9647 H2 Chemistry
... technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the teaching and learning of Chemistry. ...
... technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the teaching and learning of Chemistry. ...
Stoichiometry of Chemical Reactions
... Write a balanced equation for the decomposition of ammonium nitrate to form molecular nitrogen, molecular oxygen, and water. (Hint: Balance oxygen last, since it is present in more than one molecule on the right side of the equation.) Answer: 2NH 4 NO 3 ⟶ 2N 2 + O 2 + 4H 2 O ...
... Write a balanced equation for the decomposition of ammonium nitrate to form molecular nitrogen, molecular oxygen, and water. (Hint: Balance oxygen last, since it is present in more than one molecule on the right side of the equation.) Answer: 2NH 4 NO 3 ⟶ 2N 2 + O 2 + 4H 2 O ...
Stoichiometry of Chemical Reactions
... Write a balanced equation for the decomposition of ammonium nitrate to form molecular nitrogen, molecular oxygen, and water. (Hint: Balance oxygen last, since it is present in more than one molecule on the right side of the equation.) Answer: 2NH 4 NO 3 ⟶ 2N 2 + O 2 + 4H 2 O ...
... Write a balanced equation for the decomposition of ammonium nitrate to form molecular nitrogen, molecular oxygen, and water. (Hint: Balance oxygen last, since it is present in more than one molecule on the right side of the equation.) Answer: 2NH 4 NO 3 ⟶ 2N 2 + O 2 + 4H 2 O ...
Chains and Rings: Organic Chemistry
... or hundreds of carbon atoms bonded to each other occur in nature. This is because the C-C bond is very strong. 2. Carbon has a valency of 4, meaning that not only can carbon bonded together in a large chain, but other chemical atoms or groups of atoms may be bonded to the sides of these chains. The ...
... or hundreds of carbon atoms bonded to each other occur in nature. This is because the C-C bond is very strong. 2. Carbon has a valency of 4, meaning that not only can carbon bonded together in a large chain, but other chemical atoms or groups of atoms may be bonded to the sides of these chains. The ...
Synthesis and Characterisation of N
... I hereby certify that I am the sole author of this thesis and that no part of this thesis has been published or submitted for publication. I certify that, to the best of my knowledge, my thesis does not infringe upon anyone’s copyright nor violate any proprietary rights and that any ideas, technique ...
... I hereby certify that I am the sole author of this thesis and that no part of this thesis has been published or submitted for publication. I certify that, to the best of my knowledge, my thesis does not infringe upon anyone’s copyright nor violate any proprietary rights and that any ideas, technique ...
Chapter 16
... hydrocarbons. Adding an additional substituent, such as a methyl group, to a hydrocarbon slightly alters the melting and boiling point of the compound. For molecules also containing other elements, very small changes in structure can produce major changes in the properties. An interesting pair of ex ...
... hydrocarbons. Adding an additional substituent, such as a methyl group, to a hydrocarbon slightly alters the melting and boiling point of the compound. For molecules also containing other elements, very small changes in structure can produce major changes in the properties. An interesting pair of ex ...
Calculations on the equations reaction
... Determine reaction medium. 9. Which of the salt is hydrolyzed: LiBr; Na2CO3 Write the hydrolysis reaction in the molecular and ionic form. Determine the reaction medium. 10.Which of the salt is hydrolyzed: K2S; CaSO4 Write the hydrolysis reaction in the molecular and ionic form. Determine the reacti ...
... Determine reaction medium. 9. Which of the salt is hydrolyzed: LiBr; Na2CO3 Write the hydrolysis reaction in the molecular and ionic form. Determine the reaction medium. 10.Which of the salt is hydrolyzed: K2S; CaSO4 Write the hydrolysis reaction in the molecular and ionic form. Determine the reacti ...
Carboxylic acids, esters, and other acid derivatives
... alkoxy group instead of a hydroxyl group. ...
... alkoxy group instead of a hydroxyl group. ...
