C H
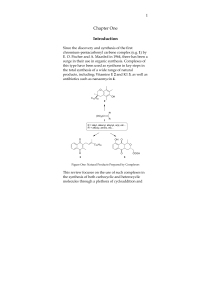
... dentistry. It also can be useful for students in environmental studies. Most students who take such courses consider bioorganic chemistry to be the most relevant part of the course of study. However, an understanding of bioorganic chemistry depends upon a sound background in organic chemistry. These ...
... dentistry. It also can be useful for students in environmental studies. Most students who take such courses consider bioorganic chemistry to be the most relevant part of the course of study. However, an understanding of bioorganic chemistry depends upon a sound background in organic chemistry. These ...
Ch-1-Alkanes and isomerism-corr
... Know the different methods of representing molecular formulas Know the different classes of carbon and hydrogen atoms know the hybridization and geometry of alkanes Know the rules for naming branched chain alkanes and how to use them. Know the physical properties of alkanes and factors aff ...
... Know the different methods of representing molecular formulas Know the different classes of carbon and hydrogen atoms know the hybridization and geometry of alkanes Know the rules for naming branched chain alkanes and how to use them. Know the physical properties of alkanes and factors aff ...
Latest Publication (still not complete)
... cyclization processes, concentrating on the synthesis of three- to seven-membered ring containing products. However, in addition to this, the structure and bonding of carbene complexes is also discussed. In particular, an analysis of information gained through computational analysis is provided. Suc ...
... cyclization processes, concentrating on the synthesis of three- to seven-membered ring containing products. However, in addition to this, the structure and bonding of carbene complexes is also discussed. In particular, an analysis of information gained through computational analysis is provided. Suc ...
Unit 1
... The Laboratory outcomes 214-10, 213-1, 213-8, 214-3, 214-8 and, in part, ACC-2 are addressed by completing Studying Reaction Rates, CORE LAB #1. Students could perform lab experiments to predict which reaction they think would be faster. Using a potassium permanganate solution to react with differen ...
... The Laboratory outcomes 214-10, 213-1, 213-8, 214-3, 214-8 and, in part, ACC-2 are addressed by completing Studying Reaction Rates, CORE LAB #1. Students could perform lab experiments to predict which reaction they think would be faster. Using a potassium permanganate solution to react with differen ...
Studies of Carbon-Sulfur Bond Cleavage by Homogeneous
... The reactive fragment [(C5Me5)Rh(PMe3)] produced by heating solutions of (C5Me5)Rh(PMe3)(Ph)H has been found to insert into a wide variety of thiophene C-S bonds (Scheme I).1 One of the first products to be structurally characterized was the adduct formed with 2,5-dimethylthiophene. This C-S insert ...
... The reactive fragment [(C5Me5)Rh(PMe3)] produced by heating solutions of (C5Me5)Rh(PMe3)(Ph)H has been found to insert into a wide variety of thiophene C-S bonds (Scheme I).1 One of the first products to be structurally characterized was the adduct formed with 2,5-dimethylthiophene. This C-S insert ...
Chapter 4
... redox equations are available, and are based on the fact that the total electrons gained in reduction equals the total lost in oxidation. The two methods: 1) Use oxidation state changes 2) Use half-reactions (the method to be used her) ...
... redox equations are available, and are based on the fact that the total electrons gained in reduction equals the total lost in oxidation. The two methods: 1) Use oxidation state changes 2) Use half-reactions (the method to be used her) ...
Essential Oils Composition
... • 1.8% geranyl acetate • 1.7% linalool • 1.1% (Z.E)-farnesol • 1.1% nerolidol • 0.6% geraniol • 0.1% benzyl salicylate ...
... • 1.8% geranyl acetate • 1.7% linalool • 1.1% (Z.E)-farnesol • 1.1% nerolidol • 0.6% geraniol • 0.1% benzyl salicylate ...
Nomenclature
... Halohydrin Formation • Bromohydrins are also formed with N-bromosuccinimide (NBS) in aqueous DMSO [(CH3)2S=O]. • In H2O, NBS decomposes to form Br2, which then goes on to form a bromohydrin by the same reaction mechanism. ...
... Halohydrin Formation • Bromohydrins are also formed with N-bromosuccinimide (NBS) in aqueous DMSO [(CH3)2S=O]. • In H2O, NBS decomposes to form Br2, which then goes on to form a bromohydrin by the same reaction mechanism. ...
Properties of Systems in Equilibrium - Le
... of the test tube gently for a few seconds using the glass stirring-rod and record your observations on your data sheet. Continue heating and stirring until a change is observed. 7. Make a cold-water bath by filling a 400-mL beaker half full with tap water and ice. Cool the test tube down by placing ...
... of the test tube gently for a few seconds using the glass stirring-rod and record your observations on your data sheet. Continue heating and stirring until a change is observed. 7. Make a cold-water bath by filling a 400-mL beaker half full with tap water and ice. Cool the test tube down by placing ...
TYPES OF HYBRIDIZATION AND GEOMETRY OF MOLECULES
... know the difference in structure between alcohols and phenols Know the different classes of alcohols Know how to name alcohols and phenols Know the physical properties (solubility, boiling and melting points) Know how hydrogen bonds are formed and its effect on boiling points of ...
... know the difference in structure between alcohols and phenols Know the different classes of alcohols Know how to name alcohols and phenols Know the physical properties (solubility, boiling and melting points) Know how hydrogen bonds are formed and its effect on boiling points of ...
