The First Chiral Organometallic Triangle for Asymmetric Catalysis
... exhibit expected ν(CtC) stretches at ∼2100 cm-1. The formulations of 1-4 are also supported by microanalysis results. All of these spectroscopic data are consistent with a cyclic trinuclear structure with approximate D3 symmetry for 1-4. Numerous attempts have however failed to produce X-ray diffrac ...
... exhibit expected ν(CtC) stretches at ∼2100 cm-1. The formulations of 1-4 are also supported by microanalysis results. All of these spectroscopic data are consistent with a cyclic trinuclear structure with approximate D3 symmetry for 1-4. Numerous attempts have however failed to produce X-ray diffrac ...
THE FRIEDEL-CRAFTS BENZYLATION OF ARENES AND THE
... First, I would like to thank my supervisor Prof. Dr. Jeffrey W. Bode for giving me the opportunity to work in his research group. After the group arrived from UPenn, I was one of his first new PhD students at ETH and I feel really fortunate that I could join the group during this exciting time. Jeff ...
... First, I would like to thank my supervisor Prof. Dr. Jeffrey W. Bode for giving me the opportunity to work in his research group. After the group arrived from UPenn, I was one of his first new PhD students at ETH and I feel really fortunate that I could join the group during this exciting time. Jeff ...
Alkyl Halides SN and E reactions
... 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 is faster. In 1° and Me° alkyl halides, SN2 occurs. 3. Weakly basic, weak nucleophiles, like H2O, EtOH, CH3COOH, etc., cannot react unless a C+ forms. This only occurs ...
... 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 is faster. In 1° and Me° alkyl halides, SN2 occurs. 3. Weakly basic, weak nucleophiles, like H2O, EtOH, CH3COOH, etc., cannot react unless a C+ forms. This only occurs ...
Reactions of Alkyl Halides (SN1, SN2, E1, and E2 reactions)
... 1. Non basic, good nucleophiles, like Br- and I- will cause substitution not elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucle ...
... 1. Non basic, good nucleophiles, like Br- and I- will cause substitution not elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucle ...
Reactions of Alkyl Halides (SN1, SN2, E1, and E2 reactions)
... 1. Non basic, good nucleophiles, like Br- and I- will cause substitution not elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucle ...
... 1. Non basic, good nucleophiles, like Br- and I- will cause substitution not elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucle ...
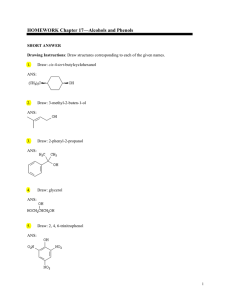
HOMEWORK Chapter 17—Alcohols and Phenols
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
PowerPoint 演示文稿
... conformations determined from electron diffraction studies by the Norwegian physical chemist Odd Hassel. Thus conformational analysis, which ever after changed the way organic chemists think about structure, synthesis and chemical reactivity, came into being. Barton and Hassel shared the 1969 Nobel ...
... conformations determined from electron diffraction studies by the Norwegian physical chemist Odd Hassel. Thus conformational analysis, which ever after changed the way organic chemists think about structure, synthesis and chemical reactivity, came into being. Barton and Hassel shared the 1969 Nobel ...
Forward
... The carbonyl carbon of a ketone bears two electron-releasing alkyl groups; an aldehyde carbonyl group has only one. Just as a disubstituted double bond in an alkene is more stable than a monosubstituted double bond, a ketone carbonyl is more stable than an aldehyde carbonyl. We’ll see later in this ...
... The carbonyl carbon of a ketone bears two electron-releasing alkyl groups; an aldehyde carbonyl group has only one. Just as a disubstituted double bond in an alkene is more stable than a monosubstituted double bond, a ketone carbonyl is more stable than an aldehyde carbonyl. We’ll see later in this ...
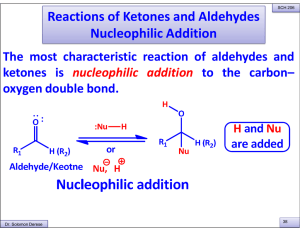
Reactions of Ketones and Aldehydes Nucleophilic Addition
... I. Addition of the organometalics to formaldehyde (CH2=O) forms a 1° alcohol. II. Addition of the organometalics to all other aldehydes forms a 2° alcohol. III.Addition of the organometalics to ketones forms a 3° alcohol. Dr. Solomon Derese ...