Unit 5 - Chemical Reactions - Student
... A catalyst is a substance that increases reaction rate without being used up by the reaction. If a catalyst is used, its formula is written above the arrow. In accordance with the law of conservation of mass, a chemical equation must be balanced. In balancing an equation, coefficients are used s ...
... A catalyst is a substance that increases reaction rate without being used up by the reaction. If a catalyst is used, its formula is written above the arrow. In accordance with the law of conservation of mass, a chemical equation must be balanced. In balancing an equation, coefficients are used s ...
Chemistry 209 - Experiment 3, Spring 2003
... Cover the tube with a cork or rubber sleeve stopper and shake it vigorously so as to mix the contents thoroughly after each addition of solvent. If the substance dissolves completely, record it as soluble. Continue shaking for at least five minutes if the substance does not dissolve at first in orde ...
... Cover the tube with a cork or rubber sleeve stopper and shake it vigorously so as to mix the contents thoroughly after each addition of solvent. If the substance dissolves completely, record it as soluble. Continue shaking for at least five minutes if the substance does not dissolve at first in orde ...
Chapter 4: Reaction Stoichiometry Reaction Stoichiometry
... 2) Determine how much 0.125 M sulfuric acid is needed (in mL) to react with 22.5 g of aluminum. 3) Determine how much of the elemental product is liberated in that reaction as well. ...
... 2) Determine how much 0.125 M sulfuric acid is needed (in mL) to react with 22.5 g of aluminum. 3) Determine how much of the elemental product is liberated in that reaction as well. ...
mass-mass problems.
... Na2O + 2CO2 + H2O → 2NaHCO3 If you start with 0.35 moles of CO2, how many grams of H2O are needed? ...
... Na2O + 2CO2 + H2O → 2NaHCO3 If you start with 0.35 moles of CO2, how many grams of H2O are needed? ...
LABORATORY MANUAL CHEMISTRY 121
... In aqueous solution the green complex trans-dichlorbis (ethylenediamine) cobalt(III) chloride dissociates into chloride ion and trans-[Co(en)2Cl2]+. This cobalt complex ion then reacts in acid solution to yield a mixture of the cis and trans forms of [Co(en)2(H2O)Cl]2+. The progress of this reaction ...
... In aqueous solution the green complex trans-dichlorbis (ethylenediamine) cobalt(III) chloride dissociates into chloride ion and trans-[Co(en)2Cl2]+. This cobalt complex ion then reacts in acid solution to yield a mixture of the cis and trans forms of [Co(en)2(H2O)Cl]2+. The progress of this reaction ...
17: Oxidation and Reduction
... We can also distill the intermediate aldehyde from the reaction mixture as it forms before it is oxidized further. This is often possible because boiling points of aldehydes are usually much lower than those of the 1° alcohols from which they are formed. Oxidation of Cyclic Ketones. When ketones rea ...
... We can also distill the intermediate aldehyde from the reaction mixture as it forms before it is oxidized further. This is often possible because boiling points of aldehydes are usually much lower than those of the 1° alcohols from which they are formed. Oxidation of Cyclic Ketones. When ketones rea ...
Experiment 1 - Melting Points - NAU jan.ucc.nau.edu web server
... melting. If a pure sample of a compound melts from 110 to 111.5°C, the addition of substantial amounts of another compound might result in a new melting point range from 85 to 100°C. An identical or near identical temperature range of melting is not, in itself, proof of the sameness of two organic c ...
... melting. If a pure sample of a compound melts from 110 to 111.5°C, the addition of substantial amounts of another compound might result in a new melting point range from 85 to 100°C. An identical or near identical temperature range of melting is not, in itself, proof of the sameness of two organic c ...
2013 Chemistry examination report
... indicated that students did not have a good understanding of the reasons why fuels are classified as renewable or non-renewable. Question 8b. and Question 8d. were challenging for some students. When asked to describe factors that might affect the accuracy of the results, students should try to answ ...
... indicated that students did not have a good understanding of the reasons why fuels are classified as renewable or non-renewable. Question 8b. and Question 8d. were challenging for some students. When asked to describe factors that might affect the accuracy of the results, students should try to answ ...
