Chapter Seven - U of L Class Index
... H Carbocation rearrangments are often promoted by the presence of Lewis Acids. In this case, the intermediates are said to be “carbocation-like” if not carbocations. ...
... H Carbocation rearrangments are often promoted by the presence of Lewis Acids. In this case, the intermediates are said to be “carbocation-like” if not carbocations. ...
1. Alcohol Oxidations
... Oxidation of 1° Alcohols to Aldehydes [Cu(II)/O2] Marko, I. E.; Giles, P. R.; Tsukazaki, M.; Brown, S. M.; Urch, C. J. Science 1996, 274, 2044. Oxidation proceeds with O2 in the presence of a catalytic amount (5 mol%) of a copper complex, which can be formed in situ from CuCl2, phenanthroline, and a ...
... Oxidation of 1° Alcohols to Aldehydes [Cu(II)/O2] Marko, I. E.; Giles, P. R.; Tsukazaki, M.; Brown, S. M.; Urch, C. J. Science 1996, 274, 2044. Oxidation proceeds with O2 in the presence of a catalytic amount (5 mol%) of a copper complex, which can be formed in situ from CuCl2, phenanthroline, and a ...
Preliminary Screening — Technical and Economic Assessment of Synthesis Gas
... production operation in a gas-to-liquids plant amounts to greater than half of the capital cost of the plant. The choice of technology for syngas production also depends on the scale of the synthesis operation. Syngas production from solid fuels can require an even greater capital investment with th ...
... production operation in a gas-to-liquids plant amounts to greater than half of the capital cost of the plant. The choice of technology for syngas production also depends on the scale of the synthesis operation. Syngas production from solid fuels can require an even greater capital investment with th ...
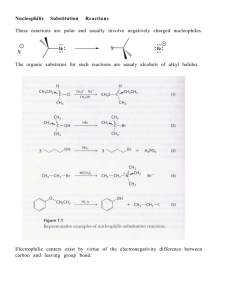
13-Elimination Reactions
... reaction mechanisms and compares them with the stereochemistry of the SN1 and SN2 reaction mechanisms, which are covered in Chapter 12. The E1 reaction mechanism is a two-step process that, as with the SN1 mechanism, usually loses all the stereochemical information of the substrate as the reaction p ...
... reaction mechanisms and compares them with the stereochemistry of the SN1 and SN2 reaction mechanisms, which are covered in Chapter 12. The E1 reaction mechanism is a two-step process that, as with the SN1 mechanism, usually loses all the stereochemical information of the substrate as the reaction p ...
Oxo-molybdenum(VI) - Repositório da Universidade Nova de Lisboa
... triggers its capability to catalyze different kind of processes such as carbon-carbon bond formation, olefin metathesis or alkene epoxidation among the most relevant transformations. ...
... triggers its capability to catalyze different kind of processes such as carbon-carbon bond formation, olefin metathesis or alkene epoxidation among the most relevant transformations. ...
Retrosynthetic Analysis of Nabumetone
... Grignard reactions that produce secondary alcohols, will require oxidation to the corresponding ketone to arrive at the desired product. Starting with the bond between the ring and the side chain, we are given two pairs of synthons as shown in Scheme 2. The synthetic equivalent to the aromatic anion ...
... Grignard reactions that produce secondary alcohols, will require oxidation to the corresponding ketone to arrive at the desired product. Starting with the bond between the ring and the side chain, we are given two pairs of synthons as shown in Scheme 2. The synthetic equivalent to the aromatic anion ...
Lecture - Ch 16
... – Substitution between two groups is rare when they are in a meta-disubstituted compound as the site is too hindered • An alternate route must be taken in the preparation of aromatic rings with three adjacent substituents ...
... – Substitution between two groups is rare when they are in a meta-disubstituted compound as the site is too hindered • An alternate route must be taken in the preparation of aromatic rings with three adjacent substituents ...
Alcohols
... Oxidation of Alcohols and Phenols Oxidations to alcohols and phenols occur by a mechanism that is closely related to the E2 reaction • The Dess-Martin oxidation involves a substitution reaction between the alcohol and the I(V) reagent to form a new periodinane intermediate followed by expulsion of ...
... Oxidation of Alcohols and Phenols Oxidations to alcohols and phenols occur by a mechanism that is closely related to the E2 reaction • The Dess-Martin oxidation involves a substitution reaction between the alcohol and the I(V) reagent to form a new periodinane intermediate followed by expulsion of ...
aldehydes and ketones
... magnitude of the positive charge on the carbonyl carbon atom. Hence, any substituent that increases the positive charge on the carbonyl carbon must increase its reactivity towards addition reactions. The introduction of negative group ( -I effect) increases the reactivity, while introduction of alky ...
