Aldehydes and Ketones - University of Nebraska Omaha
... • Given the difference in electronegativity between carbon and magnesium (2.5 – 1.3), the C-Mg bond is polar covalent, with C– and Mg+. • In its reactions, a Grignard reagent behaves as a ...
... • Given the difference in electronegativity between carbon and magnesium (2.5 – 1.3), the C-Mg bond is polar covalent, with C– and Mg+. • In its reactions, a Grignard reagent behaves as a ...
7-1 EXPERIMENT 7: Reduction of Carbonyl Compounds – Achiral
... Department of Chemistry, Iowa State University, Ames IA General Concepts A reduction is often defined as the gain of two hydrogen atoms or the loss of an oxygen atom, or both. This leads to a very important conversion reaction, where aldehydes and ketones are reduced to primary and secondary alcohol ...
... Department of Chemistry, Iowa State University, Ames IA General Concepts A reduction is often defined as the gain of two hydrogen atoms or the loss of an oxygen atom, or both. This leads to a very important conversion reaction, where aldehydes and ketones are reduced to primary and secondary alcohol ...
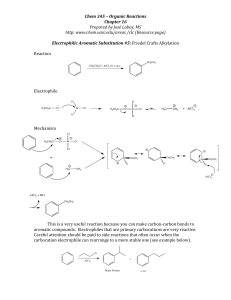
EAS Friedel-Crafts Alkylation
... aromatic compounds. Electrophiles that are primary carbocations are very reactive. Careful attention should be paid to side reactions that often occur when the carbocation electrophile can rearrange to a ...
... aromatic compounds. Electrophiles that are primary carbocations are very reactive. Careful attention should be paid to side reactions that often occur when the carbocation electrophile can rearrange to a ...
1. Acetone can bind to transition metals via oxygen only, or via both
... Why are the outcomes of a and b so different? BH4- is a more strongly coordinating ligand than AlH4-. In general, the more polar Al-H bonds are more inclined to cleave than the less polar B-H bonds. In addition, PBu2Me is a better electron-donor ligand, making the metal more basic and therefore more ...
... Why are the outcomes of a and b so different? BH4- is a more strongly coordinating ligand than AlH4-. In general, the more polar Al-H bonds are more inclined to cleave than the less polar B-H bonds. In addition, PBu2Me is a better electron-donor ligand, making the metal more basic and therefore more ...
PDF(343KB)
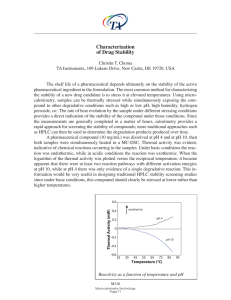
... The shelf life of a pharmaceutical depends ultimately on the stability of the active pharmaceutical ingredient in the formulation. The most common method for characterizing the stability of a new drug candidate is to stress it at elevated temperatures. Using microcalorimetry, samples can be thermall ...
... The shelf life of a pharmaceutical depends ultimately on the stability of the active pharmaceutical ingredient in the formulation. The most common method for characterizing the stability of a new drug candidate is to stress it at elevated temperatures. Using microcalorimetry, samples can be thermall ...
Formation of C-C Bonds via Catalytic Hydrogenation and Transfer
... laboratory reveals reductive C-C bond formation can be achieved under the conditions of catalytic hydrogenation. This concept is extended further via “C-C bond forming transfer hydrogenations”, wherein hydrogen exchange between alcohols and π-unsaturated reactants triggers generation of aldehyde-org ...
... laboratory reveals reductive C-C bond formation can be achieved under the conditions of catalytic hydrogenation. This concept is extended further via “C-C bond forming transfer hydrogenations”, wherein hydrogen exchange between alcohols and π-unsaturated reactants triggers generation of aldehyde-org ...