Alcohols, phenols and ethers
... • Thioethers are organic compounds in which two saturated carbon atoms are linked through a single sulfur atom. • The common naming system for thioethers is similar to that for ethers, with the name “ether” being replaced by “sulfide” ...
... • Thioethers are organic compounds in which two saturated carbon atoms are linked through a single sulfur atom. • The common naming system for thioethers is similar to that for ethers, with the name “ether” being replaced by “sulfide” ...
Structure and Synthesis of Alkenes
... Bicyclic means two rings. Bridgehead carbon is part of both rings. ...
... Bicyclic means two rings. Bridgehead carbon is part of both rings. ...
Final Exam - Dawson College
... surroundings to the system, the gas in the piston expands from 12 L to 27 L and performs work on the surroundings. If the system gains 201 J of internal energy from this process, against what constant external pressure, in atmospheres, is the piston working? ...
... surroundings to the system, the gas in the piston expands from 12 L to 27 L and performs work on the surroundings. If the system gains 201 J of internal energy from this process, against what constant external pressure, in atmospheres, is the piston working? ...
102 Lecture Ch14a
... O or the S, and are polar compounds • Alcohols and phenols contain a very polarized O-H bond, and they can H-bond with themselves and with other alcohols or water - Small alcohols (4 or less C’s) are soluble in water - Phenol is soluble in water (even with 6 C’s) because it partially ionizes in wate ...
... O or the S, and are polar compounds • Alcohols and phenols contain a very polarized O-H bond, and they can H-bond with themselves and with other alcohols or water - Small alcohols (4 or less C’s) are soluble in water - Phenol is soluble in water (even with 6 C’s) because it partially ionizes in wate ...
Si(OR - am Lehrstuhl für Bauchemie
... Hydrolysis Alkoxy silanes react readily with water by reaction called hydrolysis. The hydrolysis reaction, through the addition of water, replaces alkoxide group (OR) with hydroxyl group (OH-) which becomes attached to the silicon atom. Depending on the amount of water and catalysts present, hydroly ...
... Hydrolysis Alkoxy silanes react readily with water by reaction called hydrolysis. The hydrolysis reaction, through the addition of water, replaces alkoxide group (OR) with hydroxyl group (OH-) which becomes attached to the silicon atom. Depending on the amount of water and catalysts present, hydroly ...
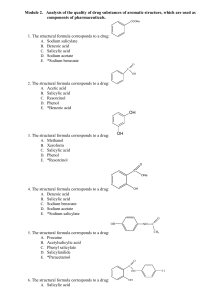
Module 2. Drug substances of aromatic structure
... C. 2-Hydroxybenzenecarboxylic acid D. Oxybenzene E. *2-Hydroxybenzenecarboxylate 56. In the medical practice salicylic acid use as means: A. Purgative B. Anti-inflammatory; analgesic C. Expectorant D. Antimicrobial preservative E. *Keratolytic 57. What indicator must be used, according to Pharmacopo ...
... C. 2-Hydroxybenzenecarboxylic acid D. Oxybenzene E. *2-Hydroxybenzenecarboxylate 56. In the medical practice salicylic acid use as means: A. Purgative B. Anti-inflammatory; analgesic C. Expectorant D. Antimicrobial preservative E. *Keratolytic 57. What indicator must be used, according to Pharmacopo ...
Chem 2A Final Review
... 9. The answer closest to the number of grams of NaH2PO4 needed to react with 38.74 mL of 0.275 M NaOH, according to the following balanced equation is: (NaH2PO4 = 119.98 g/mol) NaH2PO4 (s) + 2NaOH (aq) Na3POH4 (aq) + 3H2O ...
... 9. The answer closest to the number of grams of NaH2PO4 needed to react with 38.74 mL of 0.275 M NaOH, according to the following balanced equation is: (NaH2PO4 = 119.98 g/mol) NaH2PO4 (s) + 2NaOH (aq) Na3POH4 (aq) + 3H2O ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.