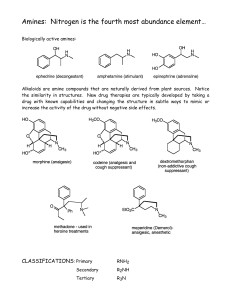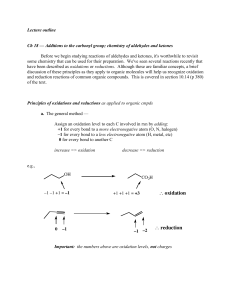
Wed March 3 lecture
... b. The easier method for alcohols and carbonyl cmpds — again focusing only on the C(s) involved in the rxn ... oxidation => gain of bonds to O and/or loss of bonds to H reduction => loss of bonds to O and/or gain of bonds to H How does this apply to the two examples above? Does this method lead to ...
... b. The easier method for alcohols and carbonyl cmpds — again focusing only on the C(s) involved in the rxn ... oxidation => gain of bonds to O and/or loss of bonds to H reduction => loss of bonds to O and/or gain of bonds to H How does this apply to the two examples above? Does this method lead to ...
Answer Key
... examination is to see whether students can follow directions, only the FIRST 2 answers were marked. a) Methoxymethane boils at a much lower temperature than ethanol has despite the fact that both compounds have the same relative molecular mass. The fact that the two molecules have the same RMM means ...
... examination is to see whether students can follow directions, only the FIRST 2 answers were marked. a) Methoxymethane boils at a much lower temperature than ethanol has despite the fact that both compounds have the same relative molecular mass. The fact that the two molecules have the same RMM means ...
Reactions of Carboxylic Acids
... Reduction with LiAlH4 (Section 15.3) Formation of acyl chlorides (Section 12.7) Esterification (Section 15.8) ...
... Reduction with LiAlH4 (Section 15.3) Formation of acyl chlorides (Section 12.7) Esterification (Section 15.8) ...
CHM134 General Chemistry I Semester Review – Dr. Steel This list
... 18. What is the likely precipitate formed by the reaction: CrCl3(aq) + AgNO3(aq) Æ 19. What salt is formed when potassium hydroxide and sulfuric acid react? 20. Calculate the oxidation number of chromium in K2Cr2O7. 21. If n = 3, what are the allowed quantum numbers for l? 22. What element has the g ...
... 18. What is the likely precipitate formed by the reaction: CrCl3(aq) + AgNO3(aq) Æ 19. What salt is formed when potassium hydroxide and sulfuric acid react? 20. Calculate the oxidation number of chromium in K2Cr2O7. 21. If n = 3, what are the allowed quantum numbers for l? 22. What element has the g ...
Naming organic compounds
... The functional group in the alcohols is the hydroxyl group (-OH). Alcohols end in the letters -ol ...
... The functional group in the alcohols is the hydroxyl group (-OH). Alcohols end in the letters -ol ...
SCH 4U
... The polymer formed from lactic acid (structure shown below) is used for surgical sutures. The polymer structure is best represented by which of the following? ...
... The polymer formed from lactic acid (structure shown below) is used for surgical sutures. The polymer structure is best represented by which of the following? ...
Chemistry -- Acids and Bases
... V. Predicting the Reactions that Occur between Acids and Bases A. How many protons can an acid donate H3PO4 can donate up to 3 H+ -- “more than one” – “proton” Polyprotic acid: An acid that can donate more than one proton Triprotic acid: An acid that can donate up to 3 ...
... V. Predicting the Reactions that Occur between Acids and Bases A. How many protons can an acid donate H3PO4 can donate up to 3 H+ -- “more than one” – “proton” Polyprotic acid: An acid that can donate more than one proton Triprotic acid: An acid that can donate up to 3 ...
PowerPoint Presentation - Chapter 1
... Which combination of reactants will give 1iodobutane as the major product? A) CH3CH2CH2CH3 + HI B) CH3CH2CH2CH2OH + KI C) CH3CH2CH2CH2Br + NaI in acetone D) CH3CH2CH=CH2 + I2 in water ...
... Which combination of reactants will give 1iodobutane as the major product? A) CH3CH2CH2CH3 + HI B) CH3CH2CH2CH2OH + KI C) CH3CH2CH2CH2Br + NaI in acetone D) CH3CH2CH=CH2 + I2 in water ...
Chapter 4
... breaking up aqueous compounds (ionic) 3. Cancel spectator ions (same on both sides including state) 4. Rewrite what’s left (reduce coefficients if necessary) See example in book pg. 151 ...
