questions based on high order thinking skill - Entrance
... Q. 1. Which solution will allow greater conductance of electricity, 1 M NaCl at 293 K or 1 M NaCl at 323 K and why ? Ans. 1 M NaCl at 323 K as the ionic mobilities increase with increase in temperature. Q. 2. What does the negative value of E°cell indicate ? Ans. G will be positive, the cell will n ...
... Q. 1. Which solution will allow greater conductance of electricity, 1 M NaCl at 293 K or 1 M NaCl at 323 K and why ? Ans. 1 M NaCl at 323 K as the ionic mobilities increase with increase in temperature. Q. 2. What does the negative value of E°cell indicate ? Ans. G will be positive, the cell will n ...
questions based on high order thinking skill
... Q. 1. Which solution will allow greater conductance of electricity, 1 M NaCl at 293 K or 1 M NaCl at 323 K and why ? Ans. 1 M NaCl at 323 K as the ionic mobilities increase with increase in temperature. Q. 2. What does the negative value of E°cell indicate ? Ans. G will be positive, the cell will n ...
... Q. 1. Which solution will allow greater conductance of electricity, 1 M NaCl at 293 K or 1 M NaCl at 323 K and why ? Ans. 1 M NaCl at 323 K as the ionic mobilities increase with increase in temperature. Q. 2. What does the negative value of E°cell indicate ? Ans. G will be positive, the cell will n ...
Chapter 3 Alcohols, Phenols, and Ethers
... – in phenols, —OH is connected to a benzene ring. (The “parent” molecule of this class is also named phenol: PhOH or C6H5OH.) • When two carbon groups are connected by single bonds to an oxygen, this is classified as the ether functional group. R ...
... – in phenols, —OH is connected to a benzene ring. (The “parent” molecule of this class is also named phenol: PhOH or C6H5OH.) • When two carbon groups are connected by single bonds to an oxygen, this is classified as the ether functional group. R ...
Organic Chemistry: Introduction
... – bonds are omitted, repeated groups put together, side chains put in brackets • CH3CH2CH2CH2CH2CH3 –or even CH3(CH2)4CH3 • CH3CH(CH3)CH3 (this is not the molecule above) ...
... – bonds are omitted, repeated groups put together, side chains put in brackets • CH3CH2CH2CH2CH2CH3 –or even CH3(CH2)4CH3 • CH3CH(CH3)CH3 (this is not the molecule above) ...
Topic 10.1 Fundametals of Organic Chemistry
... – not accepted in the IB for answers but often used in questions – every “corner” represents a carbon – hydrogens are implied ...
... – not accepted in the IB for answers but often used in questions – every “corner” represents a carbon – hydrogens are implied ...
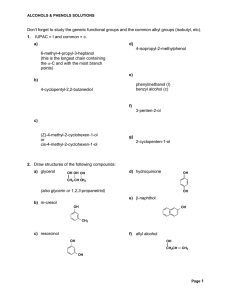
Don`t forget to study the generic functional groups and the common
... Only a rhodium catalyst allows hydrogenation of H2/Rh an aromatic ring at standard pressure and temperature. Carboxylic acids are not reduced by hydrogen. ...
... Only a rhodium catalyst allows hydrogenation of H2/Rh an aromatic ring at standard pressure and temperature. Carboxylic acids are not reduced by hydrogen. ...
4134gdisk doc..4134gdisk chapter .. Page501
... case of the oxidation of hypophosphite by trans-[RuVI(L)(O)2]2+ (L is 1,2-dimethyl3,4+9,10-dibenzo-1,12-diaza-5,8-dioxacyclopentadecane) the mechanism has hydride transfer to RuNO as a rate determining step.81 Tris(bipyridyl)ruthenium(II) modifies mono-, di- and tri-nuclear manganese complexes as e ...
... case of the oxidation of hypophosphite by trans-[RuVI(L)(O)2]2+ (L is 1,2-dimethyl3,4+9,10-dibenzo-1,12-diaza-5,8-dioxacyclopentadecane) the mechanism has hydride transfer to RuNO as a rate determining step.81 Tris(bipyridyl)ruthenium(II) modifies mono-, di- and tri-nuclear manganese complexes as e ...
On The catalytic Hydrogenation of Co2 and Carboxylic acid esters
... equivalent is added to a substrate in the presence of a catalyst, catalytic hydrogenation reactions have the greatest impact on the human society. Discovered by Paul Sabatier in 1897,1 catalytic hydrogenation sparked the rapid development of the chemical industry. Probably the most remarkable exampl ...
... equivalent is added to a substrate in the presence of a catalyst, catalytic hydrogenation reactions have the greatest impact on the human society. Discovered by Paul Sabatier in 1897,1 catalytic hydrogenation sparked the rapid development of the chemical industry. Probably the most remarkable exampl ...
Chemical Redox Agents for Organometallic
... The authors (Bill Geiger, left; Neil Connelly, right) have been at the forefront of organometallic electrochemistry for more than 20 years and have had a long-standing and fruitful collaboration. Neil Connelly took his B.Sc. (1966) and Ph.D. (1969, under the direction of Jon McCleverty) degrees at t ...
... The authors (Bill Geiger, left; Neil Connelly, right) have been at the forefront of organometallic electrochemistry for more than 20 years and have had a long-standing and fruitful collaboration. Neil Connelly took his B.Sc. (1966) and Ph.D. (1969, under the direction of Jon McCleverty) degrees at t ...
