Name - Clark College
... When electron density is shared through resonance, there is a greater extent of electron "sharing" than hyperconjugation. Since the electrons are shared by moving through like orbitals, the allylic radical is more stable. ...
... When electron density is shared through resonance, there is a greater extent of electron "sharing" than hyperconjugation. Since the electrons are shared by moving through like orbitals, the allylic radical is more stable. ...
Chem 130 Fall 2004 Exam 3 Study Guide Chapter 8.1
... Conversion into alkyl halides (with HCl, HBr, SOCl2) Dehydration to form alkene (with H2SO4, concentrated, ∆) Oxidation: Primary alcohol to aldehydes (with PCC) Primary alcohol to carboxylic acids (with CrO3 or K2Cr2O7) Secondary alcohol to ketones (with PCC or CrO3 or K2Cr2O7) Tertiary alcoho ...
... Conversion into alkyl halides (with HCl, HBr, SOCl2) Dehydration to form alkene (with H2SO4, concentrated, ∆) Oxidation: Primary alcohol to aldehydes (with PCC) Primary alcohol to carboxylic acids (with CrO3 or K2Cr2O7) Secondary alcohol to ketones (with PCC or CrO3 or K2Cr2O7) Tertiary alcoho ...
CH 3502 4500
... 16. Discuss the mechanism of cleavage of ethers by HI. 17. Explain Williamson’s synthesis of ethers. 18. Discuss Norrish type-I reaction. 19. Discuss the mechanism of Wittig reaction and its uses in organic synthesis. 20. Explain Wolf-Kishner reduction with its mechanism. 21. Give any two methods o ...
... 16. Discuss the mechanism of cleavage of ethers by HI. 17. Explain Williamson’s synthesis of ethers. 18. Discuss Norrish type-I reaction. 19. Discuss the mechanism of Wittig reaction and its uses in organic synthesis. 20. Explain Wolf-Kishner reduction with its mechanism. 21. Give any two methods o ...
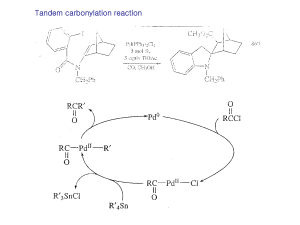
CHE 297 Organic Workshop
... Step 1 A base (use hydroxide ion) removes the acidic alpha hydrogen from a molecule of acetaldehyde (ethanal) to form an enolate anion and a molecule of water. Step 2 The enolate anion attacks the carbonyl group of a second molecule of acetaldehyde. That intermediate is then protonated to form a bet ...
... Step 1 A base (use hydroxide ion) removes the acidic alpha hydrogen from a molecule of acetaldehyde (ethanal) to form an enolate anion and a molecule of water. Step 2 The enolate anion attacks the carbonyl group of a second molecule of acetaldehyde. That intermediate is then protonated to form a bet ...
Chapter Nine: Alcohols, Ethers and Epoxides
... inversion (examples: CH3NH2, CH3O–, HO–, SH–, CN–) iii. Reduction with hydride reducing agent LiAlH4 : 12.6 ...
... inversion (examples: CH3NH2, CH3O–, HO–, SH–, CN–) iii. Reduction with hydride reducing agent LiAlH4 : 12.6 ...
Reduction [H]
... hydrazine to gaseous nitrogen, to yield the corresponding alkane. Numerous modified procedures to the classic Wolff–Kishner reduction have been reported. In general, the improvements have focused on driving hydrazone formation to completion by removal of water, and by the use of high concentrations ...
... hydrazine to gaseous nitrogen, to yield the corresponding alkane. Numerous modified procedures to the classic Wolff–Kishner reduction have been reported. In general, the improvements have focused on driving hydrazone formation to completion by removal of water, and by the use of high concentrations ...