... I. Addition of the organometalics to formaldehyde (CH2=O) forms a 1° alcohol. II. Addition of the organometalics to all other aldehydes forms a 2° alcohol. III.Addition of the organometalics to ketones forms a 3° alcohol. Dr. Solomon Derese ...
Chem E2b - Organic Chemistry II What is Organic Chemistry?
... Cl behaves like MeO for directing substitution (resonance) Cl behaves slightly like NO2 for activation (induction) As with acidity, resonance is more important than induction! ...
... Cl behaves like MeO for directing substitution (resonance) Cl behaves slightly like NO2 for activation (induction) As with acidity, resonance is more important than induction! ...
Organic Chemistry II with Dr Roche
... Normal ethers do not undergo nucleophilic substitution or eliminations because the alkoxide anion is not a good leaving group. (That is why acid catalysis is required). Epoxides are different though. The release of strain when an epoxide is opened more than compensates for the poor leaving group abi ...
... Normal ethers do not undergo nucleophilic substitution or eliminations because the alkoxide anion is not a good leaving group. (That is why acid catalysis is required). Epoxides are different though. The release of strain when an epoxide is opened more than compensates for the poor leaving group abi ...
Iridium(III) and Rhodium(III) compounds of dipyridyl-N
... such as in hydrogenation,6 hydrosilytion,7 amination,8 C-C coupling9,10 and Diels-Alder reactions.11 Furthermore, compounds of this classes exhibited anticancer12 14 and DNA intercalative properties.15,16 Owing to their wide applications, synthesis of iridium(III) and rhodium(III) complexes bearing ...
... such as in hydrogenation,6 hydrosilytion,7 amination,8 C-C coupling9,10 and Diels-Alder reactions.11 Furthermore, compounds of this classes exhibited anticancer12 14 and DNA intercalative properties.15,16 Owing to their wide applications, synthesis of iridium(III) and rhodium(III) complexes bearing ...
C 1 hapter
... most useful reactions in organic synthesis. It provides a way of transforming alkenes into many different types of C*-X and C*-R bonds through the optically enriched organoboron adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through ...
... most useful reactions in organic synthesis. It provides a way of transforming alkenes into many different types of C*-X and C*-R bonds through the optically enriched organoboron adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through ...
isomeria geometrica
... Stereocenters • Any atom at which the exchange of two groups yields a stereoisomer. • Examples: • Asymmetric carbons • Double-bonded carbons in cis-trans isomers ...
... Stereocenters • Any atom at which the exchange of two groups yields a stereoisomer. • Examples: • Asymmetric carbons • Double-bonded carbons in cis-trans isomers ...
Chiral Enolate Equivalents
... Cyanide has served as a formyl anion equivalent for many years. Its reaction with aldehydes and imines (or oxonium and iminium ions), catalyzed by Brønsted or Lewis acids or bases, lead to α-amino or α-hydroxy nitriles, which can be hydrolyzed to the corresponding carboxylic acid. Included in this c ...
... Cyanide has served as a formyl anion equivalent for many years. Its reaction with aldehydes and imines (or oxonium and iminium ions), catalyzed by Brønsted or Lewis acids or bases, lead to α-amino or α-hydroxy nitriles, which can be hydrolyzed to the corresponding carboxylic acid. Included in this c ...
Postprint
... [IrCl(CO)(PPh3)2] (Figure 1.a), a catalyst of the [L3MX]-type .4 Following parallel development with other platinum group metals, Shrock and Osborn (Figure 1.d) discovered that an [L4Rh]+X- complex could convert in situ to an [L2Rh]+X- active catalyst. Overall having a 2:1 mono-dentate ligand/metal ...
... [IrCl(CO)(PPh3)2] (Figure 1.a), a catalyst of the [L3MX]-type .4 Following parallel development with other platinum group metals, Shrock and Osborn (Figure 1.d) discovered that an [L4Rh]+X- complex could convert in situ to an [L2Rh]+X- active catalyst. Overall having a 2:1 mono-dentate ligand/metal ...
