Set 1 - ExamResults.net
... II. Answer Any FIVE of the following. ( Each questions carries 2 mark) 5 × 2 = 10 11. a) Give one example for paramagnetic substance. b) Which type of binding force existing in ice? 12. Write anodic and cathodic half-cell reactions taking place in Daniel cell. 13. Show that for first order reaction ...
... II. Answer Any FIVE of the following. ( Each questions carries 2 mark) 5 × 2 = 10 11. a) Give one example for paramagnetic substance. b) Which type of binding force existing in ice? 12. Write anodic and cathodic half-cell reactions taking place in Daniel cell. 13. Show that for first order reaction ...
summerpp_4
... The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. ...
... The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. ...
Chapter 4
... The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. ...
... The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. ...
Exploring Equilibria Name: ANSWER KEY Chem 112 This
... An acid is a compound that ionizes to form H+ ions in water. A base is a compound that ionizes or dissociates to form OH- ions in water. In an acid-base reaction, the acid donates an H+ ion and the base accepts the H+ ion. Strong bases donate OH- ions to water, which accept H+ ions from the acid to ...
... An acid is a compound that ionizes to form H+ ions in water. A base is a compound that ionizes or dissociates to form OH- ions in water. In an acid-base reaction, the acid donates an H+ ion and the base accepts the H+ ion. Strong bases donate OH- ions to water, which accept H+ ions from the acid to ...
Document
... and HI. Chloroalkanes cannot be prepared by this method because Cl- is too poor a nucleophile. ...
... and HI. Chloroalkanes cannot be prepared by this method because Cl- is too poor a nucleophile. ...
Alcohols
... alkanes because alcohols can form hydrogen bonds. • The stronger interaction between alcohol molecules will require more energy to break them resulting in a higher ...
... alkanes because alcohols can form hydrogen bonds. • The stronger interaction between alcohol molecules will require more energy to break them resulting in a higher ...
Ch 6 Lecture 2
... a) Polar covalent bonds must lead to overall dipole = polar Water, alcohols, acetonitrile, acetone b) Nonpolar = small or no net dipole: alkanes, ethers ...
... a) Polar covalent bonds must lead to overall dipole = polar Water, alcohols, acetonitrile, acetone b) Nonpolar = small or no net dipole: alkanes, ethers ...
Acids and Bases
... A protic solvent is one in which hydrogen is attached to a highly electronegative atom such as oxygen or nitrogen e.g. water Solvation of both acetic acid and acetate ion occurs in water although the acetate is more stabilized by this solvation ...
... A protic solvent is one in which hydrogen is attached to a highly electronegative atom such as oxygen or nitrogen e.g. water Solvation of both acetic acid and acetate ion occurs in water although the acetate is more stabilized by this solvation ...
NAME
... i) lithium hydroxide pellets are added to a solution of sulfuric acid, lithium sulfate and water are formed. ...
... i) lithium hydroxide pellets are added to a solution of sulfuric acid, lithium sulfate and water are formed. ...
Chapter 4 - Reactions in Aqueous Solutions
... base, or both. (a) HI, (b) CH3COO-, (c) H2PO4HI (aq) ...
... base, or both. (a) HI, (b) CH3COO-, (c) H2PO4HI (aq) ...
1 1. Give two reasons why a luminous flame is not used for heating
... b) Calculate the molar enthalpy of formation of butane C4H10 from its elements in their normal states at standard temperature and pressure. 2mks*UG* When calcium carbonate was added to a solution of dry hydrogen chloride in methyl benzene there was no observable reaction explain. 2mks*UG* The chief ...
... b) Calculate the molar enthalpy of formation of butane C4H10 from its elements in their normal states at standard temperature and pressure. 2mks*UG* When calcium carbonate was added to a solution of dry hydrogen chloride in methyl benzene there was no observable reaction explain. 2mks*UG* The chief ...
EX. Draw the structure of
... Naming and Drawing Alkyl Halides: Identify the root Identify the prefix: Name and number any alkyl side groups. Insert the number(s) of the carbon atom(s) bonded to the halogen(s). Use the prefix(es) that identify the specific halogen(s) ...
... Naming and Drawing Alkyl Halides: Identify the root Identify the prefix: Name and number any alkyl side groups. Insert the number(s) of the carbon atom(s) bonded to the halogen(s). Use the prefix(es) that identify the specific halogen(s) ...
