Supercritical Degradation of Unsaturated Polyester Resin
... Figure 3 shows the total ion chromatograms obtained after supercritical ethanol treatment and supercritical 1propanol treatment with and without addition of the catalyst R-67-7H. The experiments conducted with ethanol are similar to each other and the experiments conducted with 1-propanol are simila ...
... Figure 3 shows the total ion chromatograms obtained after supercritical ethanol treatment and supercritical 1propanol treatment with and without addition of the catalyst R-67-7H. The experiments conducted with ethanol are similar to each other and the experiments conducted with 1-propanol are simila ...
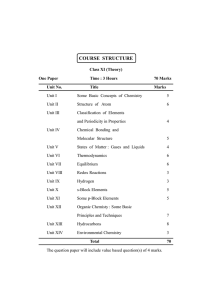
Syllabus - Chemistry
... aldehydes), carbon-carbon double bonds, alcoholic hydroxyl groups, amine groups, carbonyl and carboxyl groups. Unit-II 12 hrs One Group carbon-carbon Disconnections Examples from simple alcohols, amines (aliphatic, aromatic), simple alkenes, carbonyl compounds, acids and acid derivatives. Regioslect ...
... aldehydes), carbon-carbon double bonds, alcoholic hydroxyl groups, amine groups, carbonyl and carboxyl groups. Unit-II 12 hrs One Group carbon-carbon Disconnections Examples from simple alcohols, amines (aliphatic, aromatic), simple alkenes, carbonyl compounds, acids and acid derivatives. Regioslect ...
Word Pro
... 4. Write BALANCED NET IONIC EQUATIONS for the following reactions: (b) CaBr2(aq) + AgNO3(aq) → AgBr(s) + Ca(NO3)2(aq) Br ¯(aq) + Ag+(aq) → AgBr(s) (c) PbS(s) + HNO3(aq) → Pb(NO3)2(aq) + H2S(g) PbS(s) + 2 H+(aq) → Pb2+(aq) + H2S(g) (d) For the reaction between aqueous copper(ll) sulfate and aqueous s ...
... 4. Write BALANCED NET IONIC EQUATIONS for the following reactions: (b) CaBr2(aq) + AgNO3(aq) → AgBr(s) + Ca(NO3)2(aq) Br ¯(aq) + Ag+(aq) → AgBr(s) (c) PbS(s) + HNO3(aq) → Pb(NO3)2(aq) + H2S(g) PbS(s) + 2 H+(aq) → Pb2+(aq) + H2S(g) (d) For the reaction between aqueous copper(ll) sulfate and aqueous s ...
A COMPUTATIONAL STUDY OF CRYSTAL VIOLET
... screen. This is the center carbon atom. b) Add all 3 rings by clicking on Rings (choose Benzene if not displayed) in the builder menu. Click on the 3 bonds of the central carbon, adding a benzene ring to each. c) Go to the Inorganic menu and choose N and then the trigonal planar geometry. Add one ni ...
... screen. This is the center carbon atom. b) Add all 3 rings by clicking on Rings (choose Benzene if not displayed) in the builder menu. Click on the 3 bonds of the central carbon, adding a benzene ring to each. c) Go to the Inorganic menu and choose N and then the trigonal planar geometry. Add one ni ...
4.9 Preparation of Alkyl Halides from Alcohols and Hydrogen Halides
... Primary carbocations are too high in energy to allow SN1 mechanism. Yet, primary alcohols are converted to alkyl halides. Primary alcohols react by a mechanism called ...
... Primary carbocations are too high in energy to allow SN1 mechanism. Yet, primary alcohols are converted to alkyl halides. Primary alcohols react by a mechanism called ...
chemical kinetics type 1.mdi
... Photochemical reactions. Those reactions which take place in the presence of light are called photochemical reactions. Photosynthesis is an example of photochemical reaction. Photosensitization. The process in which a molecule that absorbs light transfers its extra energy to another molecule which m ...
... Photochemical reactions. Those reactions which take place in the presence of light are called photochemical reactions. Photosynthesis is an example of photochemical reaction. Photosensitization. The process in which a molecule that absorbs light transfers its extra energy to another molecule which m ...
carbonyl compounds
... They react with carbonates and the fizzing produced can be used to distinguish carboxylic acids from other organic compounds 2CH3COOH + CaCO3 (CH3COO)2Ca + H2O + CO2 Ethanoic acid calcium ethanoate ...
... They react with carbonates and the fizzing produced can be used to distinguish carboxylic acids from other organic compounds 2CH3COOH + CaCO3 (CH3COO)2Ca + H2O + CO2 Ethanoic acid calcium ethanoate ...
02A naming alkane alkene alkyne 2
... • If there are two or more side groups, the numbering must start with a side group and then proceed in the direction that gives the lowest possible numbers to all the side groups. • If numbers are the same for two or more side groups, the side group that comes first alphabetically is assigned as c1. ...
... • If there are two or more side groups, the numbering must start with a side group and then proceed in the direction that gives the lowest possible numbers to all the side groups. • If numbers are the same for two or more side groups, the side group that comes first alphabetically is assigned as c1. ...
Reaction Energy
... • Some standard enthalpies of formation are given in the appendix of your book. • Each entry in the table is the enthalpy of formation for the synthesis of one mole of the compound from its elements in their standard states. • The thermochemical equation to accompany an enthalpy of formation shows t ...
