chapter 22 organic and biological molecules
... functional group. The monomer for a homopolymer polyester must have an alcohol functional group and a carboxylic acid functional group present in the structure. b. A polyamide forms when an amine functional group reacts with a carboxylic acid functional group. For a copolymer polyamide, one monomer ...
... functional group. The monomer for a homopolymer polyester must have an alcohol functional group and a carboxylic acid functional group present in the structure. b. A polyamide forms when an amine functional group reacts with a carboxylic acid functional group. For a copolymer polyamide, one monomer ...
New Stereoselective Approaches to Highly Substituted
... It was during the early 1990s that the Knight9,10 group commenced research in this field. In model studies towards the valerolactone moiety of Mevinic acids, the Knight group discovered that iodolactonisations o f 3-hydroxyalk-5-enoic acid derivatives 11, gave predominately the /ra«s-3,5-disubstitut ...
... It was during the early 1990s that the Knight9,10 group commenced research in this field. In model studies towards the valerolactone moiety of Mevinic acids, the Knight group discovered that iodolactonisations o f 3-hydroxyalk-5-enoic acid derivatives 11, gave predominately the /ra«s-3,5-disubstitut ...
Elimination Reactions
... neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 M or above. WWU -- Chemistry ...
... neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 M or above. WWU -- Chemistry ...
- University of Bath Opus
... being applied and the use of the mild Swern oxidation is present in many routes to pharmaceuticals. The activation of alcohols involves conversion into halides or sulfonates on scale, both of which require an extra step which generates more waste and results in poor atom economy. More importantly, m ...
... being applied and the use of the mild Swern oxidation is present in many routes to pharmaceuticals. The activation of alcohols involves conversion into halides or sulfonates on scale, both of which require an extra step which generates more waste and results in poor atom economy. More importantly, m ...
(omit), and Epoxides
... – In ethyl vinyl ether, for example, the ether oxygen is bonded to one sp3 hybridized carbon and one sp2 hybridized carbon. ...
... – In ethyl vinyl ether, for example, the ether oxygen is bonded to one sp3 hybridized carbon and one sp2 hybridized carbon. ...
CHAPTER 21 ORGANIC CHEMISTRY
... For cis-trans isomerism (geometric isomerism), you must have at least two carbons with restricted rotation (double bond or ring) that each have two different groups bonded to them. The cis isomer will generally have the largest groups bonded to the two carbons, with restricted rotation, on the same ...
... For cis-trans isomerism (geometric isomerism), you must have at least two carbons with restricted rotation (double bond or ring) that each have two different groups bonded to them. The cis isomer will generally have the largest groups bonded to the two carbons, with restricted rotation, on the same ...
Organic Chemistry/Fourth Edition: e-Text
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
Proofs to - Research Explorer
... The character of the iron to C-alpha bond in a series of alkylidenes and heteroatom (E) stabilised carbenes has been the focus of a series of theory investigations on complexes of the type [Fe{C(ER’x)R}L2Cp]+ [36,37]. There is a clear correlation between the extent of heteroatom stabilisation and t ...
... The character of the iron to C-alpha bond in a series of alkylidenes and heteroatom (E) stabilised carbenes has been the focus of a series of theory investigations on complexes of the type [Fe{C(ER’x)R}L2Cp]+ [36,37]. There is a clear correlation between the extent of heteroatom stabilisation and t ...
Chapter 6 Addition Reactions to Alkenes
... University who won the Nobel Prize in 1977 for this work. The reaction proceeds by means of a one-step, concerted addition of the boron hydride to the alkene. Hydrogen is more electronegative than boron, so the electrophile in this reaction is boron. Note also that boron only has six electrons, so i ...
... University who won the Nobel Prize in 1977 for this work. The reaction proceeds by means of a one-step, concerted addition of the boron hydride to the alkene. Hydrogen is more electronegative than boron, so the electrophile in this reaction is boron. Note also that boron only has six electrons, so i ...
Modern Synthetic Methods for Copper-Mediated C(aryl
... the mechanistic rationale for these precise reaction conditions, although the outcome is of considerable importance. Chan and co-workers[18] first described a collection of Nand O-arylation transformations under novel reaction conditions as early as in June 1997, but only part of the work was disclo ...
... the mechanistic rationale for these precise reaction conditions, although the outcome is of considerable importance. Chan and co-workers[18] first described a collection of Nand O-arylation transformations under novel reaction conditions as early as in June 1997, but only part of the work was disclo ...
CHAPTER 22 ORGANIC AND BIOLOGICAL MOLECULES 1
... Alkanes: general formula = CnH2n + 2; all carbons are sp3 hybridized; bond angles = 109.5 Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5 bond angles, but rings with three carbons or four carbons or five carbons are ...
... Alkanes: general formula = CnH2n + 2; all carbons are sp3 hybridized; bond angles = 109.5 Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5 bond angles, but rings with three carbons or four carbons or five carbons are ...
