acidic site
... A nucleophile provides an electron pair to make a new bond to any non-hydrogen atom. As such, nucleophilic sites are either: atoms that have one or more lone pair of electrons, or bonds Nucleophiles often have negative charges since anions are, by definition, electron-rich. Any carbon atom wit ...
... A nucleophile provides an electron pair to make a new bond to any non-hydrogen atom. As such, nucleophilic sites are either: atoms that have one or more lone pair of electrons, or bonds Nucleophiles often have negative charges since anions are, by definition, electron-rich. Any carbon atom wit ...
Amines and amides
... The presence of the OH-(aq) ion means that aqueous solutions of amines are ALKALINE. Like NH3 amines react with acids, H3O+ ion is more effective proton donor than H2O, therefore reaction goes to completion therefore amines ...
... The presence of the OH-(aq) ion means that aqueous solutions of amines are ALKALINE. Like NH3 amines react with acids, H3O+ ion is more effective proton donor than H2O, therefore reaction goes to completion therefore amines ...
CHM230 OXIDATION OF CYCLOHEXANOL TO CYCLOHEXANONE
... graduated cylinder. The distillate should be a mixture of cyclohexanone and water that contains excess acetic acid. Transfer the distillate to a separatory funnel or beaker. 6. Add 3.5 grams of sodium carbonate to neutralize any excess acetic acid, and then add a small amount, about 3 grams of sodiu ...
... graduated cylinder. The distillate should be a mixture of cyclohexanone and water that contains excess acetic acid. Transfer the distillate to a separatory funnel or beaker. 6. Add 3.5 grams of sodium carbonate to neutralize any excess acetic acid, and then add a small amount, about 3 grams of sodiu ...
CHE-05 Organic Chemistry
... We hope, you are familiar with the system of evaluation to be followed for the Bachelor's Degree Programme. As this stage you may probably like to re-read the section on assignments in the Programme Guide that we sent you after your enrolment. A weightage of 30 percent, as you are aware, has been ea ...
... We hope, you are familiar with the system of evaluation to be followed for the Bachelor's Degree Programme. As this stage you may probably like to re-read the section on assignments in the Programme Guide that we sent you after your enrolment. A weightage of 30 percent, as you are aware, has been ea ...
File
... • The functional group of the carboxylic acid is -COOH (carboxyl group) • This carboxyl group is always attached to the end carbon • Carboxylic acids are polar and therefore dissolve in water • The O-H part of the carboxyl group provides hydrogen bonding (see bonding) ...
... • The functional group of the carboxylic acid is -COOH (carboxyl group) • This carboxyl group is always attached to the end carbon • Carboxylic acids are polar and therefore dissolve in water • The O-H part of the carboxyl group provides hydrogen bonding (see bonding) ...
Chemicals: What`s in? What`s out?
... Mention the word chemistry in a middle level classroom and the first thing students want to know is, “Will we be blowing anything up?” Chemistry should be fun and exciting, but much preparation and skill are needed by the teacher and students in working with chemicals. Unfortunately, accidents do ha ...
... Mention the word chemistry in a middle level classroom and the first thing students want to know is, “Will we be blowing anything up?” Chemistry should be fun and exciting, but much preparation and skill are needed by the teacher and students in working with chemicals. Unfortunately, accidents do ha ...
Oxidation of alcohols and aldehydes
... Further oxidation of a primary alcohol • Using a process known as REFLUX, the reaction contents are continually heated at their boiling point temperature, so HOTTER and LONGER heating then alcohol conversion to an aldehyde • Still uses acidified potassium dichromate Primary + Oxidising Carboxylic ...
... Further oxidation of a primary alcohol • Using a process known as REFLUX, the reaction contents are continually heated at their boiling point temperature, so HOTTER and LONGER heating then alcohol conversion to an aldehyde • Still uses acidified potassium dichromate Primary + Oxidising Carboxylic ...
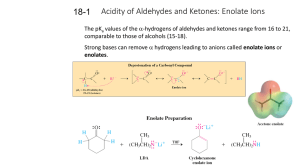
18-1 Enolates (PPT)
... For ordinary aldehydes and ketones, only traces of the enol form are present. The enol form is less stable by 8-12 kcal mol-1. However, for acetaldehyde, the enol form is about 100-times more stable than that of acetone because the less substituted aldehyde carbonyl is more stable than the more subs ...
... For ordinary aldehydes and ketones, only traces of the enol form are present. The enol form is less stable by 8-12 kcal mol-1. However, for acetaldehyde, the enol form is about 100-times more stable than that of acetone because the less substituted aldehyde carbonyl is more stable than the more subs ...
