A little less conjugation, a little more accuracy
... The modification of proteins with fluorophores, drugs and polymers is required for many applications, yet conjugation reactions often generate a heterogeneous mixture of products. A collection of articles in this issue focuses on methods to modify proteins in a site-selective manner. Proteins can fo ...
... The modification of proteins with fluorophores, drugs and polymers is required for many applications, yet conjugation reactions often generate a heterogeneous mixture of products. A collection of articles in this issue focuses on methods to modify proteins in a site-selective manner. Proteins can fo ...
Introduction to metabolism. Specific and general pathways of
... maintenance, growth and reproduction Catabolism is characterized by oxidation reactions and by release of free energy which is transformed to ATP. Anabolism is characterized by reduction reactions and by utilization of energy accumulated in ATP molecules. Catabolism and anabolism are tightly linked ...
... maintenance, growth and reproduction Catabolism is characterized by oxidation reactions and by release of free energy which is transformed to ATP. Anabolism is characterized by reduction reactions and by utilization of energy accumulated in ATP molecules. Catabolism and anabolism are tightly linked ...
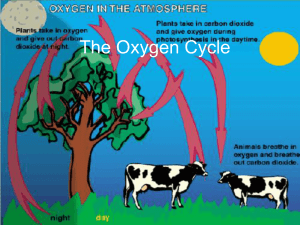
The Oxygen Cycle
... microorganisms in the pond begin to break down organic matter, consuming oxygen in the process. ...
... microorganisms in the pond begin to break down organic matter, consuming oxygen in the process. ...
ppt file/carboxilase
... 2.) Phospholipid (PL) and sphingolipid (SL) synthesis also requires sufficient carbohydrate level normally everywhere. 3.) In brain the FA transport across the blood-brain barrier is not significient, therefore CNS must synthesize its onw fats and can not take it up. In case of PC deficiency the fa ...
... 2.) Phospholipid (PL) and sphingolipid (SL) synthesis also requires sufficient carbohydrate level normally everywhere. 3.) In brain the FA transport across the blood-brain barrier is not significient, therefore CNS must synthesize its onw fats and can not take it up. In case of PC deficiency the fa ...
Lesson Plans Week of Sept. 2-5
... - Class Discussion: Water molecule Properties of water pH Organic/inorganic compounds -FA: Exit Writing- Explain how beads of sweat forming on your skin while sweating applies to the following properties of water: adhesion, cohesion, evaporative cooling. Class does not meet. ...
... - Class Discussion: Water molecule Properties of water pH Organic/inorganic compounds -FA: Exit Writing- Explain how beads of sweat forming on your skin while sweating applies to the following properties of water: adhesion, cohesion, evaporative cooling. Class does not meet. ...
Work and Energy in Muscles
... first half-hour. After this period we see that fatty acids and blood glucose takes over as important energy sources. There are approximately 20 grams of glucose in blood and extracellular fluids. Most of the glucose measured in the blood comes from breakdown of liver glycogen. Adrenalin, noradrenali ...
... first half-hour. After this period we see that fatty acids and blood glucose takes over as important energy sources. There are approximately 20 grams of glucose in blood and extracellular fluids. Most of the glucose measured in the blood comes from breakdown of liver glycogen. Adrenalin, noradrenali ...
video slide - SP New Moodle
... • The over structure of a phospholipid consists of – two fatty acids – and a phosphate group ...
... • The over structure of a phospholipid consists of – two fatty acids – and a phosphate group ...
Notes. - Glow Blogs
... carbohydrates and oxygen with the help of sunlight and chlorophyll. CARBOHYDRATE - a compound containing carbon, hydrogen and oxygen in which the ratio of hydrogen:oxygen is the same as in water. ENDOTHERMIC - a reaction in which energy is absorbed (or taken in). CHLOROPHYLL - a green chemical in th ...
... carbohydrates and oxygen with the help of sunlight and chlorophyll. CARBOHYDRATE - a compound containing carbon, hydrogen and oxygen in which the ratio of hydrogen:oxygen is the same as in water. ENDOTHERMIC - a reaction in which energy is absorbed (or taken in). CHLOROPHYLL - a green chemical in th ...
CH395 G Exam 3 Fall 2004 - Multiple Choice 1. Which of the
... a. This process depends on the fact that oxaloacetate can be transported to the bundle sheath cells where the Calvin cycle occurs. b. CO2 enters outer cells of leaves and reacts with phosphoenolpyruvate to ...
... a. This process depends on the fact that oxaloacetate can be transported to the bundle sheath cells where the Calvin cycle occurs. b. CO2 enters outer cells of leaves and reacts with phosphoenolpyruvate to ...
Enzymes
... The coenzyme helps the enzyme function properly and without it, some enzymes may not be able to function at all. Vitamins act as coenzymes. 3) Enzymes have an active site. This is where enzyme action occurs. The enzymes are usually bigger than the molecules that they act upon. ...
... The coenzyme helps the enzyme function properly and without it, some enzymes may not be able to function at all. Vitamins act as coenzymes. 3) Enzymes have an active site. This is where enzyme action occurs. The enzymes are usually bigger than the molecules that they act upon. ...
DNA
... Before the mRNA can go to the ribosome, it needs to be spliced. – The junk (parts of the DNA that are noncoding regions) called introns need to be cut out. – Exons (coding regions) are then stuck together. This is the correct concise message. ...
... Before the mRNA can go to the ribosome, it needs to be spliced. – The junk (parts of the DNA that are noncoding regions) called introns need to be cut out. – Exons (coding regions) are then stuck together. This is the correct concise message. ...
Document
... – Twenty different amino acids are used to build proteins in organisms. – Amino acids differ in side groups, or R groups. ...
