Document
... Protecting group: Temporarily convert a functional group that is incompatible with a set of reaction conditions into a new functional group (with the protecting group) that is compatible with the reaction. The protecting group is then removed giving the original functional group (deprotection). ...
... Protecting group: Temporarily convert a functional group that is incompatible with a set of reaction conditions into a new functional group (with the protecting group) that is compatible with the reaction. The protecting group is then removed giving the original functional group (deprotection). ...
Aldehydes, Ketones and Carboxylic Acids
... • Can aldehydes and ketones set up hydrogen bonds with each other? • Can they set them up with water? ...
... • Can aldehydes and ketones set up hydrogen bonds with each other? • Can they set them up with water? ...
Exam 3 Review
... focus on chemistry of alcohols, ketones, aldehydes, and organic synthesis. The following textbook sections will be covered on the exam: 17.1 to 17.9 17.12 (NMR of alcohols only) (skip sections 17.10, 17.11) ...
... focus on chemistry of alcohols, ketones, aldehydes, and organic synthesis. The following textbook sections will be covered on the exam: 17.1 to 17.9 17.12 (NMR of alcohols only) (skip sections 17.10, 17.11) ...
Reactions of Alkenes Organic Chemistry
... ** catalyst a reactant that speeds up the reaction but it is not used up ** Markovnikov’s Rule states that if the alkene is unsymmetrical, the Hydrogen atoms will be attached to the carbon atom with most hydrogen atoms. ...
... ** catalyst a reactant that speeds up the reaction but it is not used up ** Markovnikov’s Rule states that if the alkene is unsymmetrical, the Hydrogen atoms will be attached to the carbon atom with most hydrogen atoms. ...
Aldehydes and Ketones Both contain the functional group C O
... Of the two reducing agents, sodium borohydride is the milder reagent and is the one of preference for aldehydes and ketones since it is specific for these two functional groups. Lithium aluminum hydride will reduce many types of compounds very quickly. Reduction to hydrocarbons (we saw this used to ...
... Of the two reducing agents, sodium borohydride is the milder reagent and is the one of preference for aldehydes and ketones since it is specific for these two functional groups. Lithium aluminum hydride will reduce many types of compounds very quickly. Reduction to hydrocarbons (we saw this used to ...
Name
... 1. Two compounds with the same formula but different structures are called what? (1) Isomers 2. Organic compounds (1) A) always contain nitrogen. B) are synthesized by only animal cells. C) always contain carbon. D) can only be synthesized in a laboratory. E) always contain oxygen. ...
... 1. Two compounds with the same formula but different structures are called what? (1) Isomers 2. Organic compounds (1) A) always contain nitrogen. B) are synthesized by only animal cells. C) always contain carbon. D) can only be synthesized in a laboratory. E) always contain oxygen. ...
Chapter 17 - Ellis Benjamin
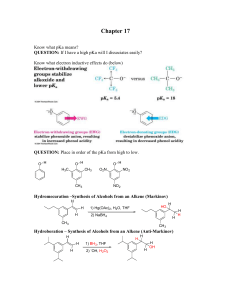
... Chapter 17 Know what pKa means? QUESTION: If I have a high pKa will I dissociates easily? Know what electron inductive effects do (below) ...
... Chapter 17 Know what pKa means? QUESTION: If I have a high pKa will I dissociates easily? Know what electron inductive effects do (below) ...
CHAPTER-6 DEHYDROHALOGENATION OF ALKYL HALIDES
... Dehydration of Alcohols to form Ethers • Simple, symmetrical ethers can be formed from the intermolecular acid‐catalyzed dehydration of 1° (or methyl) alcohols (a “substitution reaction”) • 2° and 3° alcohols can’t be used because they eliminate (intramolecular dehydration) to form alkenes ...
