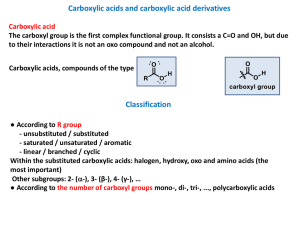
carboxylic acid
... 7.1. primary amides - In case of trivial name: „latin base” + „amide” suffix - In case of systematic name: carboxylic acid + amid suffix - In case of cyclic systems: instead of „carboxylic acid” suffix „carboxamide” 7.2. Secondary and tertiary amides – derivatives of N-(di)substituted of primary ami ...
... 7.1. primary amides - In case of trivial name: „latin base” + „amide” suffix - In case of systematic name: carboxylic acid + amid suffix - In case of cyclic systems: instead of „carboxylic acid” suffix „carboxamide” 7.2. Secondary and tertiary amides – derivatives of N-(di)substituted of primary ami ...
reactions of alcohols
... ammonia and silver nitrate. The active substance is the complex ion of [Ag(NH3)2]+ . • Conditions: heat gently • Reaction: aldehydes only are oxidised by Tollen’s reagent into a carboxylic acid and the silver(I) ions are reduced to silver atoms coating the inside of the test tube . The silver coatin ...
... ammonia and silver nitrate. The active substance is the complex ion of [Ag(NH3)2]+ . • Conditions: heat gently • Reaction: aldehydes only are oxidised by Tollen’s reagent into a carboxylic acid and the silver(I) ions are reduced to silver atoms coating the inside of the test tube . The silver coatin ...
Acids
... Proteins → AAs →phosphoric acid & sulfuric acid 4) Acidic Foods (lemons, limes, oranges, grapefruit, vinegar, tomatoes, soft drinks) 5) Stomach produces HCl acid So, our pH is usually tending to _____ …a condition called ____________ ...
... Proteins → AAs →phosphoric acid & sulfuric acid 4) Acidic Foods (lemons, limes, oranges, grapefruit, vinegar, tomatoes, soft drinks) 5) Stomach produces HCl acid So, our pH is usually tending to _____ …a condition called ____________ ...
Chapter16McMurryPPP
... The addition of bromine occurs in two steps In the first step the electrons act as a nucleophile toward Br2 (in a complex with FeBr3) This forms a cationic addition intermediate from benzene and a bromine cation The intermediate is not aromatic and therefore ...
... The addition of bromine occurs in two steps In the first step the electrons act as a nucleophile toward Br2 (in a complex with FeBr3) This forms a cationic addition intermediate from benzene and a bromine cation The intermediate is not aromatic and therefore ...
Chemistry - WordPress.com
... 65. In the assay of ephedrine HCl, we dissolve in water, add a reagent X, filter then extract with methylene chloride, what is the reagent X: a. 0.1 N NaOH (or Na2CO3) 66. Perchloric acid is stronger than acetic acid because: a. Acetic acid is more basic ??? 67. Codeine phosphate is dissolved in ace ...
... 65. In the assay of ephedrine HCl, we dissolve in water, add a reagent X, filter then extract with methylene chloride, what is the reagent X: a. 0.1 N NaOH (or Na2CO3) 66. Perchloric acid is stronger than acetic acid because: a. Acetic acid is more basic ??? 67. Codeine phosphate is dissolved in ace ...
Lecture 8a - UCLA Chemistry and Biochemistry
... aqueous layers, more difficult to dry than diethyl ether because it is more hygroscopic • A comparison of diethyl ether (m=1.15 D) and THF (m=1.75 D) shows that THF is a stronger Lewis Base because of its higher dipole moment compared to diethyl ether (d(Mg-O): 209 pm (THF), 213 pm (Et2O) (HF, 6-31G ...
... aqueous layers, more difficult to dry than diethyl ether because it is more hygroscopic • A comparison of diethyl ether (m=1.15 D) and THF (m=1.75 D) shows that THF is a stronger Lewis Base because of its higher dipole moment compared to diethyl ether (d(Mg-O): 209 pm (THF), 213 pm (Et2O) (HF, 6-31G ...
Bricks and Shapes - Graphite Cova GmbH
... CECOLIT bricks are characterised by their high cold and hot compressive strength and hardness. CECOLIT brickwork is thus exceptionally resistant to the mechanical demands made e.g. in numerous chemical processes due to the presence of erosive solid materials. An important characteristic of CECOLIT i ...
... CECOLIT bricks are characterised by their high cold and hot compressive strength and hardness. CECOLIT brickwork is thus exceptionally resistant to the mechanical demands made e.g. in numerous chemical processes due to the presence of erosive solid materials. An important characteristic of CECOLIT i ...
11.Unit 10 Haloalkanes and Haloarenes.
... Q3. When an alkyl halide is treated with ethanolic solution of KCN, the major product is alkylcyanide where as if alkyl halide is treated with AgCN, the major product is alkyl isocyanide. Ans. KCN is ionic they can attach through C or N but C-C bond is stronger than C-N bond. So RCN is major produc ...