... – Twenty different amino acids are used to build proteins in organisms. – Amino acids differ in side groups, or R groups. ...
BIOCHEMISTRY (CHEM 360)
... Why do you think the cysteine side chain is involved here instead of the serine side chain (as in protease enzymes) to form an acyl intermediate? The cysteine side chain forms a thio-ester, instead of a normal ester with serine. Thio-esters are more reactive and thus more susceptible to nucleophilic ...
... Why do you think the cysteine side chain is involved here instead of the serine side chain (as in protease enzymes) to form an acyl intermediate? The cysteine side chain forms a thio-ester, instead of a normal ester with serine. Thio-esters are more reactive and thus more susceptible to nucleophilic ...
Slide 1
... •a polysaccharide is an example of a polymer •polymer – compound made of repeating units called monomers •monomer = monosaccharide •some polysaccharides serve as storage materials – hydrolyzed into individual monosaccharides – for energy production -animals = glycogen (highly branched polymer of glu ...
... •a polysaccharide is an example of a polymer •polymer – compound made of repeating units called monomers •monomer = monosaccharide •some polysaccharides serve as storage materials – hydrolyzed into individual monosaccharides – for energy production -animals = glycogen (highly branched polymer of glu ...
Metabolism
... • Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells. ...
... • Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells. ...
1. A nucleotide is a ______. 2. DNA consists of two antiparallel
... The enzyme that catalyzes the synthesis of new DNA molecules is called The function of ribosomes in the cell is _______ Similar to the complementary purine-pyrimidine relationship observed in DNA, which of the following choices pairs with adenine in RNA? If the DNA sequence is ATCGCTCC, the corresp ...
... The enzyme that catalyzes the synthesis of new DNA molecules is called The function of ribosomes in the cell is _______ Similar to the complementary purine-pyrimidine relationship observed in DNA, which of the following choices pairs with adenine in RNA? If the DNA sequence is ATCGCTCC, the corresp ...
1 | Page Glossary: Atom: Molecule: Compound: Atomic number
... • Contractile (regulate strength & speed of heart contractions) e.g. myosin, actin - Amino acids consist of: C atom, amino group, carboxyl groups, side chain Enzymes: special proteins that catalyse (speed up) ...
... • Contractile (regulate strength & speed of heart contractions) e.g. myosin, actin - Amino acids consist of: C atom, amino group, carboxyl groups, side chain Enzymes: special proteins that catalyse (speed up) ...
Chapter 2 Outline
... a. bitter taste, slippery feel b. dissociate into hydroxyl ions NaOH Na+ + OHc. HCO3- bicarbonate ion is an important buffer in the blood d. pH range 8-14 VIII. Organic compounds – All contain carbon A. Carbohydrates – sugars and starches provide energy for the cell 1. Composed of simple monosacch ...
... a. bitter taste, slippery feel b. dissociate into hydroxyl ions NaOH Na+ + OHc. HCO3- bicarbonate ion is an important buffer in the blood d. pH range 8-14 VIII. Organic compounds – All contain carbon A. Carbohydrates – sugars and starches provide energy for the cell 1. Composed of simple monosacch ...
sickle cell anemia explained by protein shape, northeast 2012
... This unit would be taught early in an Introductory Biology course as part of a broader discussion about biological macromolecules. Students would have already been introduced to major themes in biology, such as the chemistry of life (including atomic structure and types of chemical bonds). They will ...
... This unit would be taught early in an Introductory Biology course as part of a broader discussion about biological macromolecules. Students would have already been introduced to major themes in biology, such as the chemistry of life (including atomic structure and types of chemical bonds). They will ...
Ch. 9 Cellular Respiration
... • Because the reactions that produce CO2 + alcohol or lactic acid are needed to reoxidize NADH. Without this the lack of NAD+ would stop glycolysis. ...
... • Because the reactions that produce CO2 + alcohol or lactic acid are needed to reoxidize NADH. Without this the lack of NAD+ would stop glycolysis. ...
glucose, faKy acids, amino acids
... • PhosphorylaDon is the process of adding a phosphate group to an organic molecule (oNen a protein) to acDvate or inacDvate the molecule. • ATP is oNen a source of phosphate groups for these rea ...
... • PhosphorylaDon is the process of adding a phosphate group to an organic molecule (oNen a protein) to acDvate or inacDvate the molecule. • ATP is oNen a source of phosphate groups for these rea ...
C h e m g u i d e ... CARBOXYLIC ACIDS: REDUCTION
... b) Describe the bonding between the aluminium and the four hydrogens in the [AlH4]- ion. c) Writing the reducing agent as [H], write a simple equation to show the reduction of propanoic acid. d) The reaction is carried out at room temperature in dry ethoxyethane (diethyl ether) as solvent. Why must ...
... b) Describe the bonding between the aluminium and the four hydrogens in the [AlH4]- ion. c) Writing the reducing agent as [H], write a simple equation to show the reduction of propanoic acid. d) The reaction is carried out at room temperature in dry ethoxyethane (diethyl ether) as solvent. Why must ...
57 chapter summary
... 4. Jimmy, a 12-year-old boy, was awakened suddenly by a loud crash. As he sat up in bed, straining to listen, his fright was revealed by his rapid breathing (hyperventilation), a breathing pattern effective in ridding the blood of CO2. At this point, was his blood pH rising or falling? 5. After you ...
... 4. Jimmy, a 12-year-old boy, was awakened suddenly by a loud crash. As he sat up in bed, straining to listen, his fright was revealed by his rapid breathing (hyperventilation), a breathing pattern effective in ridding the blood of CO2. At this point, was his blood pH rising or falling? 5. After you ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.