X CHEMISTRY-1-2006 TIME-1½ MARKS 80 SECTION
... Calculate the volume of methane (ctlu) measured at S.T.P released from 14.48 of aluminum carbide by excess of water (Al-27, C=12, H=1). E. a. What a kind of particles will be found in a liquid compound which is a non- electrolyte? c. If HX is a weak acid, what particles will be preseut in its dilute ...
... Calculate the volume of methane (ctlu) measured at S.T.P released from 14.48 of aluminum carbide by excess of water (Al-27, C=12, H=1). E. a. What a kind of particles will be found in a liquid compound which is a non- electrolyte? c. If HX is a weak acid, what particles will be preseut in its dilute ...
Name Page 1 F.05.215e3p1 I.
... related oxidizing agents, a 1,2-diol is formed. When phenylacetylene (Compound E) is treated with oxidizing agents such as OsO4, the corresponding addition reaction product is not observed; instead it is proposed that the initial addition reaction product, which is an ene-diol, undergoes an isomeriz ...
... related oxidizing agents, a 1,2-diol is formed. When phenylacetylene (Compound E) is treated with oxidizing agents such as OsO4, the corresponding addition reaction product is not observed; instead it is proposed that the initial addition reaction product, which is an ene-diol, undergoes an isomeriz ...
Organometallic Compounds
... Try to see what factors promote the formation of the negative charge on the carbon atoms: hybridization, resonance. ...
... Try to see what factors promote the formation of the negative charge on the carbon atoms: hybridization, resonance. ...
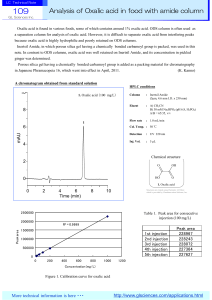
Analysis of Oxalic acid in food with amide column
... because oxalic acid is highly hydrophilic and poorly retained on ODS columns. Inertsil Amide, in which porous silica gel having a chemically bonded carbamoyl group is packed, was used in this note. In contrast to ODS columns, oxalic acid was well retained on Inertsil Amide, and its concentration in ...
... because oxalic acid is highly hydrophilic and poorly retained on ODS columns. Inertsil Amide, in which porous silica gel having a chemically bonded carbamoyl group is packed, was used in this note. In contrast to ODS columns, oxalic acid was well retained on Inertsil Amide, and its concentration in ...
1. Natures Chemistry Unit Questions
... In the reaction, the carbon atom next to the carbonyl functional group of one molecule forms a bond with the carbonyl carbon atom of the second molecule. (a) Draw a structural formula for the product formed when propanone is used instead of ethanal in this type of reaction. (1) (b) Name an aldehyde ...
... In the reaction, the carbon atom next to the carbonyl functional group of one molecule forms a bond with the carbonyl carbon atom of the second molecule. (a) Draw a structural formula for the product formed when propanone is used instead of ethanal in this type of reaction. (1) (b) Name an aldehyde ...
CET MODEL QUESTION PAPER 1. Set of quantum numbers (n, /, m
... 1] An amino acid becomes acidic 3] Amino acid will have zwitter ion form ...
... 1] An amino acid becomes acidic 3] Amino acid will have zwitter ion form ...
Notes 07 Organometallic Compounds
... and can be reacted with _____________________ to give alkanes. ...
... and can be reacted with _____________________ to give alkanes. ...
CI 12.4 - Sackville School
... CI 12.2 Ethene is unsaturated; it has a double C=C bond. It undergoes electrophilic addition reactions in which another molecule is added on. A saturated compound is produced. a. Ethene with bromine (g) or (l) or dissolved in an organic solvent. Ethene will decolourise orange bromine; it reacts rapi ...
... CI 12.2 Ethene is unsaturated; it has a double C=C bond. It undergoes electrophilic addition reactions in which another molecule is added on. A saturated compound is produced. a. Ethene with bromine (g) or (l) or dissolved in an organic solvent. Ethene will decolourise orange bromine; it reacts rapi ...
2002
... (a) How do you account for the fact that aryl and alkenyl halides are less reactive towards nucleophilic substitution reactions? ...
... (a) How do you account for the fact that aryl and alkenyl halides are less reactive towards nucleophilic substitution reactions? ...
2018 Specimen Paper 2 - Cambridge International Examinations
... 22 Acids are compounds which donate protons (hydrogen ions). NH3(aq) + H2O(l) → NH4+(aq) + OH–(aq) Which compound in this equation is behaving as an acid? A ...
... 22 Acids are compounds which donate protons (hydrogen ions). NH3(aq) + H2O(l) → NH4+(aq) + OH–(aq) Which compound in this equation is behaving as an acid? A ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.