Complete Solution Manual
... Alkanes: general formula = CnH2n + 2; all carbons are sp3 hybridized; bond angles = 109.5 Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5 bond angles, but rings with three carbons or four carbons or five carbons are ...
... Alkanes: general formula = CnH2n + 2; all carbons are sp3 hybridized; bond angles = 109.5 Cyclic alkanes: general formula CnH2n (if only one ring is present in the compound); all carbons are sp3 hybridized; prefers 109.5 bond angles, but rings with three carbons or four carbons or five carbons are ...
Copper-Catalyzed Coupling Reactions Using Carbon
... References for Chapter 2 ....................................................................................... 26 ...
... References for Chapter 2 ....................................................................................... 26 ...
CHAPTER TWENTY-TWO ORGANIC AND BIOLOGICAL MOLECULES
... hydrocarbon has only carbon-carbon single bonds in the molecule. An unsaturated hydrocarbon has one or more carbon-carbon multiple bonds, but may also contain carbon-carbon single bonds. An alkane is a saturated hydrocarbon composed of only C−C and C−H single bonds. Each carbon in an alkane is bonde ...
... hydrocarbon has only carbon-carbon single bonds in the molecule. An unsaturated hydrocarbon has one or more carbon-carbon multiple bonds, but may also contain carbon-carbon single bonds. An alkane is a saturated hydrocarbon composed of only C−C and C−H single bonds. Each carbon in an alkane is bonde ...
Elimination Reactions
... • This tells us that the C-D or C-H bonds are not broken in the rate determining step (step 1). They are broken in the fast step (step 2) in the mechanism). ...
... • This tells us that the C-D or C-H bonds are not broken in the rate determining step (step 1). They are broken in the fast step (step 2) in the mechanism). ...
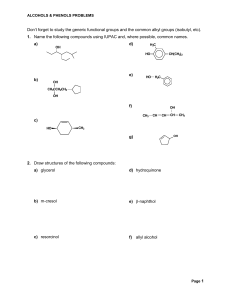
Don`t forget to study the generic functional groups and the common
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
Alcohols, Phenols and Ethers
... 44) As the molar mass of these alcohols increases, the water solubility decreases. This occurs because the polarity of the hydroxyl group, which is the reason for the interaction with the polar water molecules, becomes less important as the size of the nonpolar hydrocarbon portion of the molecule in ...
... 44) As the molar mass of these alcohols increases, the water solubility decreases. This occurs because the polarity of the hydroxyl group, which is the reason for the interaction with the polar water molecules, becomes less important as the size of the nonpolar hydrocarbon portion of the molecule in ...
Topic 22 Notes
... 2. Single and multiple covalent bonds a. Single covalent bonds (1) Where one pair of electrons is shared between two atoms. (2) Symbolized by one pair of dots or by a dash. b. Double covalent bonds (1) Where two pairs of electrons are shared between two atoms. (2) Symbolized by two pairs of dots or ...
... 2. Single and multiple covalent bonds a. Single covalent bonds (1) Where one pair of electrons is shared between two atoms. (2) Symbolized by one pair of dots or by a dash. b. Double covalent bonds (1) Where two pairs of electrons are shared between two atoms. (2) Symbolized by two pairs of dots or ...
Ch. 6 - Department of Chemistry and Biochemistry
... nucleophilic anions, and this results in “naked” anions of the Nu⊖ and makes the e⊖ pair of the Nu⊖ more available ...
... nucleophilic anions, and this results in “naked” anions of the Nu⊖ and makes the e⊖ pair of the Nu⊖ more available ...
lecture 12 catalysis_transformation of alkenes_alkynes
... two non-bonding electrons, necessary for oxidative coupling ...
... two non-bonding electrons, necessary for oxidative coupling ...
Document
... It is important to know what nucleophiles will add to carbonyl groups. • Cl¯, Br¯ and I¯ are good nucleophiles in substitution reactions at sp3 hybridized carbons, but they are ineffective nucleophiles in addition. • When these nucleophiles add to a carbonyl, they cleave the C—O bond, forming an a ...
... It is important to know what nucleophiles will add to carbonyl groups. • Cl¯, Br¯ and I¯ are good nucleophiles in substitution reactions at sp3 hybridized carbons, but they are ineffective nucleophiles in addition. • When these nucleophiles add to a carbonyl, they cleave the C—O bond, forming an a ...
Palladium(II)-Catalyzed Oxidative Cyclization Strategies Andreas K. Å. Persson
... character. Properties like these make the metal center less prone to interact with electronegative hard groups (such as alcohols). Instead it has a higher affinity towards “soft” σ- and π-donors. Consequently, alkenes, alkynes and allenes readily form π-complexes with palladium.21 ...
... character. Properties like these make the metal center less prone to interact with electronegative hard groups (such as alcohols). Instead it has a higher affinity towards “soft” σ- and π-donors. Consequently, alkenes, alkynes and allenes readily form π-complexes with palladium.21 ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.