... Dehydration of Alcohols to form Ethers • Simple, symmetrical ethers can be formed from the intermolecular acid‐catalyzed dehydration of 1° (or methyl) alcohols (a “substitution reaction”) • 2° and 3° alcohols can’t be used because they eliminate (intramolecular dehydration) to form alkenes ...
Slide 1
... was reported by Friedländer in 1882. Friedländer reactions are generally carried out either by refluxing an aqueous or alcoholic solution of reactants in the presence of base or by heating a mixture of the reactants at high temperatures ranging from 150-220°C in the absence of catalyst.(1) ...
... was reported by Friedländer in 1882. Friedländer reactions are generally carried out either by refluxing an aqueous or alcoholic solution of reactants in the presence of base or by heating a mixture of the reactants at high temperatures ranging from 150-220°C in the absence of catalyst.(1) ...
Lab 9 - Academic Computer Center
... The overall reduction of a carbonyl group to a hydroxyl group involves the addition of two H atoms. The first H atom comes from a hydride, H-, of NaBH4. The second comes from the workup of the reaction, which is normally conducted in aqueous acid. Sodium borohydride, NaBH4, is the mildest of the th ...
... The overall reduction of a carbonyl group to a hydroxyl group involves the addition of two H atoms. The first H atom comes from a hydride, H-, of NaBH4. The second comes from the workup of the reaction, which is normally conducted in aqueous acid. Sodium borohydride, NaBH4, is the mildest of the th ...
Solid phase reactions II
... introduction of a suitable leaving group instead of the OH group prior to the reaction with an amine two principle ways: (1) replacement of the OH group (e.g. acid halogenides) (2) transformation into a suitalbe leaving group (e.g. active esters, anhydrides) best conditions highly dependent on the s ...
... introduction of a suitable leaving group instead of the OH group prior to the reaction with an amine two principle ways: (1) replacement of the OH group (e.g. acid halogenides) (2) transformation into a suitalbe leaving group (e.g. active esters, anhydrides) best conditions highly dependent on the s ...
Nucleophilic Addition to Carbonyl Groups
... The reverse of acetal formation if acetal hydrolysis. This is achieved by excess water in the presence of an acid catalyst. ...
... The reverse of acetal formation if acetal hydrolysis. This is achieved by excess water in the presence of an acid catalyst. ...
Jeopardy
... product, but resulting in formation of another radical Step 3 – termination – two radicals combine to make product, removing radicals from reaction vessel ...
... product, but resulting in formation of another radical Step 3 – termination – two radicals combine to make product, removing radicals from reaction vessel ...
Q 1: Molecular formula of BHA is
... ( A) In alcohols - OH group always present at the end of the chain. ( B) Alcohols may contain one or more groups. ( C) butane-1-ol has higher boiling point than 2-methyl ...
... ( A) In alcohols - OH group always present at the end of the chain. ( B) Alcohols may contain one or more groups. ( C) butane-1-ol has higher boiling point than 2-methyl ...
C1_5_products_from_oils_crossword
... 11. Polymers that change in response to changes in their environment. 12. A hydrocarbon whose molecules contain at least one carbon-carbon double bond. Down 1. Something that cannot be replaced once it is used up. 2. An alkene with the formula C2H4. 3. The reaction used in the oil industry to break ...
... 11. Polymers that change in response to changes in their environment. 12. A hydrocarbon whose molecules contain at least one carbon-carbon double bond. Down 1. Something that cannot be replaced once it is used up. 2. An alkene with the formula C2H4. 3. The reaction used in the oil industry to break ...
Final Exam Review Sheet Chemistry 110a/1998
... The final exam questions will seek an integrated understanding of the material found in chapters 1-13. You will be allowed to use the following when working your final exam: a calculator, molecular models, 13 pieces of unlined white 8.5 x 11 inch paper on which you may hand-write any information to ...