High-Oxidation-State Palladium Catalysis: New Reactivity for
... sequence.[11] A proposed s-norbornyl PdIV intermediate is central to the overall success of the reaction. Its existence was not proven outright, but derived from the isolation of phenanthroline-stabilized allyl and benzyl model compounds, such as A and B.[12] However, some care must be exercised in ...
... sequence.[11] A proposed s-norbornyl PdIV intermediate is central to the overall success of the reaction. Its existence was not proven outright, but derived from the isolation of phenanthroline-stabilized allyl and benzyl model compounds, such as A and B.[12] However, some care must be exercised in ...
Recent developments in the synthesis of functional poly(olefin)s
... melt phase free radical polymerization [9±15]. The ability of epoxy group present in glycidyl methacrylate to react with various other functional groups like carboxyl, hydroxyl, anhydride, amine etc. renders such graft copolymers useful as compatibilizer. Melt phase free radical grafting is usually ...
... melt phase free radical polymerization [9±15]. The ability of epoxy group present in glycidyl methacrylate to react with various other functional groups like carboxyl, hydroxyl, anhydride, amine etc. renders such graft copolymers useful as compatibilizer. Melt phase free radical grafting is usually ...
Alkenes and Alkynes I
... ● Tertiary alcohols are usually so easily dehydrated that extremely mild conditions can be used. tert-Butyl alcohol, for example, dehydrates in 20% aqueous sulfuric acid at a temperature of 85°C ...
... ● Tertiary alcohols are usually so easily dehydrated that extremely mild conditions can be used. tert-Butyl alcohol, for example, dehydrates in 20% aqueous sulfuric acid at a temperature of 85°C ...
Proofs to - Research Explorer
... was confirmed by a triplet resonance in the 13C{1H} NMR spectrum at 321.5 ppm for the Cα carbene carbon, by the appearance of resonances at 70.3, 12.9 ppm and 43.7 ppm consistent with the OEt and Me substitutents respectively of the ethoxycarbene ligand and by a characteristic low field shift in the ...
... was confirmed by a triplet resonance in the 13C{1H} NMR spectrum at 321.5 ppm for the Cα carbene carbon, by the appearance of resonances at 70.3, 12.9 ppm and 43.7 ppm consistent with the OEt and Me substitutents respectively of the ethoxycarbene ligand and by a characteristic low field shift in the ...
NAME Chem 204 Spring, 1990 Final Exam FINAL EXAM: Chaps. 1
... alkyl chloride, there is NO one reaction that will convert a chiral alcohol into its alkyl chloride with "retained" configuration. Thus, it is necessary to use several reaction steps, each of 100% inversion or 100% retention of configuration, in order to convert (X) into (XI). Outlines below a serie ...
... alkyl chloride, there is NO one reaction that will convert a chiral alcohol into its alkyl chloride with "retained" configuration. Thus, it is necessary to use several reaction steps, each of 100% inversion or 100% retention of configuration, in order to convert (X) into (XI). Outlines below a serie ...
Discodermolide

(+)-Discodermolide is a polyketide natural product found to stabilize microtubule. (+)-discodermolide was isolated by Gunasekera and his co-workers at the Harbor Branch Oceanographic Institute from the deep-sea sponge Discodermia dissoluta in 1990. (+)-Discodermolide was found to be a potent inhibitor of tumor cell growth in several MDR cancer cell lines. (+)-discodermolide also shows some unique characters, including a linear backbone structure, immunosuppressive properties both in vitro and in vivo, potent induction of an accelerated senescence phenotype, and synergistic antiproliferative activity in combination with paclitaxel. Discodermolide was recognized as one of the most potent natural promoters of tubulin assembly. A large number of efforts toward the total synthesis of (+)-discodermolide were directed by its interesting biological activities and extreme scarcity of natural sources (0.002% w/w from frozen marine sponge). The compound supply necessary for complete clinical trials cannot be met by harvesting, isolation, and purification. As of 2005, attempts at synthesis or semi-synthesis by fermentation have proven unsuccessful. As a result, all discodermolide used in preclinical studies and clinical trials has come from large-scale total synthesis.