Aldehydes and Ketones
... These carbonyl compounds generally have two reaction pathways – they react with strong nucleophiles (generally, strong nucleophiles have a formal negative charge) under neutral, generally anhydrous conditions, or with weak nucleophiles (those with lone pairs, but no charge) under mild acid catalysis ...
... These carbonyl compounds generally have two reaction pathways – they react with strong nucleophiles (generally, strong nucleophiles have a formal negative charge) under neutral, generally anhydrous conditions, or with weak nucleophiles (those with lone pairs, but no charge) under mild acid catalysis ...
슬라이드 1
... A soluble bis-phosphine complexes, Ni(dppe)2Cl2, is a particularly effective catalyst. The main distinction between this reaction and Pd-catalyzed cross coupling is that the nickel reaction can be more readily applied to saturated alkyl groups because of a reduced tendency for b-elimination. ...
... A soluble bis-phosphine complexes, Ni(dppe)2Cl2, is a particularly effective catalyst. The main distinction between this reaction and Pd-catalyzed cross coupling is that the nickel reaction can be more readily applied to saturated alkyl groups because of a reduced tendency for b-elimination. ...
reduction of ketones and imines with CaH2/ZnX2 in the presence of
... aliphatic aldehydes were cleanly converted to the corresponding amines. Imines having a benzyl, alkyl, or aromatic group as an N-substituent were good substrates. Functional groups such as bromo and alkenyl moieties present in the substrate were tolerant as shown in entries 4 and 6. Ketimine was als ...
... aliphatic aldehydes were cleanly converted to the corresponding amines. Imines having a benzyl, alkyl, or aromatic group as an N-substituent were good substrates. Functional groups such as bromo and alkenyl moieties present in the substrate were tolerant as shown in entries 4 and 6. Ketimine was als ...
AMINO ACIDS Ethan Secor, John N. Gitua (Mentor)
... The proposed reaction for a-amino acid synthesis uses a metallorganic mechanism. A titanium complex coordinates to the C=N double bond, and carbon dioxide inserts into this metallocycle. Upon hydrolysis, the ring is cleaved and the product is formed. The product was isolated using a series of extrac ...
... The proposed reaction for a-amino acid synthesis uses a metallorganic mechanism. A titanium complex coordinates to the C=N double bond, and carbon dioxide inserts into this metallocycle. Upon hydrolysis, the ring is cleaved and the product is formed. The product was isolated using a series of extrac ...
Lecture 15
... • Binding of the alcohol substrate by coordination to zinc • Deprotonation of nicotinamide ribose by His-51 • Deprotonation of Ser-48 by nicotinamide ribose • Deprotonation of the alcohol by Ser-48 • Hydride transfer from the alkoxide ion to NAD+, leading to NADH and a zinc bound aldehyde or ketone ...
... • Binding of the alcohol substrate by coordination to zinc • Deprotonation of nicotinamide ribose by His-51 • Deprotonation of Ser-48 by nicotinamide ribose • Deprotonation of the alcohol by Ser-48 • Hydride transfer from the alkoxide ion to NAD+, leading to NADH and a zinc bound aldehyde or ketone ...
SCH4U Organic Chemistry Portfolio Name: This portfolio is due on
... This portfolio is due on April 22nd, no later. 1. In household fires, it is often the smoke produced that creates a danger as great as the flames. Many plastic materials in the house contain polyvinylchloride (PVC); the formula for this compound is (C2H3Cl)n. Write an equation for the combustion of ...
... This portfolio is due on April 22nd, no later. 1. In household fires, it is often the smoke produced that creates a danger as great as the flames. Many plastic materials in the house contain polyvinylchloride (PVC); the formula for this compound is (C2H3Cl)n. Write an equation for the combustion of ...
A NEW APROACH TO N-SUBSTITUTED OXAZOLIDINE VIA NITRILIUM ION TRAPPING
... Abstract: We have recently demonstrated a direct conversion of secondary alcohols to amides with retention of configuration. This method involved the in situ formation of chlorosulfites followed by a reaction with nitrile complexes of Ti(IV) fluoride. We hypothesize that these amidation reactions in ...