Topic 4
... A molecular/formula unit equation is one in which the reactants and products are written as if they were molecules/formula units, even though they may actually exist in solution as ions. Many ionic compounds undergo a displacement reaction between the cation of one species with the anion of another. ...
... A molecular/formula unit equation is one in which the reactants and products are written as if they were molecules/formula units, even though they may actually exist in solution as ions. Many ionic compounds undergo a displacement reaction between the cation of one species with the anion of another. ...
the suzuki-miyaura reaction and boron reagents – mechanism
... mild reaction conditions, especially versatile with palladium(II) complexes. ...
... mild reaction conditions, especially versatile with palladium(II) complexes. ...
Chapter-16B
... There are two major differences between acidcatalyzed and base-promoted ester hydrolysis 1. for acid-catalyzed hydrolysis, acid is required in only catalytic amounts; for base-promoted hydrolysis, base is required in equimolar amounts 2. hydrolysis of an ester in aqueous acid is reversible; base-pro ...
... There are two major differences between acidcatalyzed and base-promoted ester hydrolysis 1. for acid-catalyzed hydrolysis, acid is required in only catalytic amounts; for base-promoted hydrolysis, base is required in equimolar amounts 2. hydrolysis of an ester in aqueous acid is reversible; base-pro ...
Document
... – Reduction of an aldehyde gives a primary alcohol (-CH2OH). – Reduction of a ketone gives a secondary alcohol (-CHOH-). ...
... – Reduction of an aldehyde gives a primary alcohol (-CH2OH). – Reduction of a ketone gives a secondary alcohol (-CHOH-). ...
13.0 Redox Reactions PowerPoint
... ▫ Helpful reference: Table 6 pg. 575 ▫ Remember: In solutions, molecules and ions behave independently of each other. ...
... ▫ Helpful reference: Table 6 pg. 575 ▫ Remember: In solutions, molecules and ions behave independently of each other. ...
380 KB / 39 pages
... tells us that each dimethylglyoxime anion must have a charge of –1 in order to balance the cationic charge. When you analyzed the data in Check This 6.11, you probably focused on the sample that produced the largest amount of precipitate and found that the molar ratio of nickel to dimethylglyoxime i ...
... tells us that each dimethylglyoxime anion must have a charge of –1 in order to balance the cationic charge. When you analyzed the data in Check This 6.11, you probably focused on the sample that produced the largest amount of precipitate and found that the molar ratio of nickel to dimethylglyoxime i ...
Part-1
... Osmotic pressure of a solution is directly proportional to the number of moles of solute dissolved per litre of solution at a given temperature. Solutions having equal molar concentration and equal osmotic pressure at a given temperature are called isotonic solutions, e.g., A 0.90% (mass/volume) sol ...
... Osmotic pressure of a solution is directly proportional to the number of moles of solute dissolved per litre of solution at a given temperature. Solutions having equal molar concentration and equal osmotic pressure at a given temperature are called isotonic solutions, e.g., A 0.90% (mass/volume) sol ...
Study Guide 2 - Chemistry Teaching Resources
... KHS Jan 2015 - Cheviot Learning Community - based on Challenge Chemistry © R.I.S.E ...
... KHS Jan 2015 - Cheviot Learning Community - based on Challenge Chemistry © R.I.S.E ...
Chem 226 — Problem Set #2
... “Fundamentals of Organic Chemistry,” 4th edition, John McMurry. Chapter 2 ...
... “Fundamentals of Organic Chemistry,” 4th edition, John McMurry. Chapter 2 ...
comparison of tranestrification efficiency using alkaline and acid
... Biodiesel FAME, were obtained from the two-stage process through catalytic commixtures with the following alkaline catalysts: KOH and NaOH. Potassium and sodium hydroxides are good and cheap catalysts. Their fault results from their hygroscopic characteristics; even vestigial traces of moisture caus ...
... Biodiesel FAME, were obtained from the two-stage process through catalytic commixtures with the following alkaline catalysts: KOH and NaOH. Potassium and sodium hydroxides are good and cheap catalysts. Their fault results from their hygroscopic characteristics; even vestigial traces of moisture caus ...
5. Stoichiometry - Sakshi Education
... c) In acid medium include enough number of water molecules where there is a deficiency of oxygen and include enough number of H + ions on the side where there is a deficiency of Hydrogen. d) In alkaline medium include enough number of OH⎯ ions on the side where there is a deficiency of oxygen and en ...
... c) In acid medium include enough number of water molecules where there is a deficiency of oxygen and include enough number of H + ions on the side where there is a deficiency of Hydrogen. d) In alkaline medium include enough number of OH⎯ ions on the side where there is a deficiency of oxygen and en ...
Key Words Questions and Problems
... Cinnamic alcohol is used mainly in perfumery, particularly in soaps and cosmetics. Its molecular formula is C9H10O. (a) Calculate the percent composition by mass of C, H, and O in cinnamic alcohol. (b) How many molecules of cinnamic alcohol are contained in a sample of mass 0.469 g? All of the subst ...
... Cinnamic alcohol is used mainly in perfumery, particularly in soaps and cosmetics. Its molecular formula is C9H10O. (a) Calculate the percent composition by mass of C, H, and O in cinnamic alcohol. (b) How many molecules of cinnamic alcohol are contained in a sample of mass 0.469 g? All of the subst ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.