Relative Reactivity of Aldehydes and Ketones: Generally
... Acidic conditions use neutral water, H2O, as the nucleophile… Neutral carbonyl and neutral water aren’t so very attracted to each other… The oxygen atom in water is electronegative and less willing to share its electron density to form a bond to the carbonyl. We must tweak the system to make them mo ...
... Acidic conditions use neutral water, H2O, as the nucleophile… Neutral carbonyl and neutral water aren’t so very attracted to each other… The oxygen atom in water is electronegative and less willing to share its electron density to form a bond to the carbonyl. We must tweak the system to make them mo ...
Chem 263 Nov 3 2016 notes
... A general example of nucleophilic attack by hydride is shown below. The mechanism involves nucleophilic attack of the hydride to the electrophilic carbon atom of the carbonyl group. Rehybridization of the carbonyl carbon from sp2 to sp3 occurs, and a tetrahedral alkoxide ion intermediate is produced ...
... A general example of nucleophilic attack by hydride is shown below. The mechanism involves nucleophilic attack of the hydride to the electrophilic carbon atom of the carbonyl group. Rehybridization of the carbonyl carbon from sp2 to sp3 occurs, and a tetrahedral alkoxide ion intermediate is produced ...
Properties of , -Unsaturated Aldehydes and Ketones
... ,-Unsaturated aldehydes and ketones undergo the reactions typical of their component functional groups. The conjugated carbonyl group of ,-unsaturated aldehydes and ketones can undergo reactions involving the entire functional system by: ...
... ,-Unsaturated aldehydes and ketones undergo the reactions typical of their component functional groups. The conjugated carbonyl group of ,-unsaturated aldehydes and ketones can undergo reactions involving the entire functional system by: ...
Weekly Review Lecture
... c. Dehydration to nitrile using P2O5 10) Reactions of Weinreb amides a. (single) Grignard addition b. LAH reduction to produce aldehyde 11) Reactions of nitriles a. Hydrolysis b. Reduction with LiAlH4 ...
... c. Dehydration to nitrile using P2O5 10) Reactions of Weinreb amides a. (single) Grignard addition b. LAH reduction to produce aldehyde 11) Reactions of nitriles a. Hydrolysis b. Reduction with LiAlH4 ...
Carboxylic Acid Derivatives
... We've seen this step before in the acid-catalyzed additions of nucleophiles to carbonyl groups. Its purpose is to increase the reactivity of the carbonyl carbon as an electrophile, so that it can be easily attacked by the alcohol oxygen. After nucleophilic attack, there is a proton transfer. Its pur ...
... We've seen this step before in the acid-catalyzed additions of nucleophiles to carbonyl groups. Its purpose is to increase the reactivity of the carbonyl carbon as an electrophile, so that it can be easily attacked by the alcohol oxygen. After nucleophilic attack, there is a proton transfer. Its pur ...
Types of Reactions in Organic Chemistry Chemistry
... Types of Reactions in Organic Chemistry The mechanism of a reaction is the detailed step-by-step description of how the overall reaction occurs. A substitution reaction is a chemical reaction in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms. A substitut ...
... Types of Reactions in Organic Chemistry The mechanism of a reaction is the detailed step-by-step description of how the overall reaction occurs. A substitution reaction is a chemical reaction in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms. A substitut ...
投影片 1
... The positive charge (+) is placed at the carbon attached to the E class function group (e.g.,=O,-OH, -Br) Consonant pattern: Positives charges are placed at carbon atoms bonded to the E class groups ...
... The positive charge (+) is placed at the carbon attached to the E class function group (e.g.,=O,-OH, -Br) Consonant pattern: Positives charges are placed at carbon atoms bonded to the E class groups ...
Exam - Chemistry With BT
... More than one step may be required. Show all reagents and intermediate products. ...
... More than one step may be required. Show all reagents and intermediate products. ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034 PART-A
... 07. How is catalytic hydrogenation different from dissolving metal reduction? Give reasons. 08. What are natural synthons? Give suitable examples. 09. What are protecting groups? How is carbonyl protected and deprotected? 10. What are regiospecific control elements? ...
