Assigning Oxidation Numbers
... reduce the cost of textbook materials for the K-12 market both in the U.S. and worldwide. Using an open-content, web-based collaborative model termed the FlexBook®, CK-12 intends to pioneer the generation and distribution of high-quality educational content that will serve both as core text as well ...
... reduce the cost of textbook materials for the K-12 market both in the U.S. and worldwide. Using an open-content, web-based collaborative model termed the FlexBook®, CK-12 intends to pioneer the generation and distribution of high-quality educational content that will serve both as core text as well ...
Aromatic Compounds Early in the history of organic chemistry (late
... For benzene there are 6 atomic p orbitals in conjugation therefore there will be 6 MO’s ...
... For benzene there are 6 atomic p orbitals in conjugation therefore there will be 6 MO’s ...
AQA GCSE Chemistry My Revision Notes
... When sodium reacts with chlorine, sodium ions and chloride ions are formed. (a) Explain how a sodium atom changes into a sodium ion. (2 marks) (b) Explain how a chlorine atom changes into a chloride ion. (2 marks) Rubidium (Rb) is in the same group of the periodic table as sodium. Rubidium reacts wi ...
... When sodium reacts with chlorine, sodium ions and chloride ions are formed. (a) Explain how a sodium atom changes into a sodium ion. (2 marks) (b) Explain how a chlorine atom changes into a chloride ion. (2 marks) Rubidium (Rb) is in the same group of the periodic table as sodium. Rubidium reacts wi ...
12 - einstein classes
... is broken into small lumps and put into the ammonia convertor, where the Fe3O4 is reduced to give small crystals of iron in a refractory matrix. This is the active catalyst. The actual plant is more complicated than this one-stage reaction implies, since the N2 and H2 must be made before they can be ...
... is broken into small lumps and put into the ammonia convertor, where the Fe3O4 is reduced to give small crystals of iron in a refractory matrix. This is the active catalyst. The actual plant is more complicated than this one-stage reaction implies, since the N2 and H2 must be made before they can be ...
Name: Period:______ Let`s make some sandwiches! Introduction: If
... 4. Notice that the labels changed from molecules to moles. This does not change the mole ratio, as a mole is simply a large number of molecules. How many molecules are in a mole? 5. In Trial #6, what reactant limits you from making more water molecules? Which reactant was present in excess and remai ...
... 4. Notice that the labels changed from molecules to moles. This does not change the mole ratio, as a mole is simply a large number of molecules. How many molecules are in a mole? 5. In Trial #6, what reactant limits you from making more water molecules? Which reactant was present in excess and remai ...
5.1 questions - DrBravoChemistry
... Give the names of the enthalpy changes represented by H1, H2 and H5. H1 ........................................................................................................................... H2 ................................................................................................ ...
... Give the names of the enthalpy changes represented by H1, H2 and H5. H1 ........................................................................................................................... H2 ................................................................................................ ...
elements of chemistry unit
... or groups of atoms) to another. These reactions are called oxidation reduction reactions. REDOX REACTIONS The species that loses electrons is oxidized and the species gaining electrons is reduced. Oxidation reduction reactions are also known as redox reactions (red = reduction, ox = oxidation). OXID ...
... or groups of atoms) to another. These reactions are called oxidation reduction reactions. REDOX REACTIONS The species that loses electrons is oxidized and the species gaining electrons is reduced. Oxidation reduction reactions are also known as redox reactions (red = reduction, ox = oxidation). OXID ...
Lipids I do pdf
... In estrified form In a free form It is 10% -15% of dry weight of brain It is substrate for hormone synthesis ...
... In estrified form In a free form It is 10% -15% of dry weight of brain It is substrate for hormone synthesis ...
Document
... 12. Write the IUPAC name of the compound when 1-butene undergoes the process of hydration. 13. Write the IUPAC name of the compound formed by the reaction between propene and hydrogen iodide. Essay 14. Alkanes are compounds of carbon and hydrogen. a Write the molecular formula of an alkane having fi ...
... 12. Write the IUPAC name of the compound when 1-butene undergoes the process of hydration. 13. Write the IUPAC name of the compound formed by the reaction between propene and hydrogen iodide. Essay 14. Alkanes are compounds of carbon and hydrogen. a Write the molecular formula of an alkane having fi ...
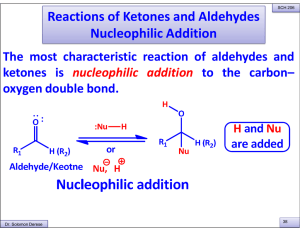
Reactions of Ketones and Aldehydes Nucleophilic Addition
... Like hydrate formation, the synthesis of acetals is reversible, and often the equilibrium favors reactants, not products. In acetal synthesis, however, water is formed as a by-product, so the equilibrium can be driven to the right by removing the water as it is formed by using drying agents or disti ...
... Like hydrate formation, the synthesis of acetals is reversible, and often the equilibrium favors reactants, not products. In acetal synthesis, however, water is formed as a by-product, so the equilibrium can be driven to the right by removing the water as it is formed by using drying agents or disti ...
chemistry notes on the mole - lessons
... and hydrogen peroxide H2O2(l). Both of these compounds contain hydrogen and oxygen. The only difference between the two, is that there is one extra oxygen atom in the hydrogen peroxide. Even though this difference seems very small, it results in significant differences in the properties of both subs ...
