Background Information
... The orange-yellow Jones reagent will immediately turn green in the presence of 1 ° and 2 ° alcohols and aldehydes. The color change is typically observed only for these functional groups and this color change constitutes a “positive” result. Tertiary alcohols do not react with Jones reagent. Sometim ...
... The orange-yellow Jones reagent will immediately turn green in the presence of 1 ° and 2 ° alcohols and aldehydes. The color change is typically observed only for these functional groups and this color change constitutes a “positive” result. Tertiary alcohols do not react with Jones reagent. Sometim ...
The Thomas Hardye School Summer Preparation Task Chemistry AS
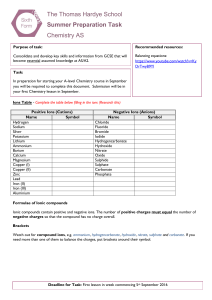
... Name Symbol Chloride Fluoride Bromide Iodide Hydrogencarbonate Hydroxide Nitrate Oxide Sulphide Sulphate Carbonate Phosphate ...
... Name Symbol Chloride Fluoride Bromide Iodide Hydrogencarbonate Hydroxide Nitrate Oxide Sulphide Sulphate Carbonate Phosphate ...
L-13
... the reaction and led to the production of allylated product 3a in 80% yield (entry 2).[6] Strong Lewis acids such as AlCl3 or BF3・OEt2 were not effective for the allylation (entries 3 and 4), probably because these catalysts are not stable under protic conditions. Sc(OTf)3 only gave a low yield of 3 ...
... the reaction and led to the production of allylated product 3a in 80% yield (entry 2).[6] Strong Lewis acids such as AlCl3 or BF3・OEt2 were not effective for the allylation (entries 3 and 4), probably because these catalysts are not stable under protic conditions. Sc(OTf)3 only gave a low yield of 3 ...
Functional Groups
... amines: derivatives of ammonia (NH3) in which 1 or more H atoms are replaced by organic groups (alkyl or aryl groups) ...
... amines: derivatives of ammonia (NH3) in which 1 or more H atoms are replaced by organic groups (alkyl or aryl groups) ...
File - Loreto Science
... • It is important to note that in the preaparation of aldehydes the acidified sodium dichromate is added slowly and the aldehyde is distilled off immediately to prevent further oxidation to carboxylic ...
... • It is important to note that in the preaparation of aldehydes the acidified sodium dichromate is added slowly and the aldehyde is distilled off immediately to prevent further oxidation to carboxylic ...
Presentation11_108
... Whereas electron donating substituents decrease the acidity . HCOOH > CH3COOH > CH3CH2COOH > CH3CH2CH2CH2COOH ( size of R group) Cl3CCOOH > Cl2CHCOOH > ClCH2COOH > CH3COOH > (OCH3)CH2COOH ( number of e.w.g. and e.d. g.) CH3CH2CH2CH(Cl)COOH > CH3CH2CH(Cl)CH2COOH > CH3CH(Cl)CH2CH2COOH > CH2(Cl)CH2CH2C ...
... Whereas electron donating substituents decrease the acidity . HCOOH > CH3COOH > CH3CH2COOH > CH3CH2CH2CH2COOH ( size of R group) Cl3CCOOH > Cl2CHCOOH > ClCH2COOH > CH3COOH > (OCH3)CH2COOH ( number of e.w.g. and e.d. g.) CH3CH2CH2CH(Cl)COOH > CH3CH2CH(Cl)CH2COOH > CH3CH(Cl)CH2CH2COOH > CH2(Cl)CH2CH2C ...
06_reactions
... Oxidation of an aldehyde under acidic conditions aldehyde + oxidising agent + H+ → carboxylic acid (there will be more reactants or products depending on the oxidising agent) Example: ethanal + acidified dichromate → ethanoic acid + chromium ions CH3CHO + Cr2O72- + 12H+ → CH3COOH + 2Cr3+ + 6H2O ...
... Oxidation of an aldehyde under acidic conditions aldehyde + oxidising agent + H+ → carboxylic acid (there will be more reactants or products depending on the oxidising agent) Example: ethanal + acidified dichromate → ethanoic acid + chromium ions CH3CHO + Cr2O72- + 12H+ → CH3COOH + 2Cr3+ + 6H2O ...
