Sheet #24 (archive) Medicinal Plants
... coloring agents . The primary classes of tetratepines ,we have: carotene ,indicating already hydrocarbons Xanthophylls, composed of carbon, hydrogen & oxygen, both of them are C40 compounds there is small group considered as degradation product of carotenes & carotinoids are CAROTINOID ACID( the nam ...
... coloring agents . The primary classes of tetratepines ,we have: carotene ,indicating already hydrocarbons Xanthophylls, composed of carbon, hydrogen & oxygen, both of them are C40 compounds there is small group considered as degradation product of carotenes & carotinoids are CAROTINOID ACID( the nam ...
Alcohols phenols
... By hydrolysis of alkyl halides. Alkyl halides on hydrolysis with aqueous alkalies or moist silver oxide give alcohols. In general, alkyl halides are prepared from alcohols as the latter are easily available. It is a nucleophilic substitution reaction in which hydroxide ion substitutes halide ions. A ...
... By hydrolysis of alkyl halides. Alkyl halides on hydrolysis with aqueous alkalies or moist silver oxide give alcohols. In general, alkyl halides are prepared from alcohols as the latter are easily available. It is a nucleophilic substitution reaction in which hydroxide ion substitutes halide ions. A ...
... Since the establishment of the Nobel Prize over a century ago advances in synthetic methodology in organic chemistry have received recognition of five of the Nobel Chemistry Prizes. All involve the carbon-carbon bond – its construction and chemistry. This is not surprising given the importance of c ...
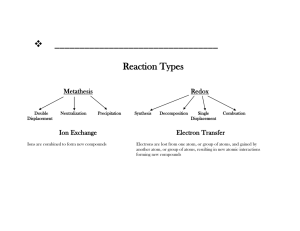
The Major Classes of Chemical Reactions
... do not conduct an electric current, these substances are called nonelectrolytes. Many other covalent substances, such as benzene (C6H6) and octane (C8H18), do not contain polar bonds, and these substances do not dissolve appreciably in water. A small, but very important, group of H-containing covale ...
... do not conduct an electric current, these substances are called nonelectrolytes. Many other covalent substances, such as benzene (C6H6) and octane (C8H18), do not contain polar bonds, and these substances do not dissolve appreciably in water. A small, but very important, group of H-containing covale ...
CHEM 1212 Principles of Chemistry II Course Study Guide
... •Taking quizzes….even mock quizzes your generate for each other in a study group •Explaining to someone else how to get an answer in your own words Retrieval is what you will be expected to do on tests. Why wouldn’t you practice it beforehand? *Spaced repetition means taking breaks between practice, ...
... •Taking quizzes….even mock quizzes your generate for each other in a study group •Explaining to someone else how to get an answer in your own words Retrieval is what you will be expected to do on tests. Why wouldn’t you practice it beforehand? *Spaced repetition means taking breaks between practice, ...
Organic Acids and Bases and Some of Their Derivatives
... found in carboxylic acids, esters, and amides. However, in these compounds, the carbonyl group is only part of the functional group. A carboxylic acid1 is an organic compound that has a carboxyl group2. The carboxyl group is a functional group that contains a carbon–oxygen double bond and an OH grou ...
... found in carboxylic acids, esters, and amides. However, in these compounds, the carbonyl group is only part of the functional group. A carboxylic acid1 is an organic compound that has a carboxyl group2. The carboxyl group is a functional group that contains a carbon–oxygen double bond and an OH grou ...
File
... such that they are ________ from one phase to another, while an equal number are moving in the reverse direction e.g. H2O(l) → H20(g) 3. Chemical Reaction Equilibrium - Quantitative reactions are those reactions where ___ of the limiting reagent is ___________ in the reaction e.g. CaCO3 (s) → CaO(s) ...
... such that they are ________ from one phase to another, while an equal number are moving in the reverse direction e.g. H2O(l) → H20(g) 3. Chemical Reaction Equilibrium - Quantitative reactions are those reactions where ___ of the limiting reagent is ___________ in the reaction e.g. CaCO3 (s) → CaO(s) ...
Basic definitions for organic chemistry
... The molecular formula is an exact multiple of the empirical formula. Comparing the molecular mass with the empirical mass allows one to find the true formula. if the empirical formula is CH (relative mass = 13) and the molecular mass is 78 the molecular formula will be 78/13 or 6 times the empirical ...
... The molecular formula is an exact multiple of the empirical formula. Comparing the molecular mass with the empirical mass allows one to find the true formula. if the empirical formula is CH (relative mass = 13) and the molecular mass is 78 the molecular formula will be 78/13 or 6 times the empirical ...
acid
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
Chapter 18 Carboxylic Acids
... The problem of formation of precipitates in hard water was overcome by using a molecule containing a sulfonate (-SO3) group in the place of a carboxylate (-CO2-) group. • Calcium, magnesium and iron salts of sulfonic acids, RSO3H, are more soluble in water than are their salts of ...
... The problem of formation of precipitates in hard water was overcome by using a molecule containing a sulfonate (-SO3) group in the place of a carboxylate (-CO2-) group. • Calcium, magnesium and iron salts of sulfonic acids, RSO3H, are more soluble in water than are their salts of ...
Ch12-Alcohols-Grignard
... What remains is the combination of Grignard reagent and carbonyl compound that can be used to prepare the alcohol. ...
... What remains is the combination of Grignard reagent and carbonyl compound that can be used to prepare the alcohol. ...
Chapter 17
... – treatment of a Grignard reagent with carbon dioxide followed by acidification gives a carboxylic acid O MgBr + C O Carb on dioxid e O C-O - [MgBr] + A magnesium carboxylate ...
