File
... • A protein with a D amino acid instead of L will have its R group sticking out in the wrong direction. • Many other kinds of organic molecules exist as enantiomers. Usually only one form is active in biological systems. For example, if one form binds to a receptor protein on the surface of a cell, ...
... • A protein with a D amino acid instead of L will have its R group sticking out in the wrong direction. • Many other kinds of organic molecules exist as enantiomers. Usually only one form is active in biological systems. For example, if one form binds to a receptor protein on the surface of a cell, ...
Organic Chemistry Unit Test
... The molecule above is which of the following? a. a saturated fatty acid d. palmitin b. a triglyceride e. a natural oil found in grain c. an unsaturated fatty acid ____ 2. What is the monomer for the polymer Kel-F shown below? ...
... The molecule above is which of the following? a. a saturated fatty acid d. palmitin b. a triglyceride e. a natural oil found in grain c. an unsaturated fatty acid ____ 2. What is the monomer for the polymer Kel-F shown below? ...
PowerPoint Presentation - No Slide Title
... Compare a molecular formula to an alkane’s: every TWO Hydrogens less = I degree of unsaturation ...
... Compare a molecular formula to an alkane’s: every TWO Hydrogens less = I degree of unsaturation ...
4.4 Formation of Esters from Carboxylic Acids and Alcohols
... process. Note that a water molecule is removed in the process of forming the ester from the carboxylic acid and the alcohol. The water comes from removing an OH group on the carboxylic acid and combining it with a H. One can show this using “lasso” chemistry as shown ...
... process. Note that a water molecule is removed in the process of forming the ester from the carboxylic acid and the alcohol. The water comes from removing an OH group on the carboxylic acid and combining it with a H. One can show this using “lasso” chemistry as shown ...
BIOCHEMISTRY TEST – PRACTICE QUESTIONS
... condition or event manipulated by the investigator (B) condition or event that may change due to the other variable (C) condition which the investigator attempts to keep the same (D) same as the dependent variable ...
... condition or event manipulated by the investigator (B) condition or event that may change due to the other variable (C) condition which the investigator attempts to keep the same (D) same as the dependent variable ...
www.xtremepapers.net
... excess HCl(aq) at a low temperature (below 5oC) converts phenylamine into (a solution of) benzenediazonium chloride. On warming to about 50oC, this decomposes to phenol and nitrogen gas (usually in not a very good yield, but enough to be able to smell the phenol). Both these reactions can be illustr ...
... excess HCl(aq) at a low temperature (below 5oC) converts phenylamine into (a solution of) benzenediazonium chloride. On warming to about 50oC, this decomposes to phenol and nitrogen gas (usually in not a very good yield, but enough to be able to smell the phenol). Both these reactions can be illustr ...
WM5 The synthesis of salicylic acid and aspirin
... 1898: 2-ethanoylhydroxybenzoic acid (a.k.a acetylsalicylic acid or aspirin) produced ...
... 1898: 2-ethanoylhydroxybenzoic acid (a.k.a acetylsalicylic acid or aspirin) produced ...
Name__________________________Review Organic Reactions
... 2. Given the balanced equation for an organic reaction: C2H2 + 2Cl 2 C2H2Cl 4 This reaction is best classified as A) addition C) fermentation ...
... 2. Given the balanced equation for an organic reaction: C2H2 + 2Cl 2 C2H2Cl 4 This reaction is best classified as A) addition C) fermentation ...
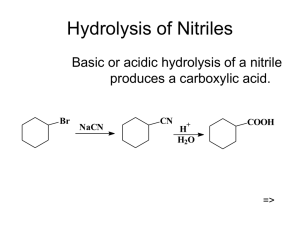
Hydrolysis of Nitriles
... CH2N2 reacts with carboxylic acids to produce methyl esters quantitatively. Very toxic, explosive. Dissolve in ether. O ...
... CH2N2 reacts with carboxylic acids to produce methyl esters quantitatively. Very toxic, explosive. Dissolve in ether. O ...
last year`s April exam
... b) Draw a picture that shows how water molecules can interact with an aldehyde through H-bonding, showing all possible interactions. ...
... b) Draw a picture that shows how water molecules can interact with an aldehyde through H-bonding, showing all possible interactions. ...
INTRODUCTION
... Second generation breathalyzer tests measure the amount of water produced in the reaction, for a more accurate measure. Secondary alcohols (OH attached to a non-terminal carbon) may be oxidized in a single stage to the corresponding ketone. The general reaction is: secondary alcohol + oxidizing agen ...
... Second generation breathalyzer tests measure the amount of water produced in the reaction, for a more accurate measure. Secondary alcohols (OH attached to a non-terminal carbon) may be oxidized in a single stage to the corresponding ketone. The general reaction is: secondary alcohol + oxidizing agen ...
32 GRIGNARD REACTION Alkyl halides can react with magnesium
... reagent reacts with water to give hydrocarbons (benzene in this case). Mount a 50 mL round bottom flask with a condenser. Do not run water through your condenser. Place 0.7 g Mg turnings in the flask. Add 10 mL of dry ether, followed by 0.5 mL of bromobenzene. Your TA will then come around and add a ...
... reagent reacts with water to give hydrocarbons (benzene in this case). Mount a 50 mL round bottom flask with a condenser. Do not run water through your condenser. Place 0.7 g Mg turnings in the flask. Add 10 mL of dry ether, followed by 0.5 mL of bromobenzene. Your TA will then come around and add a ...
