purdue university - IUPUI ScholarWorks
... Three parts consist of my thesis work centered on the synthesis of inorganic phosphates and then metal organic frame work (MOF). The first part is the synthesis of mesoporous chromium phosphates using the room temperature solid state reaction (SSR) approach. One of the major aims of this work is to ...
... Three parts consist of my thesis work centered on the synthesis of inorganic phosphates and then metal organic frame work (MOF). The first part is the synthesis of mesoporous chromium phosphates using the room temperature solid state reaction (SSR) approach. One of the major aims of this work is to ...
PREPARATION OF ALDEHYDES
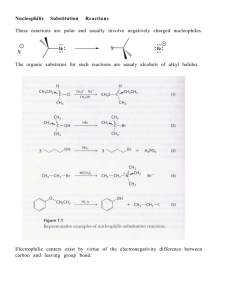
... EXAMPLES OF NUCLEOPHILIC ADDITION TO ALDEHYDES & KETONES Addition of HCN (neutral-basic conditions). CN Ө is a very good nucleophile (ionic nucleophile). The use of the actual compound HCN is not experimentally feasible, as it is a lethal gas, bp 26 oC. Addition of the elements of HCN to a C=O grou ...
... EXAMPLES OF NUCLEOPHILIC ADDITION TO ALDEHYDES & KETONES Addition of HCN (neutral-basic conditions). CN Ө is a very good nucleophile (ionic nucleophile). The use of the actual compound HCN is not experimentally feasible, as it is a lethal gas, bp 26 oC. Addition of the elements of HCN to a C=O grou ...
Chapter - FIU Faculty Websites
... Sometimes, acyl groups must be named as substituents. The three most common acyl groups are shown below: ...
... Sometimes, acyl groups must be named as substituents. The three most common acyl groups are shown below: ...
Carbonyl Condensation Reactions
... 24.3 Directed Aldol Reactions A directed aldol reaction is a variation of the crossed aldol reaction that clearly defines which carbonyl compound becomes the nucleophilic enolate and which reacts at the electrophilic carbonyl carbon. The strategy of a directed aldol reaction is as follows: [1] Prepa ...
... 24.3 Directed Aldol Reactions A directed aldol reaction is a variation of the crossed aldol reaction that clearly defines which carbonyl compound becomes the nucleophilic enolate and which reacts at the electrophilic carbonyl carbon. The strategy of a directed aldol reaction is as follows: [1] Prepa ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... electron-rich arenes, such as 1,4-dimethoxybenzene (2b), 1,3,5trimethoxybenzene (2c), and anisole (2d) furnished the desired products in excellent yields. It is worthwhile to note that the use of these nucleophiles provides an efficient route to methoxy-substituted unsymmetrical diarylmethanes which c ...
... electron-rich arenes, such as 1,4-dimethoxybenzene (2b), 1,3,5trimethoxybenzene (2c), and anisole (2d) furnished the desired products in excellent yields. It is worthwhile to note that the use of these nucleophiles provides an efficient route to methoxy-substituted unsymmetrical diarylmethanes which c ...
Preparation and Physical Properties of Chitosan Benzoic Acid
... Chitin, the second abundant natural polymer in Nature, and chitosan, a partially deacetylated form of chitin, have recently received much of attention owing to their applicability in wide range of fields such as pharmaceuticals, cosmetics, agriculture, foods, and material sciences [1–5]. Their film ...
... Chitin, the second abundant natural polymer in Nature, and chitosan, a partially deacetylated form of chitin, have recently received much of attention owing to their applicability in wide range of fields such as pharmaceuticals, cosmetics, agriculture, foods, and material sciences [1–5]. Their film ...
Zn(BH4)2/Al2O3: A new synthetic method for the efficient
... reduction reactions were performed efficiently in short reaction times (20–40 min) (92–95 %) (Table V). Under different conditions, attempts to reduce α-diketones into acyloins were unsatisfactory and only vicinal diols were identified as the sole products. In addition, reduction of acyloins to vici ...
... reduction reactions were performed efficiently in short reaction times (20–40 min) (92–95 %) (Table V). Under different conditions, attempts to reduce α-diketones into acyloins were unsatisfactory and only vicinal diols were identified as the sole products. In addition, reduction of acyloins to vici ...
Chapter Seven PPT
... • Unsaturated Compounds (Less than Maximum H Atoms) • Alkenes also Referred to as Olefins • Properties Similar to those of Corresponding Alkanes • Slightly Soluble in Water • Dissolve Readily in Nonpolar or Low Polarity Solvents • Densities of Alkenes and Alkynes Less than Water ...
... • Unsaturated Compounds (Less than Maximum H Atoms) • Alkenes also Referred to as Olefins • Properties Similar to those of Corresponding Alkanes • Slightly Soluble in Water • Dissolve Readily in Nonpolar or Low Polarity Solvents • Densities of Alkenes and Alkynes Less than Water ...