... 07. How is catalytic hydrogenation different from dissolving metal reduction? Give reasons. 08. What are natural synthons? Give suitable examples. 09. What are protecting groups? How is carbonyl protected and deprotected? 10. What are regiospecific control elements? ...
Selectivity of sodium borohydride
... Lithium aluminum hydride is very reactive and must be used with caution under anhydrous conditions, typically in diethyl ether or tetrahydrofuran (THF) solvents, and is capable of reducing a wide range of functional groups including aldehydes, ketones, esters, carboxylic acids, amides, nitriles and ...
... Lithium aluminum hydride is very reactive and must be used with caution under anhydrous conditions, typically in diethyl ether or tetrahydrofuran (THF) solvents, and is capable of reducing a wide range of functional groups including aldehydes, ketones, esters, carboxylic acids, amides, nitriles and ...
Experiment 7-Reduction
... A reduction is often defined as the gain of two hydrogen atoms or the loss of an oxygen atom, or both. This leads to a very important conversion reaction, where aldehydes and ketones are reduced to primary and secondary alcohols. O ...
... A reduction is often defined as the gain of two hydrogen atoms or the loss of an oxygen atom, or both. This leads to a very important conversion reaction, where aldehydes and ketones are reduced to primary and secondary alcohols. O ...
lec-2- 211(ES +Add)
... HX which can donate a proton, H2O should be able to add to alkenes in the same way as HBr, for example, resulting in the hydration of an alkene. However, for the addition of H2O to alkenes to occur acid catalysts are required. ...
... HX which can donate a proton, H2O should be able to add to alkenes in the same way as HBr, for example, resulting in the hydration of an alkene. However, for the addition of H2O to alkenes to occur acid catalysts are required. ...
Johnson Group Research
... organic transformations. One area of current interest is the development of methodology for the cleavage and functionalization of carbon-carbon bonds. While carbon-carbon single bonds are inert under a vast majority of standard reaction conditions, certain transition metal complexes promote the acti ...
... organic transformations. One area of current interest is the development of methodology for the cleavage and functionalization of carbon-carbon bonds. While carbon-carbon single bonds are inert under a vast majority of standard reaction conditions, certain transition metal complexes promote the acti ...
Chap Thirteen: Alcohols
... its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and reactivity. Predict the reagents needed to produce a given product from an alcohol in one or ...
... its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and reactivity. Predict the reagents needed to produce a given product from an alcohol in one or ...
Outline_CH13_Klein
... its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and reactivity. Predict the reagents needed to produce a given product from an alcohol in one or ...
... its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and reactivity. Predict the reagents needed to produce a given product from an alcohol in one or ...
C h e m g u i d e ... ALCOHOLS: THE REACTION WITH SODIUM
... a) Why is it important to test the pH of the liquid before adding the sodium? b) What would you observe if the liquid was actually an alcohol? c) Observing this isn’t enough to be sure that you have an alcohol. What else might cause the result you described in part (b)? d) Suppose you added a small ...
... a) Why is it important to test the pH of the liquid before adding the sodium? b) What would you observe if the liquid was actually an alcohol? c) Observing this isn’t enough to be sure that you have an alcohol. What else might cause the result you described in part (b)? d) Suppose you added a small ...
Technical Data Sheet - Lučební závody Kolín
... industry and in production of pesticides. In numerous reduction reactions Synhydrid can successfully replace traditional complex hydrides such as LiAlH4, NaAlH4, NaBH4 or their derivatives. It is often preferable to use Synhydrid because of its unusually high solubility in various solvents and becau ...
... industry and in production of pesticides. In numerous reduction reactions Synhydrid can successfully replace traditional complex hydrides such as LiAlH4, NaAlH4, NaBH4 or their derivatives. It is often preferable to use Synhydrid because of its unusually high solubility in various solvents and becau ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.