纳米结构体系物理化学性质的理论研究方法与实例
... 15.6 Amides • Carboxylic acids can react with ammonia to form nitrogen-containing organic compounds called amides. This is a two-step process, as shown below. O ...
... 15.6 Amides • Carboxylic acids can react with ammonia to form nitrogen-containing organic compounds called amides. This is a two-step process, as shown below. O ...
Spring 2015 CH 421 Name ________________________________________ Section ___________ Post‐lab 3: The Grignard Reaction: Preparation of an Alcohol
... 4) Aldehydes undergo reaction with a Grignard reagent to provide an alcohol product. Many aldehydes are prone to air oxidation. For instance, a bottle of benzaldehyde will turn from a clear liquid to a white solid if left open over time. What is the oxidation produ ...
... 4) Aldehydes undergo reaction with a Grignard reagent to provide an alcohol product. Many aldehydes are prone to air oxidation. For instance, a bottle of benzaldehyde will turn from a clear liquid to a white solid if left open over time. What is the oxidation produ ...
CARBON COMPOUNDS - SMK Raja Perempuan Ipoh
... Alkanes are saturated hydrocarbons that contain single covalent bonds. This single covalent bonds are strong and not reactive. Alkanes are neutral compound, they have no effect on blue or red litmus paper. Alkanes burn readily in air or in excess oxygen to produce carbon dioxide and water. The alkan ...
... Alkanes are saturated hydrocarbons that contain single covalent bonds. This single covalent bonds are strong and not reactive. Alkanes are neutral compound, they have no effect on blue or red litmus paper. Alkanes burn readily in air or in excess oxygen to produce carbon dioxide and water. The alkan ...
H + - uaschemistry
... • If it is on the side chain, it undergoes nucleophilic substitution just like a halogenoalkane. What is needed as a reagent and what would the ...
... • If it is on the side chain, it undergoes nucleophilic substitution just like a halogenoalkane. What is needed as a reagent and what would the ...
Chemical Equation Interpretations – Match the chemical equation
... D. When octane and oxygen gas are burned in our cards, carbon dioxide and water come out in the exhaust. E. Methanol , if ingested, reacts with oxygen to form formaldehyde, which is toxic. Water is also formed in this reaction. F. Liquid mercury evaporates to produce mercury vapor. G. Saturated fatt ...
... D. When octane and oxygen gas are burned in our cards, carbon dioxide and water come out in the exhaust. E. Methanol , if ingested, reacts with oxygen to form formaldehyde, which is toxic. Water is also formed in this reaction. F. Liquid mercury evaporates to produce mercury vapor. G. Saturated fatt ...
Chapter 18 Carboxylic Acids
... The problem of formation of precipitates in hard water was overcome by using a molecule containing a sulfonate (-SO3) group in the place of a carboxylate (-CO2-) group. • Calcium, magnesium and iron salts of sulfonic acids, RSO3H, are more soluble in water than are their salts of ...
... The problem of formation of precipitates in hard water was overcome by using a molecule containing a sulfonate (-SO3) group in the place of a carboxylate (-CO2-) group. • Calcium, magnesium and iron salts of sulfonic acids, RSO3H, are more soluble in water than are their salts of ...
Document
... • Oxidation/Reduction Reactions: Review (Section 12.2) • Reduction of Carbonyls to Alcohols (Section 12.3) • Oxidation of Alcohols (Section 12.4) • Organometallic Compounds (Section 12.5) • Organolithium and Magnesium Compounds (Section 12.6) • Reactions of Organolithium/Magnesium Species (Section 1 ...
... • Oxidation/Reduction Reactions: Review (Section 12.2) • Reduction of Carbonyls to Alcohols (Section 12.3) • Oxidation of Alcohols (Section 12.4) • Organometallic Compounds (Section 12.5) • Organolithium and Magnesium Compounds (Section 12.6) • Reactions of Organolithium/Magnesium Species (Section 1 ...
Biopolymers
... Two distinct ends N-terminus is an amine C-terminus is a carboxylic acid Principles of Chemistry II ...
... Two distinct ends N-terminus is an amine C-terminus is a carboxylic acid Principles of Chemistry II ...