Chemical Redox Agents for Organometallic
... The authors (Bill Geiger, left; Neil Connelly, right) have been at the forefront of organometallic electrochemistry for more than 20 years and have had a long-standing and fruitful collaboration. Neil Connelly took his B.Sc. (1966) and Ph.D. (1969, under the direction of Jon McCleverty) degrees at t ...
... The authors (Bill Geiger, left; Neil Connelly, right) have been at the forefront of organometallic electrochemistry for more than 20 years and have had a long-standing and fruitful collaboration. Neil Connelly took his B.Sc. (1966) and Ph.D. (1969, under the direction of Jon McCleverty) degrees at t ...
Ch14_PT MULTIPLE CHOICE. Choose the one alternative that best
... 44) As the molar mass of these alcohols increases, the water solubility decreases. This occurs because the polarity of the hydroxyl group, which is the reason for the interaction with the polar water molecules, becomes less important as the size of the nonpolar hydrocarbon portion of the molecule in ...
... 44) As the molar mass of these alcohols increases, the water solubility decreases. This occurs because the polarity of the hydroxyl group, which is the reason for the interaction with the polar water molecules, becomes less important as the size of the nonpolar hydrocarbon portion of the molecule in ...
Organic handouts - Moore Chemistry
... 8-carbons octane Cycloalkanes These hydrocarbons exist in ring structures with general formula CnH2n. There is considerable bond strain for 3 and 4 carbon rings. 5 and 6 carbon rings have much less bond strain. They can buckle their structures to achieve more stability. You name them just as alkanes ...
... 8-carbons octane Cycloalkanes These hydrocarbons exist in ring structures with general formula CnH2n. There is considerable bond strain for 3 and 4 carbon rings. 5 and 6 carbon rings have much less bond strain. They can buckle their structures to achieve more stability. You name them just as alkanes ...
(omit), and Epoxides
... • IUPAC: the longest carbon chain is the parent. Name the OR group as an alkoxy substituent. • Common names: name the groups bonded to oxygen in alphabetical order followed by the word ether. OH ...
... • IUPAC: the longest carbon chain is the parent. Name the OR group as an alkoxy substituent. • Common names: name the groups bonded to oxygen in alphabetical order followed by the word ether. OH ...
- University of Bath Opus
... sulfonates are classified as genotoxic and the pharmaceutical industry is required to control these compounds to extremely low levels in APIs for patient safety. It follows that any methodology that avoids the need to convert alcohols to halides or sulfonates will offer significant benefits to both ...
... sulfonates are classified as genotoxic and the pharmaceutical industry is required to control these compounds to extremely low levels in APIs for patient safety. It follows that any methodology that avoids the need to convert alcohols to halides or sulfonates will offer significant benefits to both ...
08 Redox Reactions
... The redox reactions are a very important group of reactions which occur with almost every element. These reactions are involved in large number of processes in nature (biological and industrial). They take place in burning of fuels such as gasoline, oil, natural gas, organic substances of carbon and ...
... The redox reactions are a very important group of reactions which occur with almost every element. These reactions are involved in large number of processes in nature (biological and industrial). They take place in burning of fuels such as gasoline, oil, natural gas, organic substances of carbon and ...
Infrared - ResearchGate
... General trends: •Stretching frequencies are higher than bending frequencies (it is easier to bend a bond than stretching or compresing them) ...
... General trends: •Stretching frequencies are higher than bending frequencies (it is easier to bend a bond than stretching or compresing them) ...
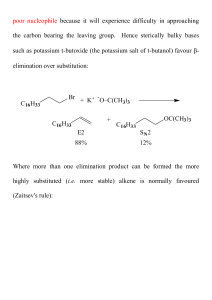
Alkenes 4 - ChemWeb (UCC)
... Unlike the case of the E2 mechanism - there is no steric requirement for the conformation of the substrate in an E1 reaction. ...
... Unlike the case of the E2 mechanism - there is no steric requirement for the conformation of the substrate in an E1 reaction. ...
File
... In other words, a ________ value for Keq means that at equilibrium, there is _____________ and very little reactant left. The larger the value for Keq the closer to completion the reaction is at equilibrium. (NOTE: "Completion" means reactants have been completely converted to products.) A very smal ...
... In other words, a ________ value for Keq means that at equilibrium, there is _____________ and very little reactant left. The larger the value for Keq the closer to completion the reaction is at equilibrium. (NOTE: "Completion" means reactants have been completely converted to products.) A very smal ...
Asymmetric induction

Asymmetric induction (also enantioinduction) in stereochemistry describes the preferential formation in a chemical reaction of one enantiomer or diastereoisomer over the other as a result of the influence of a chiral feature present in the substrate, reagent, catalyst or environment. Asymmetric induction is a key element in asymmetric synthesis.Asymmetric induction was introduced by Hermann Emil Fischer based on his work on carbohydrates. Several types of induction exist.Internal asymmetric induction makes use of a chiral center bound to the reactive center through a covalent bond and remains so during the reaction. The starting material is often derived from chiral pool synthesis. In relayed asymmetric induction the chiral information is introduced in a separate step and removed again in a separate chemical reaction. Special synthons are called chiral auxiliaries. In external asymmetric induction chiral information is introduced in the transition state through a catalyst of chiral ligand. This method of asymmetric synthesis is economically most desirable.