... Q3. When an alkyl halide is treated with ethanolic solution of KCN, the major product is alkylcyanide where as if alkyl halide is treated with AgCN, the major product is alkyl isocyanide. Ans. KCN is ionic they can attach through C or N but C-C bond is stronger than C-N bond. So RCN is major produc ...
Section II - School District 27J
... Identify the oxidizing agent. c. If a 0.300 mol sample of copper reacts with 10.0 mL of 12.0 M nitric acid, how many moles of nitrogen monoxide gas will form? d. If 0.672 g of nitrogen monoxide is recovered from this reaction, calculate your percent error. ...
... Identify the oxidizing agent. c. If a 0.300 mol sample of copper reacts with 10.0 mL of 12.0 M nitric acid, how many moles of nitrogen monoxide gas will form? d. If 0.672 g of nitrogen monoxide is recovered from this reaction, calculate your percent error. ...
Organic Chemistry
... They are mainly used as fuels. The large Mr alkanes do not ignite easily so there is little demand for them as fuels so they are CRACKED to make smaller more useful alkanes and alkenes. Apart from combustion alkanes undergo few chemical reactions. This is for two main reasons: ...
... They are mainly used as fuels. The large Mr alkanes do not ignite easily so there is little demand for them as fuels so they are CRACKED to make smaller more useful alkanes and alkenes. Apart from combustion alkanes undergo few chemical reactions. This is for two main reasons: ...
Organic molecules with functional groups containing oxygen
... fertilizer (natural gas (CH4) is one of the raw materials used in its manufacture), energy used (machinery/transport/ ...
... fertilizer (natural gas (CH4) is one of the raw materials used in its manufacture), energy used (machinery/transport/ ...
ANSWERS: Types of Reactions - Chemical Minds
... 6) Addition – occurs in alkenes because they have double bonds. Ethene is an alkene so will undergo addition reactions. The chlorine (molecule) will add (across the double bond.) CH2 = CH2 + Cl2 → CH2Cl – CH2Cl Substitution occurs in alkanes (because they have single bonds). Ethane is an alkane, so ...
... 6) Addition – occurs in alkenes because they have double bonds. Ethene is an alkene so will undergo addition reactions. The chlorine (molecule) will add (across the double bond.) CH2 = CH2 + Cl2 → CH2Cl – CH2Cl Substitution occurs in alkanes (because they have single bonds). Ethane is an alkane, so ...
QUESTIONS FOR PRACTICE HYDROCARBONS 1. Name the least
... 10. Explain the reaction of alkylhalides with alcoholic ammonia in a sealed tube. 11. How do you convert ethyl bromide to ethylene? 12. Give any four differences between SN1 and SN2 mechanism. ...
... 10. Explain the reaction of alkylhalides with alcoholic ammonia in a sealed tube. 11. How do you convert ethyl bromide to ethylene? 12. Give any four differences between SN1 and SN2 mechanism. ...
Chem 1411 Chapt2
... ‘ide’. Also, one uses prefixes (mono, di, tri, tetra, penta, hexa, hepta, octa, nona, deca) to indicate the number of atoms of each element. If there is only one atom of the more electropositive element, do not use the prefix ‘mono’. Example: CO carbon monoxide; CO2 carbon dioxide NO2 nitrogen dioxi ...
... ‘ide’. Also, one uses prefixes (mono, di, tri, tetra, penta, hexa, hepta, octa, nona, deca) to indicate the number of atoms of each element. If there is only one atom of the more electropositive element, do not use the prefix ‘mono’. Example: CO carbon monoxide; CO2 carbon dioxide NO2 nitrogen dioxi ...
Document
... Only one product General form: element or compound + element or compound compound ...
... Only one product General form: element or compound + element or compound compound ...
Gen Chem Final--review problems Fall 2006
... For the precipitation reaction/s above in problem 1, please add the appropriate ‘state’ (i.e. solid or aq) to each species. For the oxidation/reduction reaction/s above, please identify the species that is being oxidized and the species being reduced and assign oxidation numbers to each atom. For th ...
... For the precipitation reaction/s above in problem 1, please add the appropriate ‘state’ (i.e. solid or aq) to each species. For the oxidation/reduction reaction/s above, please identify the species that is being oxidized and the species being reduced and assign oxidation numbers to each atom. For th ...
Alkynes - IIT Portal.com
... acetylene in liquid ammonia as the solvent. Terminal alkynes react similarly to give species of the type RC=CNa. Anions of acetylene and terminal alkynes are nucleophilic and react with methyl and primary alkyl halides to form carbon-carbon bonds by nucleophilic substitution. Some useful applicatio ...
... acetylene in liquid ammonia as the solvent. Terminal alkynes react similarly to give species of the type RC=CNa. Anions of acetylene and terminal alkynes are nucleophilic and react with methyl and primary alkyl halides to form carbon-carbon bonds by nucleophilic substitution. Some useful applicatio ...
Document
... • NaH is an especially good base for forming alkoxide because the by-product of the reaction, H2, is a gas that just bubbles out of the reaction mixture. ...
... • NaH is an especially good base for forming alkoxide because the by-product of the reaction, H2, is a gas that just bubbles out of the reaction mixture. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.