Chapter 15
... • Activators are electron donors, so their stabilizing effects would be greater with the carbocation directly bonded to them. • Thus, all activators are ortho/para directors • By contrast, deactivator are electron withdrawing groups, so they want to avoid being directly bonded to the carbocation • T ...
... • Activators are electron donors, so their stabilizing effects would be greater with the carbocation directly bonded to them. • Thus, all activators are ortho/para directors • By contrast, deactivator are electron withdrawing groups, so they want to avoid being directly bonded to the carbocation • T ...
The Carboxylic Acid Group as an Effective Director of Ortho
... acid (2a).To a 500-mL round-bottomed flask equipped with a condenser and a rubber septum under argon was added 12.8 g (0.110mol) of redistilled (bp 120.5-121.0 "C) NJV,N',"-tetramethyl-l,2-ethylenediamine and 100 mL of dry THF. The mixture was stirred magnetically and cooled to -90 "C. After consecu ...
... acid (2a).To a 500-mL round-bottomed flask equipped with a condenser and a rubber septum under argon was added 12.8 g (0.110mol) of redistilled (bp 120.5-121.0 "C) NJV,N',"-tetramethyl-l,2-ethylenediamine and 100 mL of dry THF. The mixture was stirred magnetically and cooled to -90 "C. After consecu ...
Carbohydrates important reactions
... I. Chemical reactions of - COOH group Oxidation As noted above, sugars may be classified as reducing or non-reducing based on their reactivity with Tollens', Benedict's or Fehling's reagents. If a sugar is oxidized by these reagents it is called reducing, since the oxidant (Ag(+) or Cu(+2)) is reduc ...
... I. Chemical reactions of - COOH group Oxidation As noted above, sugars may be classified as reducing or non-reducing based on their reactivity with Tollens', Benedict's or Fehling's reagents. If a sugar is oxidized by these reagents it is called reducing, since the oxidant (Ag(+) or Cu(+2)) is reduc ...
Chapter 16. Biological Reagents
... any organic reactions that use the carbon dioxide lost from acetoacetic acid (or malonic acid) use in a coupled synthetic step? 16.5 Show the analogous mechanism between decarboxylation of a β-ketoacid and the thiamin decarboxylation of pyruvate. 16.6 Another biological reagent is vitamin B6 which d ...
... any organic reactions that use the carbon dioxide lost from acetoacetic acid (or malonic acid) use in a coupled synthetic step? 16.5 Show the analogous mechanism between decarboxylation of a β-ketoacid and the thiamin decarboxylation of pyruvate. 16.6 Another biological reagent is vitamin B6 which d ...
part 1
... Aldehydes form hydrates in water (an equilibrium) l An aldehyde hydrate is a diol and can react by the same ...
... Aldehydes form hydrates in water (an equilibrium) l An aldehyde hydrate is a diol and can react by the same ...
organic synthesis
... the nucleophile can attack from above and below there is an equal chance of each possibility a mixture of optically active isomers is produced only occurs if different groups are attached to the carbonyl group ...
... the nucleophile can attack from above and below there is an equal chance of each possibility a mixture of optically active isomers is produced only occurs if different groups are attached to the carbonyl group ...
0_specific - ugc-inno
... happen unhindered even in presence of water even if water does not seem to be significantly faciliting what happens without its presence. Geometrical Variations At this juncture the question of greater concern seems to be to find ways for proton transfer from the carboxylic acid function (of the non ...
... happen unhindered even in presence of water even if water does not seem to be significantly faciliting what happens without its presence. Geometrical Variations At this juncture the question of greater concern seems to be to find ways for proton transfer from the carboxylic acid function (of the non ...
Reaction of amino acids with exo-3,6-epoxy-1,2,3,6
... When an aqueous solution of glycine and the anhydride 2 was heated and subsequently evaporated by azeotropic distillation of toluene as described by Ondruš et al., we obtained material which corresponded with that reported by them.6 However, upon attempted crystallization from chloroform, as previou ...
... When an aqueous solution of glycine and the anhydride 2 was heated and subsequently evaporated by azeotropic distillation of toluene as described by Ondruš et al., we obtained material which corresponded with that reported by them.6 However, upon attempted crystallization from chloroform, as previou ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.