enantioselective zeolite-catalyzed reactions
... Beginning with the first homogeneous asymmetric metal-catalyzed reaction performed by Noyori—a cyclopropanation—the area of enantioselective catalysis in organic chemistry has become a major topic of discussion and research.1 Homogeneous, asymmetry-inducing catalysts have been developed for a wide r ...
... Beginning with the first homogeneous asymmetric metal-catalyzed reaction performed by Noyori—a cyclopropanation—the area of enantioselective catalysis in organic chemistry has become a major topic of discussion and research.1 Homogeneous, asymmetry-inducing catalysts have been developed for a wide r ...
Sample Chapter - Chapter 4
... Water separates ions in a process that greatly reduces the electrostatic force of attraction between them. To see how it does this, let’s examine the water molecule closely. Water’s power as an ionizing solvent results from two features of the water molecule: the distribution of its bonding electron ...
... Water separates ions in a process that greatly reduces the electrostatic force of attraction between them. To see how it does this, let’s examine the water molecule closely. Water’s power as an ionizing solvent results from two features of the water molecule: the distribution of its bonding electron ...
Carefully detach the last page. It is the Data Sheet.
... 40 Ethanoic acid, CH3COOH, is a weak acid in water. What happens when 0.01 moles of HCl are added to a 0.1 mol L−1 solution of ethanoic acid? ...
... 40 Ethanoic acid, CH3COOH, is a weak acid in water. What happens when 0.01 moles of HCl are added to a 0.1 mol L−1 solution of ethanoic acid? ...
Problem Set Chapter 8: Introduction to Alkyl Halides, Alcohols
... 8.56 When sec-butylbenzene undergoes free-radical bromination, one major product is formed, as shown in Fig. P8.56. If the starting material is optically active, predict whether the substitution product should also be optically active. (Hint: Consider the geometry of the free-radical intermediate; s ...
... 8.56 When sec-butylbenzene undergoes free-radical bromination, one major product is formed, as shown in Fig. P8.56. If the starting material is optically active, predict whether the substitution product should also be optically active. (Hint: Consider the geometry of the free-radical intermediate; s ...
CHEMICAL EQUATIONS - Clayton State University
... HCl(aq) + NaHCO3(aq) → NaCl(aq) + H2CO3(aq) 2HCl(aq) + Na2CO3(aq) → 2NaCl(aq) + H2CO3(aq) - H2CO3 (carbonic acid) is unstable and decomposes to produce CO2 H2CO3(aq) → H2O(l) + CO2(g) - Hydrogen sulfide (H2S) is produced when Na2S reacts with an acid 2HCl(aq) + Na2S(aq) → 2NaCl(aq) + H2S(g) ...
... HCl(aq) + NaHCO3(aq) → NaCl(aq) + H2CO3(aq) 2HCl(aq) + Na2CO3(aq) → 2NaCl(aq) + H2CO3(aq) - H2CO3 (carbonic acid) is unstable and decomposes to produce CO2 H2CO3(aq) → H2O(l) + CO2(g) - Hydrogen sulfide (H2S) is produced when Na2S reacts with an acid 2HCl(aq) + Na2S(aq) → 2NaCl(aq) + H2S(g) ...
For Peer Review Only
... Vegetable oils can be epoxidized (reaction A, Table 1), leading to interesting building blocks for biobased chemistry 21. The most currently used method to epoxidize vegetable oil is based on peracetic acid formed in situ from reaction between acetic acid ...
... Vegetable oils can be epoxidized (reaction A, Table 1), leading to interesting building blocks for biobased chemistry 21. The most currently used method to epoxidize vegetable oil is based on peracetic acid formed in situ from reaction between acetic acid ...
isompp
... Carbonyl compounds undergo nucleophilic addition. If there are two different groups attached to the C=O bond, the possibility of forming optical isomers arises. THE NUCLEOPHILIC ADDITION OF HCN TO ETHANAL If the nucleophilic cyanide ion attacks from above one optical isomer is formed ...