II. Acids and Bases
... substance that contains a hydroxide group and dissociates to produce a hydroxide ion in aqueous solution (ex: NaOH). 4. HCl(g) H+(aq) + Cl-(aq) 5. NaOH(s) Na+(aq) + OH-(aq) ...
... substance that contains a hydroxide group and dissociates to produce a hydroxide ion in aqueous solution (ex: NaOH). 4. HCl(g) H+(aq) + Cl-(aq) 5. NaOH(s) Na+(aq) + OH-(aq) ...
Lecture Notes 7 - La Salle University
... catalyzes the oxidation of substrates with hydrogen peroxide. The related enzyme catalase, also containing heme, catalyzes the breakdown of hydrogen peroxide to water and oxygen. Other heme-containing proteins include the cytochromes, which serve as one-electron carriers in the electron transport ...
... catalyzes the oxidation of substrates with hydrogen peroxide. The related enzyme catalase, also containing heme, catalyzes the breakdown of hydrogen peroxide to water and oxygen. Other heme-containing proteins include the cytochromes, which serve as one-electron carriers in the electron transport ...
Lab Stoichiometry problems Dr. Baxley 1. Lithium metal reacts with
... a. What mass of mercury (II) bromide can be produced from 5.00 mL of mercury (d = 13.6 g/mL) when added to 5.00 mL of bromine (d = 3.10 g/mL). b. What mass of which reactant is left unreacted? 4. Aluminum nitrite and ammonium chloride react to form aluminum chloride, nitrogen and water. a. What mass ...
... a. What mass of mercury (II) bromide can be produced from 5.00 mL of mercury (d = 13.6 g/mL) when added to 5.00 mL of bromine (d = 3.10 g/mL). b. What mass of which reactant is left unreacted? 4. Aluminum nitrite and ammonium chloride react to form aluminum chloride, nitrogen and water. a. What mass ...
I. ALDEHYDES AND KETONES Carbonyl compounds are
... most often used in addition reactions involving carbonyl compounds, as shown above. The product of these addition reactions is typically a secondary or tertiary alcohol (primary alcohols can be formed by reaction with formaldehyde), as shown in the examples below; in these the carbonyl and halide po ...
... most often used in addition reactions involving carbonyl compounds, as shown above. The product of these addition reactions is typically a secondary or tertiary alcohol (primary alcohols can be formed by reaction with formaldehyde), as shown in the examples below; in these the carbonyl and halide po ...
CAMBRIDGE INTERNATIONAL EXAMINATIONS
... The diagrams show mixtures of chemicals that react to produce gases. In which reaction will the litmus paper change colour? A ...
... The diagrams show mixtures of chemicals that react to produce gases. In which reaction will the litmus paper change colour? A ...
Study Guide KEY Exam III F 2012
... Primary (1o) structure of proteins is the amino acid sequence. It is maintained by covalent bonds called peptide bonds. Secondary (2o) structure is a regular repeating structure due to folding of the polypeptide chain. The main types are alpha-helix and beta sheet (either parallel or anti-parallel). ...
... Primary (1o) structure of proteins is the amino acid sequence. It is maintained by covalent bonds called peptide bonds. Secondary (2o) structure is a regular repeating structure due to folding of the polypeptide chain. The main types are alpha-helix and beta sheet (either parallel or anti-parallel). ...
PPT
... • Hydrogen bonding between unsubstituted amides causes them to have high melting points. ...
... • Hydrogen bonding between unsubstituted amides causes them to have high melting points. ...
1.7 FUNCTIONAL GROUPS
... 3) A carboxylic acid has the C atom of the carbonyl group (C=O) bonded to an OH group, as shown below. Even though you would be tempted to label the OH on the carbonyl group as an alcohol, the presence of the C=O completely changes the chemical properties of the OH and it does not have the propertie ...
... 3) A carboxylic acid has the C atom of the carbonyl group (C=O) bonded to an OH group, as shown below. Even though you would be tempted to label the OH on the carbonyl group as an alcohol, the presence of the C=O completely changes the chemical properties of the OH and it does not have the propertie ...
Optical Isomers or Enantiomers
... Polarity of the molecule is not as important as the strength of the carbon-halogen bond. Iodine is the least polar, but the most reactive, since the carbon-iodine bond is the weakest. Reactivity of halogenoalkanes depends on the ability of the halogen bond to break and leave, which is why the haloge ...