Chapter 9
... presence of hydroxide ion (OH-) they split to form a carboxylate ion and an alcohol. Reaction used to make soap. ...
... presence of hydroxide ion (OH-) they split to form a carboxylate ion and an alcohol. Reaction used to make soap. ...
Document
... Water (hydro) is used to split (lyse) a molecule. Esters are one class of molecules that undergo hydrolysis – when treated with water in the presence of hydroxide ion (OH-) they split to form a carboxylate ion and an alcohol. Reaction used to make soap. ...
... Water (hydro) is used to split (lyse) a molecule. Esters are one class of molecules that undergo hydrolysis – when treated with water in the presence of hydroxide ion (OH-) they split to form a carboxylate ion and an alcohol. Reaction used to make soap. ...
CHEM 202_ Part 2
... Aspirin is a common carboxylic acid. They are weak acids (pKa around 5) compared with mineral acids (pKa around -1), but they are more acidic than alcohols or phenols (because of resonance structure). ...
... Aspirin is a common carboxylic acid. They are weak acids (pKa around 5) compared with mineral acids (pKa around -1), but they are more acidic than alcohols or phenols (because of resonance structure). ...
L-13
... of allylmagnesium chloride), which is a typical highly nucleophilic reagent, instead of 1, but the starting alcohol was recovered after work-up under conditions with or without the InCl3 catalyst. These results strongly suggest that the appropriate nucleophilicity of the allylic reagent and Lewis ac ...
... of allylmagnesium chloride), which is a typical highly nucleophilic reagent, instead of 1, but the starting alcohol was recovered after work-up under conditions with or without the InCl3 catalyst. These results strongly suggest that the appropriate nucleophilicity of the allylic reagent and Lewis ac ...
Part (d) The Birch Reduction of Nitrogen
... As the constituents of poly amides (ie peptides) these functional groups are essential parts of biological systems. We can hydrolyse an amide bond in the laboratory, but require harsh acidic or basic conditions to do it ...
... As the constituents of poly amides (ie peptides) these functional groups are essential parts of biological systems. We can hydrolyse an amide bond in the laboratory, but require harsh acidic or basic conditions to do it ...
polymerisation
... which readily breaks up to form radicals which, in turn, initiate a chain reaction. Another famous type of catalyst is a Ziegler-Natta catalyst (named after the scientists who developed it). Such catalysts are based on the compound TiCl4. ...
... which readily breaks up to form radicals which, in turn, initiate a chain reaction. Another famous type of catalyst is a Ziegler-Natta catalyst (named after the scientists who developed it). Such catalysts are based on the compound TiCl4. ...
SAMPLE PAPER -2 Time Allowed: 3 Hrs
... give different colour with same solution and have different magnetic properties.(At. No. Of Fe=26u) How will you distinguish the following pair of compounds? (i) Aniline and benzyl amine. (ii) Aniline and N-Methyl aniline. Write the following name reaction: (i) Gabriel phthalimide reaction. (ii) Hof ...
... give different colour with same solution and have different magnetic properties.(At. No. Of Fe=26u) How will you distinguish the following pair of compounds? (i) Aniline and benzyl amine. (ii) Aniline and N-Methyl aniline. Write the following name reaction: (i) Gabriel phthalimide reaction. (ii) Hof ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034 PART-A
... ‘Aryldiazonium salt involves both in electrophilic and nucleophilic substitutions’. Justify. What is first order asymmetric transformation? Give an example. How do inductive and field effects affect the second substitution in aromatic systems? In 4-t-butylcyclohexanecarboxylic acid, trans form is mo ...
... ‘Aryldiazonium salt involves both in electrophilic and nucleophilic substitutions’. Justify. What is first order asymmetric transformation? Give an example. How do inductive and field effects affect the second substitution in aromatic systems? In 4-t-butylcyclohexanecarboxylic acid, trans form is mo ...
Esters
... • Ethanoyl chloride is much more reactive, but, also toxic and hazardous to use. • Ethanoic anhydride & ethanoyl chloride are described as acylating agents using an acylating agent is the only way to esterify a phenol alcohols can be esterified using acylating agents or by reacting with a carboxyl ...
... • Ethanoyl chloride is much more reactive, but, also toxic and hazardous to use. • Ethanoic anhydride & ethanoyl chloride are described as acylating agents using an acylating agent is the only way to esterify a phenol alcohols can be esterified using acylating agents or by reacting with a carboxyl ...
Petasis reaction

The Petasis reaction (alternatively called the Petasis borono–Mannich (PBM) reaction) is the chemical reaction of an amine, aldehyde, and vinyl- or aryl-boronic acid to form substituted amines.Reported in 1993 by Nicos Petasis as a practical method towards the synthesis of a geometrically pure antifungal agent, naftifine, the Petasis reaction can be described as a variation of the Mannich reaction. Rather than generating an enolate to form the substituted amine product, in the Petasis reaction, the vinyl group of the organoboronic acid serves as the nucleophile. In comparison to other methods of generating allyl amines, the Petasis reaction tolerates a multifunctional scaffold, with a variety of amines and organoboronic acids as potential starting materials. Additionally, the reaction does not require anhydrous or inert conditions. As a mild, selective synthesis, the Petasis reaction is useful in generating α-amino acids, and is utilized in combinatorial chemistry and drug discovery.