... breaking up aqueous compounds (ionic) 3. Cancel spectator ions (same on both sides including state) 4. Rewrite what’s left (reduce coefficients if necessary) See example in book pg. 151 ...
chemical equilibrium in a liquid phase
... Sulfuric Acid BY C. M. SUTERAND ELMEROBERG' It has been shown by other investigators that the reaction between sulfuric acid and a primary aliphatic alcohol at ordinary temperatures gives only the mono-alkyl ester regardless of whether the acid2 or the alcohol3 is present in excess. Quantitative stu ...
... Sulfuric Acid BY C. M. SUTERAND ELMEROBERG' It has been shown by other investigators that the reaction between sulfuric acid and a primary aliphatic alcohol at ordinary temperatures gives only the mono-alkyl ester regardless of whether the acid2 or the alcohol3 is present in excess. Quantitative stu ...
Nucleophilic
... Nucleophilicity usually increases going down a column of the periodic chart. Thus, sulfur nucleophiles are more reactive than oxygen nucleophiles. Halides: I– > Br– > Cl– > F–. Negatively charged nucleophiles are usually more reactive than neutral nucleophiles. Note that elimination is a competing ...
... Nucleophilicity usually increases going down a column of the periodic chart. Thus, sulfur nucleophiles are more reactive than oxygen nucleophiles. Halides: I– > Br– > Cl– > F–. Negatively charged nucleophiles are usually more reactive than neutral nucleophiles. Note that elimination is a competing ...
Kazzie`s Guide to Orgo 2
... Predict all possible products from the following reaction (Hints: 1- There is one for the cis formation and one for the trans formation (therefore stereochemistry is important) 2- One never works with a single molecule 3-No acid is available): NaBH4 O ...
... Predict all possible products from the following reaction (Hints: 1- There is one for the cis formation and one for the trans formation (therefore stereochemistry is important) 2- One never works with a single molecule 3-No acid is available): NaBH4 O ...
PPT file
... The mechanism for this reaction is very similar to that for addition of water to a carbonyl group; however, because amines are better nucleophiles than alcohols/water, the carbonyl group doesn’t have to be protonated first. A small amount of acid is necessary to generate a leaving group of water (R- ...
... The mechanism for this reaction is very similar to that for addition of water to a carbonyl group; however, because amines are better nucleophiles than alcohols/water, the carbonyl group doesn’t have to be protonated first. A small amount of acid is necessary to generate a leaving group of water (R- ...
- Palisades School District
... 2. The oxidation number of a monatomic ion equal the charge on the ion. 3. The more electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine in a compound is always –1 5. Oxygen has an oxidation numbe ...
... 2. The oxidation number of a monatomic ion equal the charge on the ion. 3. The more electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine in a compound is always –1 5. Oxygen has an oxidation numbe ...
Organic Chemistry
... CH3OH methyl alcohol, methanol, “wood alcohol”. Impurity in moonshine; causes blindness CH3CH2OH ethyl alcohol, ethanol, “alcohol” Prepared by fermentation of sugar-containing plant material. ...
... CH3OH methyl alcohol, methanol, “wood alcohol”. Impurity in moonshine; causes blindness CH3CH2OH ethyl alcohol, ethanol, “alcohol” Prepared by fermentation of sugar-containing plant material. ...
Organic Chemistry
... Acetic acid CH3COOH is a carboxylic acid - it has the -COOH group The compound CH3NH2 is methyl amine An amino acid like glycine (see picture) has both the carboxylate and the amino group ...
... Acetic acid CH3COOH is a carboxylic acid - it has the -COOH group The compound CH3NH2 is methyl amine An amino acid like glycine (see picture) has both the carboxylate and the amino group ...
1 Chapter 8: Nucleophilic Substitution 8.1: Functional Group
... Nucleophilicity usually increases going down a column of the periodic chart. Thus, sulfur nucleophiles are more reactive than oxygen nucleophiles. Halides: I– > Br– > Cl– > F–. Negatively charged nucleophiles are usually more reactive than neutral nucleophiles. Note that elimination is a competing ...
... Nucleophilicity usually increases going down a column of the periodic chart. Thus, sulfur nucleophiles are more reactive than oxygen nucleophiles. Halides: I– > Br– > Cl– > F–. Negatively charged nucleophiles are usually more reactive than neutral nucleophiles. Note that elimination is a competing ...
name - cloudfront.net
... 10. Vanadium(V) oxide reacts with calcium according to the chemical equation below. When 10.0 moles of ...
... 10. Vanadium(V) oxide reacts with calcium according to the chemical equation below. When 10.0 moles of ...
Weathering and Erosion - School District 67 Okanagan Skaha
... • Water weathers rock by dissolving it • Water combines with some minerals (eg. Feldspar) to produce clay • Chemical reaction of water with other substances is called hydrolysis ...
... • Water weathers rock by dissolving it • Water combines with some minerals (eg. Feldspar) to produce clay • Chemical reaction of water with other substances is called hydrolysis ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.