... Polarity of the molecule is not as important as the strength of the carbon-halogen bond. Iodine is the least polar, but the most reactive, since the carbon-iodine bond is the weakest. Reactivity of halogenoalkanes depends on the ability of the halogen bond to break and leave, which is why the haloge ...
Halogens - Cronodon
... The chlorine has displaced the bromide because chlorine is a stronger oxidising agent than bromine (equivalently bromide is a stronger reducing agent than chloride). The oxidising power of the halogens decreases in the order: F2 > Cl2 > Br2 > I2 (oxidising strength) Q.16. When a halogen oxidises ano ...
... The chlorine has displaced the bromide because chlorine is a stronger oxidising agent than bromine (equivalently bromide is a stronger reducing agent than chloride). The oxidising power of the halogens decreases in the order: F2 > Cl2 > Br2 > I2 (oxidising strength) Q.16. When a halogen oxidises ano ...
AlCl3 in modern chemistry of polyfluoroarenes
... AlCl3 application in the chemistry of polyfluoroarenes beginning since 1995y. As substrates or reagents there are given fluorinated compounds containing two and more fluorine atoms in one aromatic ring and the transformations are systematized according to the type of the reactions taking place. 1. F ...
... AlCl3 application in the chemistry of polyfluoroarenes beginning since 1995y. As substrates or reagents there are given fluorinated compounds containing two and more fluorine atoms in one aromatic ring and the transformations are systematized according to the type of the reactions taking place. 1. F ...
Preparation of Alcohols
... F: Hydroxide attacks the less hindered carbon to give the trans diol. Carey 16.12-13. G: Alcohols are formed by the use of hydride reagents: LiAlH4, BH3. NaBH4 doesn't work (not reactive enough), even though BH3 does. Go figure. Carey Table 15.3. H: Two ways to do this, depending on which part of th ...
... F: Hydroxide attacks the less hindered carbon to give the trans diol. Carey 16.12-13. G: Alcohols are formed by the use of hydride reagents: LiAlH4, BH3. NaBH4 doesn't work (not reactive enough), even though BH3 does. Go figure. Carey Table 15.3. H: Two ways to do this, depending on which part of th ...
Working with Hazardous Chemicals
... In some articles in Organic Syntheses, chemical-specific hazards are highlighted in red “Caution Notes” within a procedure. It is important to recognize that the absence of a caution note does not imply that no significant hazards are associated with the chemicals involved in that procedure. Prior t ...
... In some articles in Organic Syntheses, chemical-specific hazards are highlighted in red “Caution Notes” within a procedure. It is important to recognize that the absence of a caution note does not imply that no significant hazards are associated with the chemicals involved in that procedure. Prior t ...
Chem Reactions (and Balancing Equations)
... • Two things replace each other. • Reactants must be two ionic compounds or acids. • Usually in aqueous solution AB + CD AD + CB ...
... • Two things replace each other. • Reactants must be two ionic compounds or acids. • Usually in aqueous solution AB + CD AD + CB ...
File
... EXAMPLE 2 (Mass-Mass): If excess sulfuric acid reacts with the 30.0 g of sodium chloride, how many grams of sodium sulfate are produced? H2SO4 + 2NaCl 2HCl + Na2SO4 EXAMPLE 3 (Volume-Volume): What volume of oxygen is used to convert 75.0 L of SO 2 to SO3? Assume STP. 2SO2 + O2 2SO3 EXAMPLE 4 (Vo ...
... EXAMPLE 2 (Mass-Mass): If excess sulfuric acid reacts with the 30.0 g of sodium chloride, how many grams of sodium sulfate are produced? H2SO4 + 2NaCl 2HCl + Na2SO4 EXAMPLE 3 (Volume-Volume): What volume of oxygen is used to convert 75.0 L of SO 2 to SO3? Assume STP. 2SO2 + O2 2SO3 EXAMPLE 4 (Vo ...
CH1710 PrEX#2 Sp2013 answers
... _____ 18. The titration of 25.0 mL of an unknown concentration H2SO4 solution requires 83.6 mL of 0.12 M LiOH solution. What is the concentration of the H2SO4 solution (in M)? A) 0.20 M B) 0.40 M C) 0.10 M D) 0.36 M _____ 19. Which of the following is an acid base reaction? A) C(s) + O2(g) → CO2(g) ...
... _____ 18. The titration of 25.0 mL of an unknown concentration H2SO4 solution requires 83.6 mL of 0.12 M LiOH solution. What is the concentration of the H2SO4 solution (in M)? A) 0.20 M B) 0.40 M C) 0.10 M D) 0.36 M _____ 19. Which of the following is an acid base reaction? A) C(s) + O2(g) → CO2(g) ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.