... and hydrogen peroxide H2O2(l). Both of these compounds contain hydrogen and oxygen. The only difference between the two, is that there is one extra oxygen atom in the hydrogen peroxide. Even though this difference seems very small, it results in significant differences in the properties of both subs ...
A Few More Notes on Acidity
... Weak inorganic acids (H2S, HCN, NH4+), amine salts (RNH3+), phenols (ArOH), thiols (RSH), aromatic amides (ArCONH2) H2O, alcohols, thiols (RSH), amides RCONH2 ketones (the alpha proton H-CH2COR) Esters (the alpha proton H-CH2CO2R), alkynes RCCH, nitriles (H-CH2CN) ...
... Weak inorganic acids (H2S, HCN, NH4+), amine salts (RNH3+), phenols (ArOH), thiols (RSH), aromatic amides (ArCONH2) H2O, alcohols, thiols (RSH), amides RCONH2 ketones (the alpha proton H-CH2COR) Esters (the alpha proton H-CH2CO2R), alkynes RCCH, nitriles (H-CH2CN) ...
CHAPTER 3 STOICHIOMETRY:
... and (aq) for aqueous to identify the states of matter of the reactants and products. Catalysts, photons (hν), or heat (∆) may stand above the reaction arrow (→). CaCO3(s) + 2HCl(aq) → Ca2+(aq) + 2Cl–(aq) + CO2(g) + H2O(l) ...
... and (aq) for aqueous to identify the states of matter of the reactants and products. Catalysts, photons (hν), or heat (∆) may stand above the reaction arrow (→). CaCO3(s) + 2HCl(aq) → Ca2+(aq) + 2Cl–(aq) + CO2(g) + H2O(l) ...
- Angelo State University
... • In the pyranose form of glucose, carbon-1 is chiral, and thus two stereoisomers are possible: one in which the OH group points down (-hydroxy group) and one in which the OH group points up (hydroxy group). These forms are anomers of each other, and carbon-1 is called the anomeric carbon. ...
... • In the pyranose form of glucose, carbon-1 is chiral, and thus two stereoisomers are possible: one in which the OH group points down (-hydroxy group) and one in which the OH group points up (hydroxy group). These forms are anomers of each other, and carbon-1 is called the anomeric carbon. ...
ism ismismismismismrapidrevisionquestionsismismismismismism
... (i) Combination between nitrogen and hydrogen to form ammonia in the presence of iron catalyst in Haber’s process. Fe N2 + 3H2 2NH3 (ii) Zeolites catalyst ZSM-5 is used to convert alcohol to gasoline by dehydration. (iii) The enzyme zymase converts glucose into ethyl alcohol and carbon dioxide. Zyma ...
... (i) Combination between nitrogen and hydrogen to form ammonia in the presence of iron catalyst in Haber’s process. Fe N2 + 3H2 2NH3 (ii) Zeolites catalyst ZSM-5 is used to convert alcohol to gasoline by dehydration. (iii) The enzyme zymase converts glucose into ethyl alcohol and carbon dioxide. Zyma ...
Reactions of Alcohols and Thiols
... When a secondary alcohol is oxidized, [O], • one H is removed from the –OH. • another H is removed from the carbon bonded to the OH. • a ketone is produced. [O] secondary alcohol ...
... When a secondary alcohol is oxidized, [O], • one H is removed from the –OH. • another H is removed from the carbon bonded to the OH. • a ketone is produced. [O] secondary alcohol ...
98 pts
... • (T) All E1 reactions involve formation of carbocations; • (T) More stable carbocations are generated faster; • (T) Carbocations are electrophiles; • (T) Carbocations are electron deficient; • (T) Free radicals are electron deficient; • (T) Alcohols are Brønsted bases; • (F) The rate-determining st ...
... • (T) All E1 reactions involve formation of carbocations; • (T) More stable carbocations are generated faster; • (T) Carbocations are electrophiles; • (T) Carbocations are electron deficient; • (T) Free radicals are electron deficient; • (T) Alcohols are Brønsted bases; • (F) The rate-determining st ...
1 Q. What are Saturated and unsaturated hydrocarbons?imp
... CYCLOALKANES: General formula is CnH2n (n is a positive whole number for all H/Cs) ...
... CYCLOALKANES: General formula is CnH2n (n is a positive whole number for all H/Cs) ...
Alcohols, Phenols, and Ethers
... (2) The compound ethyl methyl ether contains two alkoxy groups. (3) Rubbing alcohol is a 50-50 mixture by volume of isopropyl alcohol and water. a) All three statements are true. b) Two of the three statements are true. c) Only one of the statements is true. d) None of the statements is true. 14.38 ...
... (2) The compound ethyl methyl ether contains two alkoxy groups. (3) Rubbing alcohol is a 50-50 mixture by volume of isopropyl alcohol and water. a) All three statements are true. b) Two of the three statements are true. c) Only one of the statements is true. d) None of the statements is true. 14.38 ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.