... Carbonyl compounds undergo nucleophilic addition. If there are two different groups attached to the C=O bond, the possibility of forming optical isomers arises. THE NUCLEOPHILIC ADDITION OF HCN TO ETHANAL If the nucleophilic cyanide ion attacks from above one optical isomer is formed ...
No Slide Title
... Carbonyl compounds undergo nucleophilic addition. If there are two different groups attached to the C=O bond, the possibility of forming optical isomers arises. THE NUCLEOPHILIC ADDITION OF HCN TO ETHANAL If the nucleophilic cyanide ion attacks from above one optical isomer is formed ...
... Carbonyl compounds undergo nucleophilic addition. If there are two different groups attached to the C=O bond, the possibility of forming optical isomers arises. THE NUCLEOPHILIC ADDITION OF HCN TO ETHANAL If the nucleophilic cyanide ion attacks from above one optical isomer is formed ...
Isomerism Guide
... Carbonyl compounds undergo nucleophilic addition. If there are two different groups attached to the C=O bond, the possibility of forming optical isomers arises. THE NUCLEOPHILIC ADDITION OF HCN TO ETHANAL If the nucleophilic cyanide ion attacks from above one optical isomer is formed ...
... Carbonyl compounds undergo nucleophilic addition. If there are two different groups attached to the C=O bond, the possibility of forming optical isomers arises. THE NUCLEOPHILIC ADDITION OF HCN TO ETHANAL If the nucleophilic cyanide ion attacks from above one optical isomer is formed ...
Drying Solvents: Note: When the solvent is to be distilled after
... give a dark blue ketyl radical which is protonated by water to give colorless products. Thus the sodium-benzophenone system is particularly convenient because it is selfindicating, and it is the preferred reagent for rigorous drying of diethyl ether, THF, DME, and other ethereal solvents. Sodium-pot ...
... give a dark blue ketyl radical which is protonated by water to give colorless products. Thus the sodium-benzophenone system is particularly convenient because it is selfindicating, and it is the preferred reagent for rigorous drying of diethyl ether, THF, DME, and other ethereal solvents. Sodium-pot ...
Carboxylic acids, esters, and other acid derivatives
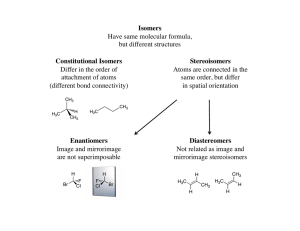
... • Recall that constitutional isomers are molecules that share the same formula but differ in their atom-to-atom connectivities. • Three kinds of constitutional isomers (in the order we encountered them): – positional isomers – skeletal isomers – functional group isomers ...
... • Recall that constitutional isomers are molecules that share the same formula but differ in their atom-to-atom connectivities. • Three kinds of constitutional isomers (in the order we encountered them): – positional isomers – skeletal isomers – functional group isomers ...
Diastereoselective Allylation of Carbonyl Compounds and Imines:
... chiral α-alkoxyaldehydes under these reaction conditions provided excellent yields with high stereoselectivity, a single diastereomer being observed in many cases by NMR analyses of the crude reaction product (Table 2, entries 1−3 and 5). Regarding the relative configuration of the created stereogeni ...
... chiral α-alkoxyaldehydes under these reaction conditions provided excellent yields with high stereoselectivity, a single diastereomer being observed in many cases by NMR analyses of the crude reaction product (Table 2, entries 1−3 and 5). Regarding the relative configuration of the created stereogeni ...
chemistry-subject test5 w. solutions
... dispersion forces (in the order of decreasing strength). For the ideal gas law to give an accurate prediction of the volume, then, we are looking for gases that do not have ahttp://doc.guandang.net/bbca35c11081d34250955e480.html strong dipole moment. Methane, CH4, does not have a dipole moment: What ...
... dispersion forces (in the order of decreasing strength). For the ideal gas law to give an accurate prediction of the volume, then, we are looking for gases that do not have ahttp://doc.guandang.net/bbca35c11081d34250955e480.html strong dipole moment. Methane, CH4, does not have a dipole moment: What ...
