
Chapter 4 Student Notes
... o Bases are substances that are able to ionize in aqueous solution to form OH 1- (Arrhenius definition). o Bases are substances that accept or react with the H1+ ions formed by acids (proton acceptor). o Hydroxide ions, OH1-, react with the H1+ ions to form water: o H1+(aq) + OH1-(aq) H2O(l) o Com ...
... o Bases are substances that are able to ionize in aqueous solution to form OH 1- (Arrhenius definition). o Bases are substances that accept or react with the H1+ ions formed by acids (proton acceptor). o Hydroxide ions, OH1-, react with the H1+ ions to form water: o H1+(aq) + OH1-(aq) H2O(l) o Com ...
ALKENES INTRODUCING
... High density poly(ethene) has very little branching along the hydrocarbon chains - the crystallinity is 95% or better. This better packing means that van der Waals attractions between the chains are greater and so the plastic is stronger and has a higher melting point. Its density is also higher bec ...
... High density poly(ethene) has very little branching along the hydrocarbon chains - the crystallinity is 95% or better. This better packing means that van der Waals attractions between the chains are greater and so the plastic is stronger and has a higher melting point. Its density is also higher bec ...
CH 13
... H2O2(aq) H2O + ½ O2(g) It’s rate of decomposition is measured by titrating samples of the solution with potassium permanganate (KMnO4) at certain ...
... H2O2(aq) H2O + ½ O2(g) It’s rate of decomposition is measured by titrating samples of the solution with potassium permanganate (KMnO4) at certain ...
chemistry sp.indd
... Permission to reproduce items where third-party owned material protected by copyright is included has been sought and cleared where possible. Every reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included ...
... Permission to reproduce items where third-party owned material protected by copyright is included has been sought and cleared where possible. Every reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included ...
heterogeneous chiral catalyst derived from hydrolyzed
... development of efficient routes to enantiopure compounds was perceived as cumbersome and expensive. However, following the understanding that different enantiomers may have qualitatively distinct physiological effects, [16-20] and instigated by stricter regulations from health authorities, a growing ...
... development of efficient routes to enantiopure compounds was perceived as cumbersome and expensive. However, following the understanding that different enantiomers may have qualitatively distinct physiological effects, [16-20] and instigated by stricter regulations from health authorities, a growing ...
3 - Rates
... energy to react a short lived activated complex is formed. At this point no bond breaking or bond making is occuring. 3 - At the same time new bonds are starting to form between H and Cl as H nuclei attract Cl electrons and Cl nuclei attract H electrons 2 - As the reacting molecules5 - If the reacti ...
... energy to react a short lived activated complex is formed. At this point no bond breaking or bond making is occuring. 3 - At the same time new bonds are starting to form between H and Cl as H nuclei attract Cl electrons and Cl nuclei attract H electrons 2 - As the reacting molecules5 - If the reacti ...
The Cardiac Glycosides Cardio
... 1- Squill Glycosides • They have 6-membered lactone ring. • Obtained from Squill bulbs. • Aglycone contains only two hydroxyl groups at C-3 and C-14. ...
... 1- Squill Glycosides • They have 6-membered lactone ring. • Obtained from Squill bulbs. • Aglycone contains only two hydroxyl groups at C-3 and C-14. ...
16.1 The Carbonyl Group
... by a double bond. • The bond angles between the three substituents on the carbonyl carbon atom are 120°, or close to it. ...
... by a double bond. • The bond angles between the three substituents on the carbonyl carbon atom are 120°, or close to it. ...
Biology 251 Fall 2015 1 TOPIC 23: ACID BASE BALANCE I
... Person can (unless they have a respiratory disease) always alter ventilation rates to change plasma acid-base balance. H. Respiratory system usually only returns pH 50% to 75% of normal, because as pH gets closer to normal, the less the ventilation rates are influenced. IV. Kidneys are third line of ...
... Person can (unless they have a respiratory disease) always alter ventilation rates to change plasma acid-base balance. H. Respiratory system usually only returns pH 50% to 75% of normal, because as pH gets closer to normal, the less the ventilation rates are influenced. IV. Kidneys are third line of ...
NO - Blue Devil Chem
... 1. 3Co2+ + 2Al → 3Co + 2Al3+ 2. 2Na + 2H2O → 2NaOH + H2 3. 2HCl + Zn → ZnCl2 + H2 4. 2HNO3 + Mg(OH)2 → 2H2O + Mg(NO3)2 5. CH4 + 2O2 → CO2 + 2H2O • Double replacement reactions are never oxidation reduction reactions since none of the atoms change oxidation numbers. ...
... 1. 3Co2+ + 2Al → 3Co + 2Al3+ 2. 2Na + 2H2O → 2NaOH + H2 3. 2HCl + Zn → ZnCl2 + H2 4. 2HNO3 + Mg(OH)2 → 2H2O + Mg(NO3)2 5. CH4 + 2O2 → CO2 + 2H2O • Double replacement reactions are never oxidation reduction reactions since none of the atoms change oxidation numbers. ...
Name_____________________________________ 22 • Organic
... is added. The resulting product will react with a base to form a salt and water. When the new product in container X is mixed with container Y and H2SO4 is added, the chemist is able to confirm that: ...