... Abstract: We have recently demonstrated a direct conversion of secondary alcohols to amides with retention of configuration. This method involved the in situ formation of chlorosulfites followed by a reaction with nitrile complexes of Ti(IV) fluoride. We hypothesize that these amidation reactions in ...
Elimination reactions under acidic conditions
... 1. Provide a mechanism for these elimination reactions of alcohols under acidic conditions. ...
... 1. Provide a mechanism for these elimination reactions of alcohols under acidic conditions. ...
Organic Reactions 2.1- 2.3 - mccormack-sch4u-2013
... • Carbon atom forms fewer bonds to Oxygen or more bonds to Hydrogen • Aldehydes, ketones and carboxyliic acids can be “reduced” to alcohols • Alkenes and alkynes can be reduced to become alkanes • Occurs in the presence of reducing agents such as LiAlH4, and H2 where Hydrogen [H] is added ...
... • Carbon atom forms fewer bonds to Oxygen or more bonds to Hydrogen • Aldehydes, ketones and carboxyliic acids can be “reduced” to alcohols • Alkenes and alkynes can be reduced to become alkanes • Occurs in the presence of reducing agents such as LiAlH4, and H2 where Hydrogen [H] is added ...
R-c-H+H-oH:n-J-u oo o il o o o I o
... 14. \tVhichof the following compounds contains a diether linkage? (a) a hemiacetal (b) chloral hydrate (c) aketal (d) camphor 15. \tVhichof the following substancesis used as a preservative of biological specimens? (a) paraldehyde (b) formalin (c) methyl ethyl ketone (d) cinnamaldehyde 16. \Mhich of ...
... 14. \tVhichof the following compounds contains a diether linkage? (a) a hemiacetal (b) chloral hydrate (c) aketal (d) camphor 15. \tVhichof the following substancesis used as a preservative of biological specimens? (a) paraldehyde (b) formalin (c) methyl ethyl ketone (d) cinnamaldehyde 16. \Mhich of ...
chapter 2: reactions of organic compounds
... • Carbon atom forms fewer bonds to Oxygen or more bonds to Hydrogen • Aldehydes, ketones and carboxyliic acids can be “reduced” to alcohols • Alkenes and alkynes can be reduced to become alkanes • Occurs in the presence of reducing agents such as LiAlH4, and H2 where Hydrogen [H] is added ...
... • Carbon atom forms fewer bonds to Oxygen or more bonds to Hydrogen • Aldehydes, ketones and carboxyliic acids can be “reduced” to alcohols • Alkenes and alkynes can be reduced to become alkanes • Occurs in the presence of reducing agents such as LiAlH4, and H2 where Hydrogen [H] is added ...
today`s PowerPoint
... • Both aldehydes and ketones will test positively. No other compounds (e.g. Carboxylic acids or esters) will • The precipitate is called 2,4-dinitrophenylhydrazone ...
... • Both aldehydes and ketones will test positively. No other compounds (e.g. Carboxylic acids or esters) will • The precipitate is called 2,4-dinitrophenylhydrazone ...
1 Carbonyl Condensation Reactions (Conjugate Addition) If we look
... present, then you’re likely looking at an e no late type reaction. 2) GOOD electrophiles. If there’s another strong electrophile around, such as a halogen molecule (Br2, for example), or something like iodomethane (CH3I), you’re looking at an en ola te type of reaction. 3) Catalytic amounts of a wea ...
... present, then you’re likely looking at an e no late type reaction. 2) GOOD electrophiles. If there’s another strong electrophile around, such as a halogen molecule (Br2, for example), or something like iodomethane (CH3I), you’re looking at an en ola te type of reaction. 3) Catalytic amounts of a wea ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.





![Reduction [H]](http://s1.studyres.com/store/data/007148356_1-25f5210a5c809c157bb6e0d32d66fd9d-300x300.png)