... – treatment of a Grignard reagent with carbon dioxide followed by acidification gives a carboxylic acid O MgBr + C O Carb on dioxid e O C-O - [MgBr] + A magnesium carboxylate ...
Reaction Rates/Chemical Kinetics
... In this reaction, SO2 and O2 are placed in a container. Initially, the forward reaction proceeds and SO3 is produced. The rate of the forward reaction is much greater than the rate of the reverse reaction. As SO3 builds up, it starts to decompose into SO2 and O2. The rate of the forward reaction is ...
... In this reaction, SO2 and O2 are placed in a container. Initially, the forward reaction proceeds and SO3 is produced. The rate of the forward reaction is much greater than the rate of the reverse reaction. As SO3 builds up, it starts to decompose into SO2 and O2. The rate of the forward reaction is ...
Resolution of Diols via Catalytic Asymmetric Acetalization
... diols with tertiary alcohol stereocenters with high enantioselectivity are much less common.2c,7d,e Since racemic diols are readily accessible, kinetic resolution can be an attractive alternative strategy for the preparation of enantioenriched diols.8 Methods for kinetic resolution of diols via asym ...
... diols with tertiary alcohol stereocenters with high enantioselectivity are much less common.2c,7d,e Since racemic diols are readily accessible, kinetic resolution can be an attractive alternative strategy for the preparation of enantioenriched diols.8 Methods for kinetic resolution of diols via asym ...
A Review of using Spray Pyrolysis through Sol-gel
... Received 3 July 2015, revised 30 March 2016, accepted 30 March 2016. ...
... Received 3 July 2015, revised 30 March 2016, accepted 30 March 2016. ...
Prep UK-intro.p65
... 198 trivial names of most important (ca 5) f.acids 199 general metabolism of fats 200 beta-oxidation of fatty acids (formulas & ...
... 198 trivial names of most important (ca 5) f.acids 199 general metabolism of fats 200 beta-oxidation of fatty acids (formulas & ...
Determination of Cystein and Methionine by Oscillating Chemical
... Several methods, such as pH-metery [10], potentiometry [11,12], luminometry [13,14], conductometery [15] and fluorimetry [16] have been applied to investigate of oscillating reaction One of us (Samadi-Maybodi) for the first time reported that the oscillating system, H2O2– KSCN–CuSO4–NaOH, could be e ...
... Several methods, such as pH-metery [10], potentiometry [11,12], luminometry [13,14], conductometery [15] and fluorimetry [16] have been applied to investigate of oscillating reaction One of us (Samadi-Maybodi) for the first time reported that the oscillating system, H2O2– KSCN–CuSO4–NaOH, could be e ...
Carbonyl Condensation Reactions
... Much of what is presented in Chapter 24 applies principles you have already learned. Many of the reactions may look more complicated than those in previous chapters, but they are fundamentally the same. Nucleophiles attack electrophilic carbonyl groups to form the products of nucleophilic addition o ...
... Much of what is presented in Chapter 24 applies principles you have already learned. Many of the reactions may look more complicated than those in previous chapters, but they are fundamentally the same. Nucleophiles attack electrophilic carbonyl groups to form the products of nucleophilic addition o ...
Chemistry 110 Oxidation Reduction Reactions Oxidation Number
... We can see that the oxidation number of C increases from -4 to +4 in this reaction, so C is oxidized. We can also see that the oxidation number of O decreases from zero (0) to -2, so O is reduced. Notice that the oxidation number of hydrogen does not change. It is always the case that if any element ...
... We can see that the oxidation number of C increases from -4 to +4 in this reaction, so C is oxidized. We can also see that the oxidation number of O decreases from zero (0) to -2, so O is reduced. Notice that the oxidation number of hydrogen does not change. It is always the case that if any element ...
Writing and Predicting Chemical Reactions
... Amphoteric metals (such as Al, Zn, Pb, and Hg) have properties that may be intermediate between those of metals and those of nonmetals. They will react with a base to form a complex ion with oxygen. This is a rare problem. 1. A piece of solid aluminum is added to a 6 M solution of sodium hydroxide: ...
... Amphoteric metals (such as Al, Zn, Pb, and Hg) have properties that may be intermediate between those of metals and those of nonmetals. They will react with a base to form a complex ion with oxygen. This is a rare problem. 1. A piece of solid aluminum is added to a 6 M solution of sodium hydroxide: ...
full text pdf
... the process speed to resistance of diffusion through the gas film (external film diffusion), diffusion inside the grain (diffusion through reacted material) and the chemical reaction. Another model of the process was presented by Kimura and Smith8, who have conducted the studies in the temperature r ...
... the process speed to resistance of diffusion through the gas film (external film diffusion), diffusion inside the grain (diffusion through reacted material) and the chemical reaction. Another model of the process was presented by Kimura and Smith8, who have conducted the studies in the temperature r ...
Collected Essays chapter 13 answers
... (c) Determine Kp, in terms of Kc for this system. Kp = 5.0 (d) When the system is cooled from 2,000 K to a lower temperature, 30.0 percent of the CO(g) is converted back to CO2(g). Calculate the value of Kc at this lower temperature. 0.92 (e) In a different experiment, 0.50 mole of H2(g) is mixed wi ...
... (c) Determine Kp, in terms of Kc for this system. Kp = 5.0 (d) When the system is cooled from 2,000 K to a lower temperature, 30.0 percent of the CO(g) is converted back to CO2(g). Calculate the value of Kc at this lower temperature. 0.92 (e) In a different experiment, 0.50 mole of H2(g) is mixed wi ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.