... The final exam questions will seek an integrated understanding of the material found in chapters 1-13. You will be allowed to use the following when working your final exam: a calculator, molecular models, 13 pieces of unlined white 8.5 x 11 inch paper on which you may hand-write any information to ...
doc CHEM 222 Lab exam with Answers
... is obtained via insertion into the double bond. 5.__T___ Carbenes are very reactive towards nucleophiles because of an empty orbital on the carbene carbon. 6.__F___ An emulsion is essential in the Carbene lab in order to increase the temperature and thus maximize the yield. 7.__ T__Biphenyl is solub ...
... is obtained via insertion into the double bond. 5.__T___ Carbenes are very reactive towards nucleophiles because of an empty orbital on the carbene carbon. 6.__F___ An emulsion is essential in the Carbene lab in order to increase the temperature and thus maximize the yield. 7.__ T__Biphenyl is solub ...
Unit 4_Carbonyl and carboxylic acid questions
... 4. Explain in terms of bonding why carbonyls have higher b.ps than corresponding alkanes but lower b.ps than corresponding alcohols. ...
... 4. Explain in terms of bonding why carbonyls have higher b.ps than corresponding alkanes but lower b.ps than corresponding alcohols. ...
Sources of hydride ion
... Reduction of esters with h DIBALH -- DIBALH is less reactive than LiAlH4 ...
... Reduction of esters with h DIBALH -- DIBALH is less reactive than LiAlH4 ...
Document
... • R groups are inductively donating and reduce delta positive charge on C of C=O. Ketones have two R groups and so are less reactive with nucleophiles than aldehydes. • Nucleophiles react with carbonyls as shown below – they attack at delta positive C of C=O breaking pi bond and generating intermedi ...
... • R groups are inductively donating and reduce delta positive charge on C of C=O. Ketones have two R groups and so are less reactive with nucleophiles than aldehydes. • Nucleophiles react with carbonyls as shown below – they attack at delta positive C of C=O breaking pi bond and generating intermedi ...
Assignment 2 Group A and B
... 9) Which of the following alcohols can be prepared by the reaction of methyl formate with excess Grignard reagent? A) 1-pentanol B) 2-pentanol C) 3-pentanol D) 2-methyl-2-pentanol E) 3-methyl-3-pentanol 10) What reagent(s) would you use to accomplish the following conversion? ...
... 9) Which of the following alcohols can be prepared by the reaction of methyl formate with excess Grignard reagent? A) 1-pentanol B) 2-pentanol C) 3-pentanol D) 2-methyl-2-pentanol E) 3-methyl-3-pentanol 10) What reagent(s) would you use to accomplish the following conversion? ...
CHEM 201 Name Quiz 10 (Ch 17) ID Q1. Which of the following
... the ester shown below with LiAlH4? O O ...
... the ester shown below with LiAlH4? O O ...
Preface - Wiley Online Library
... unusual class of reactions that involve the loss of small molecular fragments from a ring in service of uniting separate carbon atoms in that ring to form alkenes. These so called “extrusion reactions” are more commonly known for the loss of a single molecular fragment as in, e.g. the Ramberg-B¨ackl ...
... unusual class of reactions that involve the loss of small molecular fragments from a ring in service of uniting separate carbon atoms in that ring to form alkenes. These so called “extrusion reactions” are more commonly known for the loss of a single molecular fragment as in, e.g. the Ramberg-B¨ackl ...
10.5 Carbonyl Compounds (a) describe: (i) the
... (i) the formation of aldehydes and ketones from primary and secondary alcohols respectively using Cr2O72-/H+ (ii) the reduction of aldehydes and ketones e.g. using NaBH4 (b) describe the mechanism of nucleophilic addition reactions of hydrogen cyanide with aldehydes and ketones (c) describe the use ...
... (i) the formation of aldehydes and ketones from primary and secondary alcohols respectively using Cr2O72-/H+ (ii) the reduction of aldehydes and ketones e.g. using NaBH4 (b) describe the mechanism of nucleophilic addition reactions of hydrogen cyanide with aldehydes and ketones (c) describe the use ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.