... is added. The resulting product will react with a base to form a salt and water. When the new product in container X is mixed with container Y and H2SO4 is added, the chemist is able to confirm that: ...
Glycosyl amines
... According to the non-saccharidic nature of the amino group, they are devided into unsubstituted, aliphatic and aromatic glycosyl amines. Aromatic glycosyl amines are much more stable than aliphatic ones. Similarly as free aldoses or ketoses (glycoses), they undergo mutarotation. A treatment with min ...
... According to the non-saccharidic nature of the amino group, they are devided into unsubstituted, aliphatic and aromatic glycosyl amines. Aromatic glycosyl amines are much more stable than aliphatic ones. Similarly as free aldoses or ketoses (glycoses), they undergo mutarotation. A treatment with min ...
Organic_1_6.1ed_2012_02nd_module_functional_groups_and_IR
... For each of the following IR spectra, identify whether it is consistent with the structure of a ketone, an alcohol, a carboxylic acid, a primary amine, or a secondary amine. Explain your answer. ...
... For each of the following IR spectra, identify whether it is consistent with the structure of a ketone, an alcohol, a carboxylic acid, a primary amine, or a secondary amine. Explain your answer. ...
Microsoft Word - Open Access Repository of Indian Theses
... The conjugate addition (1,4-addition or Michael addition) of nucleophiles to α,βunsaturated compounds is one of the most important new bond-forming strategies in synthetic organic chemistry. Aza-Michael addition is one of the important reactions especially for the synthesis of C-N heterocycles conta ...
... The conjugate addition (1,4-addition or Michael addition) of nucleophiles to α,βunsaturated compounds is one of the most important new bond-forming strategies in synthetic organic chemistry. Aza-Michael addition is one of the important reactions especially for the synthesis of C-N heterocycles conta ...
Bio 2 alkanes+isomerism
... Suppose we want to add one more C atom to propane. We have two different kinds of H we can replace, a methylene H or a methyl H. If we replace the methyl H, we have butane. If we replace the methylene H, we have a skeletal isomer of butane. This isomer of butane has a common or trivial name, isobuta ...
... Suppose we want to add one more C atom to propane. We have two different kinds of H we can replace, a methylene H or a methyl H. If we replace the methyl H, we have butane. If we replace the methylene H, we have a skeletal isomer of butane. This isomer of butane has a common or trivial name, isobuta ...
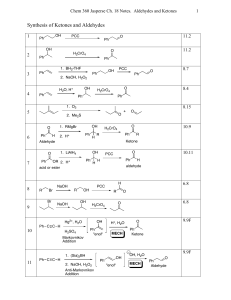
Synthesis of Ketones and Aldehydes
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
Multiple Pathways To Success Quarter 3 Learning Module
... 21. A student writes the chemical formula for the ionic compound calcium hydroxide as CaOH2. a. Write the chemical formula for each ion in the compound. ...
... 21. A student writes the chemical formula for the ionic compound calcium hydroxide as CaOH2. a. Write the chemical formula for each ion in the compound. ...
Chapter 14 - faculty at Chemeketa
... Neutralization of Carboxylic Acids Carboxylic acid salts • are a product of the neutralization of a carboxylic acid with a strong base. CH3—COOH + NaOH CH3—COO– Na+ + H2O acetic acid sodium acetate (carboxylic acid salt) ...
... Neutralization of Carboxylic Acids Carboxylic acid salts • are a product of the neutralization of a carboxylic acid with a strong base. CH3—COOH + NaOH CH3—COO– Na+ + H2O acetic acid sodium acetate (carboxylic acid salt) ...
[SESSION-2014-2015] SUBJECT - SCIENCE PATNA REGION
... Some salts precipitate due to mutual exchange of ions between the two salts. For example ...
... Some salts precipitate due to mutual exchange of ions between the two salts. For example ...
Synthesis of Ketones and Aldehydes
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
Reactions You Should Know When You Begin Organic II
... Adding agent can be symmetrical or asymmetrical. Ex. H2 vs. HCl Symmetrical: H2, C12, Br2, and I2 (I2 slow and readily reversible) Asymmetrical: HCl, RBr, HOH (H2O) Addition of symmetrical agents may be anti or syn depending on mechanism or catalyst. Addition of asymmetrical agents follows Markovnik ...
... Adding agent can be symmetrical or asymmetrical. Ex. H2 vs. HCl Symmetrical: H2, C12, Br2, and I2 (I2 slow and readily reversible) Asymmetrical: HCl, RBr, HOH (H2O) Addition of symmetrical agents may be anti or syn depending on mechanism or catalyst. Addition of asymmetrical agents follows Markovnik ...
Chapter 16 Alkanes and alkenes
... alkenes burn in air to form CO2 and H2O incomplete combustion results in the formation of carbon monoxide, CO and soot, C alkenes tend to produce more soot than alkanes when they burn in air example: combustion of ethene ...
... alkenes burn in air to form CO2 and H2O incomplete combustion results in the formation of carbon monoxide, CO and soot, C alkenes tend to produce more soot than alkanes when they burn in air example: combustion of ethene ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.



















![[SESSION-2014-2015] SUBJECT - SCIENCE PATNA REGION](http://s1.studyres.com/store/data/008930072_1-5a35e1ae8e3204ea88999f1418a93013-300x300.png)