Part I Carbohydrate Auxiliaries - Wiley-VCH
... (modified Strecker reaction) are fundamental carbon–carbon bond-forming processes [3], which are efficient methods for preparing α-amino acids (Scheme 1.1). In 1987 Kunz and coworkers first reported pivaloyl protected d-galactosyl amine 3 as a very useful tool for asymmetric aminonitrile syntheses [4]. ...
... (modified Strecker reaction) are fundamental carbon–carbon bond-forming processes [3], which are efficient methods for preparing α-amino acids (Scheme 1.1). In 1987 Kunz and coworkers first reported pivaloyl protected d-galactosyl amine 3 as a very useful tool for asymmetric aminonitrile syntheses [4]. ...
幻灯片 1
... 1846.His ability to work in laboratory was hampered by a childhood injury that caused the loss of an arm. From 1849,utill 1887, he was professor of Chemistry at University College, London. ...
... 1846.His ability to work in laboratory was hampered by a childhood injury that caused the loss of an arm. From 1849,utill 1887, he was professor of Chemistry at University College, London. ...
Chemistry 360 - Athabasca University
... Should you stopper the test tubes prior to heating the tubes in the Ethanolic Silver Nitrate and Sodium ...
... Should you stopper the test tubes prior to heating the tubes in the Ethanolic Silver Nitrate and Sodium ...
Document
... dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one stereoisomer is formed preferentially. Why? ...
... dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one stereoisomer is formed preferentially. Why? ...
Document
... dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one stereoisomer is formed preferentially. Why? ...
... dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one stereoisomer is formed preferentially. Why? ...
Kinetic Modeling Of Methanol Synthesis From Carbon Monoxide
... in POLYMATH. Another aim was to use the results from the best fit model to explain some aspects and resolve some questions related to methanol synthesis kinetics. Two statistically sound kinetic models were chosen from literature based on their goodness of fit to the respective kinetic data. POLYMAT ...
... in POLYMATH. Another aim was to use the results from the best fit model to explain some aspects and resolve some questions related to methanol synthesis kinetics. Two statistically sound kinetic models were chosen from literature based on their goodness of fit to the respective kinetic data. POLYMAT ...
1 THE BARTON-McCOMBIE REACTION STUART W. McCOMBIE 28
... (2S and 2R)-2-Deuterio-3',5'-O-(1,1,3,3-tetraisopropyldisilox-1,3-diyl)uridine [Deoxygenation of a Secondary O-(N-Phenylthiocarbamoyl) Derivative with Bu3SnD] 1,2-Diphenyl-1-methoxyethane [Deoxygenation of a Secondary O-(Phenoxythiocarbonyl) Derivative with Poly(methylhydrosiloxane) and Catalytic (B ...
... (2S and 2R)-2-Deuterio-3',5'-O-(1,1,3,3-tetraisopropyldisilox-1,3-diyl)uridine [Deoxygenation of a Secondary O-(N-Phenylthiocarbamoyl) Derivative with Bu3SnD] 1,2-Diphenyl-1-methoxyethane [Deoxygenation of a Secondary O-(Phenoxythiocarbonyl) Derivative with Poly(methylhydrosiloxane) and Catalytic (B ...
Chapter Seven - U of L Class Index
... H Carbocation rearrangments are often promoted by the presence of Lewis Acids. In this case, the intermediates are said to be “carbocation-like” if not carbocations. ...
... H Carbocation rearrangments are often promoted by the presence of Lewis Acids. In this case, the intermediates are said to be “carbocation-like” if not carbocations. ...
Discodermolide

(+)-Discodermolide is a polyketide natural product found to stabilize microtubule. (+)-discodermolide was isolated by Gunasekera and his co-workers at the Harbor Branch Oceanographic Institute from the deep-sea sponge Discodermia dissoluta in 1990. (+)-Discodermolide was found to be a potent inhibitor of tumor cell growth in several MDR cancer cell lines. (+)-discodermolide also shows some unique characters, including a linear backbone structure, immunosuppressive properties both in vitro and in vivo, potent induction of an accelerated senescence phenotype, and synergistic antiproliferative activity in combination with paclitaxel. Discodermolide was recognized as one of the most potent natural promoters of tubulin assembly. A large number of efforts toward the total synthesis of (+)-discodermolide were directed by its interesting biological activities and extreme scarcity of natural sources (0.002% w/w from frozen marine sponge). The compound supply necessary for complete clinical trials cannot be met by harvesting, isolation, and purification. As of 2005, attempts at synthesis or semi-synthesis by fermentation have proven unsuccessful. As a result, all discodermolide used in preclinical studies and clinical trials has come from large-scale total synthesis.