Types of reactions you know:
... b. CuLi and alkyl halide (use this if you need to add to the middle of a chain) ...
... b. CuLi and alkyl halide (use this if you need to add to the middle of a chain) ...
CHAPTER V Fischer-Tropsch Syncrude
... hydrocarbons (>C20), do not have a single α-value, but two. The first α-value (α1) describes the carbon number distribution from C3 to C12, while the second α-value (α2) describes the distribution of the C20 and heavier fraction.(4) This seems to be a mathematical convenience, since the α-value seem ...
... hydrocarbons (>C20), do not have a single α-value, but two. The first α-value (α1) describes the carbon number distribution from C3 to C12, while the second α-value (α2) describes the distribution of the C20 and heavier fraction.(4) This seems to be a mathematical convenience, since the α-value seem ...
Alcohols
... HYDROXIDE ION (OH-) WHICH IS A NEGATIVELY CHARGED PARTICLE FOUND IN BASES. IN BASES, THE HYDROXIDE ION IS HELD TO A POSITIVE ION BY AN A IONIC BOND. • ALCOHOLS HAVE THE GENERAL FORMULA CnH 2n+ 1OH ...
... HYDROXIDE ION (OH-) WHICH IS A NEGATIVELY CHARGED PARTICLE FOUND IN BASES. IN BASES, THE HYDROXIDE ION IS HELD TO A POSITIVE ION BY AN A IONIC BOND. • ALCOHOLS HAVE THE GENERAL FORMULA CnH 2n+ 1OH ...
Chapter 4
... Therefore if a given solution has 90% of one enantiomer (say R) and 10% of the other enantiomer (S) then the enantiomeric excess is 80% ...
... Therefore if a given solution has 90% of one enantiomer (say R) and 10% of the other enantiomer (S) then the enantiomeric excess is 80% ...
Chapter Twenty-One
... For a molecule to be optically active, it must have a carbon atom bonded to four different species. For 2-chlorobutane, the carbon at the 2 position is bonded to a methyl group, a chlorine, a hydrogen, and an ethyl group. ...
... For a molecule to be optically active, it must have a carbon atom bonded to four different species. For 2-chlorobutane, the carbon at the 2 position is bonded to a methyl group, a chlorine, a hydrogen, and an ethyl group. ...
Chemical Equilibrium Equilibrium A state where the reactants and
... If the reaction is multiplied by a factor, ½ N2 + 3/2 H2 < -- > NH3 then, K// = ...
... If the reaction is multiplied by a factor, ½ N2 + 3/2 H2 < -- > NH3 then, K// = ...
Thermodynamics and kinetics of the hydrolysis of atmospherically
... isoprene-derived organonitrates are able to efficiently convert to organosulfates and alcohols on ambient SOA. In order to better understand the structure activity relationships previously observed for the isoprene-derived organonitrates and organosulfates, the hydrolysis reactions of a number of mo ...
... isoprene-derived organonitrates are able to efficiently convert to organosulfates and alcohols on ambient SOA. In order to better understand the structure activity relationships previously observed for the isoprene-derived organonitrates and organosulfates, the hydrolysis reactions of a number of mo ...
carboxylic acids and their derivatives
... the bonding electrons along the chain of atoms between it and the carboxyl proton. This produces a succession of electron shifts along the chain, which, for an electron-attracting substituent, increases the acid strength by making it more energetically feasible for the -OH hydrogen of the carboxyl g ...
... the bonding electrons along the chain of atoms between it and the carboxyl proton. This produces a succession of electron shifts along the chain, which, for an electron-attracting substituent, increases the acid strength by making it more energetically feasible for the -OH hydrogen of the carboxyl g ...
1. What energy changes occur when chemical bonds are formed
... Bond breaking is endothermic and bond making is exothermic. ...
... Bond breaking is endothermic and bond making is exothermic. ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.