... • Some standard enthalpies of formation are given in the appendix of your book. • Each entry in the table is the enthalpy of formation for the synthesis of one mole of the compound from its elements in their standard states. • The thermochemical equation to accompany an enthalpy of formation shows t ...
Exam Edge Digital
... be familiar with how overlap of atomic orbitals is used to explain the formation of covalent bonds and be able to distinguish between sigma and pi bonding. You should also know how the number of bonding electron pairs and lone electron pairs influences the shape of covalent molecules. It is essentia ...
... be familiar with how overlap of atomic orbitals is used to explain the formation of covalent bonds and be able to distinguish between sigma and pi bonding. You should also know how the number of bonding electron pairs and lone electron pairs influences the shape of covalent molecules. It is essentia ...
Calculations from Balanced Equations
... 20g of calcium carbonate reacts with excess hydrochloric acid. Calculate (a) the mass of calcium chloride formed. (b) the volume of carbon dioxide gas formed. (Take the molar volume to be 23.0 litre mol-1) Write the balanced equation Show mole ratio ...
... 20g of calcium carbonate reacts with excess hydrochloric acid. Calculate (a) the mass of calcium chloride formed. (b) the volume of carbon dioxide gas formed. (Take the molar volume to be 23.0 litre mol-1) Write the balanced equation Show mole ratio ...
Synthesis and Structure of Alcohols
... Besides –OH groups, other acidic groups like N-H and S-H cannot be tolerated by organometallics. The terminal C-H of alkynes is also sufficiently acidic to be deprotonated by organometallics. These groups are considered incompatible with organometallics. ...
... Besides –OH groups, other acidic groups like N-H and S-H cannot be tolerated by organometallics. The terminal C-H of alkynes is also sufficiently acidic to be deprotonated by organometallics. These groups are considered incompatible with organometallics. ...
2014_S4_CHM_NORMAL (ALL)
... In which of the following pairs would both members have the same electronic configuration? A. ...
... In which of the following pairs would both members have the same electronic configuration? A. ...
CHE 1031 Lab Manual
... Experimental chemistry is inherently dangerous. Many experiments can be hazardous unless the scientist is aware of the nature of the materials being used and uses careful technique, thinking steps through bef ...
... Experimental chemistry is inherently dangerous. Many experiments can be hazardous unless the scientist is aware of the nature of the materials being used and uses careful technique, thinking steps through bef ...
CHEM 494 Lecture 10b - UIC Department of Chemistry
... The man…. laid his hands on him and after a while examined his feet and legs, then pinched his foot hard and asked if he felt it. He said ‘No’; then after that, his thighs; and passing upwards in this way he showed us that he was growing cold and rigid. And then again he touched him and said that ...
... The man…. laid his hands on him and after a while examined his feet and legs, then pinched his foot hard and asked if he felt it. He said ‘No’; then after that, his thighs; and passing upwards in this way he showed us that he was growing cold and rigid. And then again he touched him and said that ...
answer ch6 - Mr Khaled Nasr
... The number of atoms, molecules or ions in one mole of any substance. A unit of quantity that consists of 6.02 x 1023 particles. The mass in grams of one mole of any pure substance. The volumes of gases involved in a reaction and the gases produced exist in fixed ratios. The law which states that equ ...
... The number of atoms, molecules or ions in one mole of any substance. A unit of quantity that consists of 6.02 x 1023 particles. The mass in grams of one mole of any pure substance. The volumes of gases involved in a reaction and the gases produced exist in fixed ratios. The law which states that equ ...
Chemical Equilibrium - Chemistry Teaching Resources
... amounts of CoCl2 (blue) and CoCl2.6H2O (pink) will depend on factors such as temperature of the room, humidity of the air etc. The pieces of paper will still be losing and gaining water molecules:- the reactions are still taking place. The situation is still dynamic, not static. However, the number ...
... amounts of CoCl2 (blue) and CoCl2.6H2O (pink) will depend on factors such as temperature of the room, humidity of the air etc. The pieces of paper will still be losing and gaining water molecules:- the reactions are still taking place. The situation is still dynamic, not static. However, the number ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... In a 250 ml R.B.F. 2-amino-4-(3'-nitro phenyl) thiazole) (2.21 g, 0.01 mole) (1) and 2-chloro-N-(4nitrophenyl)acetamide (2.14 g, 0.01 mole) (2) in 30 ml glacial acetic acid was taken, then slowly K2CO3 (6.9 g, 0.05 mole) was added into reaction mixture. When addition was completed, the reaction mixt ...
... In a 250 ml R.B.F. 2-amino-4-(3'-nitro phenyl) thiazole) (2.21 g, 0.01 mole) (1) and 2-chloro-N-(4nitrophenyl)acetamide (2.14 g, 0.01 mole) (2) in 30 ml glacial acetic acid was taken, then slowly K2CO3 (6.9 g, 0.05 mole) was added into reaction mixture. When addition was completed, the reaction mixt ...
Curriculum Vitae
... “Development of Hydrogen Technologies in Greece”, Greek General Secretariat of Research and Technology, 1998‐2001 ...
... “Development of Hydrogen Technologies in Greece”, Greek General Secretariat of Research and Technology, 1998‐2001 ...
BIOC201W1_Carbohydrate Chemistry_2014
... Hence, sucrose does not undergo reaction characteristic to aldehydes and ketones so it is called a NON-REDUCING ...
... Hence, sucrose does not undergo reaction characteristic to aldehydes and ketones so it is called a NON-REDUCING ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.