... magnitude of the positive charge on the carbonyl carbon atom. Hence, any substituent that increases the positive charge on the carbonyl carbon must increase its reactivity towards addition reactions. The introduction of negative group ( -I effect) increases the reactivity, while introduction of alky ...
RUMPLE-DISSERTATION-2014 - SMARTech Home
... advice and mentorship. Her advice and vision have proven instrumental in my development as a chemist, and she has really gone above and beyond in her efforts to help and guide us. To all my colleagues, it’s been a pleasure working with you and joking with you. We’ve all become good friends over the ...
... advice and mentorship. Her advice and vision have proven instrumental in my development as a chemist, and she has really gone above and beyond in her efforts to help and guide us. To all my colleagues, it’s been a pleasure working with you and joking with you. We’ve all become good friends over the ...
synthetic.applicatio..
... ters.22,23 Chiral non-racemic α-methylcysteine (S)-24 was prepared by this method (Scheme 6).24 The introduction of oxygen functionality β to the carboxylate group, as present in substituted serine derivatives, was achieved using acid promoted ring opening. When chiral aziridines (SS,2S,3S)-25 and ( ...
... ters.22,23 Chiral non-racemic α-methylcysteine (S)-24 was prepared by this method (Scheme 6).24 The introduction of oxygen functionality β to the carboxylate group, as present in substituted serine derivatives, was achieved using acid promoted ring opening. When chiral aziridines (SS,2S,3S)-25 and ( ...
Amines
... to -CH2Amides can be reduced by LiAlH4 but NOT the less reactive NaBH4 Typical reagents : LiAlH4 / ether solvent, followed by aqueous work-up. Note that this reaction is different to that of other C=O compounds which reduce to alcohols (for example esters) The nature of the amine obtained depends on ...
... to -CH2Amides can be reduced by LiAlH4 but NOT the less reactive NaBH4 Typical reagents : LiAlH4 / ether solvent, followed by aqueous work-up. Note that this reaction is different to that of other C=O compounds which reduce to alcohols (for example esters) The nature of the amine obtained depends on ...
Synthesis of Inorganic Nanostructures in Reverse Micelles
... exchange.[25] As the reaction takes place in reverse micelles, more and more droplets could contain products and reactants simultaneously. The reaction inside nanodroplets can be catalyzed by the surface of an existing product particle, and a large particle has a greater probability of acting as a c ...
... exchange.[25] As the reaction takes place in reverse micelles, more and more droplets could contain products and reactants simultaneously. The reaction inside nanodroplets can be catalyzed by the surface of an existing product particle, and a large particle has a greater probability of acting as a c ...
10 Haloalkanes and Haloarenes
... Direct halogenation of an alkane is a chain reaction and follows free radical mechanism. ii. This method of preparation gives the mixture of mono, di, tri and tetra halogen derivatives of an alkane and it is difficult to separate each component in pure form. eg. Preparation of methyl chloride by dir ...
... Direct halogenation of an alkane is a chain reaction and follows free radical mechanism. ii. This method of preparation gives the mixture of mono, di, tri and tetra halogen derivatives of an alkane and it is difficult to separate each component in pure form. eg. Preparation of methyl chloride by dir ...
Organic Chemistry Fifth Edition
... The transition state that leads to 1-butene is less crowded than the one leading to cis or trans-2-butene. ...
... The transition state that leads to 1-butene is less crowded than the one leading to cis or trans-2-butene. ...
Discodermolide

(+)-Discodermolide is a polyketide natural product found to stabilize microtubule. (+)-discodermolide was isolated by Gunasekera and his co-workers at the Harbor Branch Oceanographic Institute from the deep-sea sponge Discodermia dissoluta in 1990. (+)-Discodermolide was found to be a potent inhibitor of tumor cell growth in several MDR cancer cell lines. (+)-discodermolide also shows some unique characters, including a linear backbone structure, immunosuppressive properties both in vitro and in vivo, potent induction of an accelerated senescence phenotype, and synergistic antiproliferative activity in combination with paclitaxel. Discodermolide was recognized as one of the most potent natural promoters of tubulin assembly. A large number of efforts toward the total synthesis of (+)-discodermolide were directed by its interesting biological activities and extreme scarcity of natural sources (0.002% w/w from frozen marine sponge). The compound supply necessary for complete clinical trials cannot be met by harvesting, isolation, and purification. As of 2005, attempts at synthesis or semi-synthesis by fermentation have proven unsuccessful. As a result, all discodermolide used in preclinical studies and clinical trials has come from large-scale total synthesis.