Chemical Reactions and Equations - 2012 Book Archive
... Up to now, we have presented chemical reactions as a topic, but we have not discussed how the products of a chemical reaction can be predicted. Here we will begin our study of certain types of chemical reactions that allow us to predict what the products of the reaction will be. A single-replacement ...
... Up to now, we have presented chemical reactions as a topic, but we have not discussed how the products of a chemical reaction can be predicted. Here we will begin our study of certain types of chemical reactions that allow us to predict what the products of the reaction will be. A single-replacement ...
8 Chemical Equations Chapter Outline Chemical Equations
... Write the balanced chemical equation for the reaction of magnesium hydroxide and phosphoric acid to form magnesium phosphate and water. a. 3 Mg(OH)2 + 2 H3PO4 b. Mg(OH)2 + H3PO4 ...
... Write the balanced chemical equation for the reaction of magnesium hydroxide and phosphoric acid to form magnesium phosphate and water. a. 3 Mg(OH)2 + 2 H3PO4 b. Mg(OH)2 + H3PO4 ...
- Opus: Online Publications Store
... Figure 1: Difference between the installation of a functional group by a typical conventional conversion (left) and by C-H activation and functionalization method (right). Circles in color denote random functional groups. * Indicates less toxic byproduct/s. However, the transformation of a C–H bond ...
... Figure 1: Difference between the installation of a functional group by a typical conventional conversion (left) and by C-H activation and functionalization method (right). Circles in color denote random functional groups. * Indicates less toxic byproduct/s. However, the transformation of a C–H bond ...
Document
... • Problem: draw condensed structural formulas for the two aldehydes with the molecular formula C4H8O. • Solution: • First draw the functional group of an aldehyde and add the remaining three carbons; these may be bonded in two ways. • Then add the seven hydrogens necessary to complete the four bonds ...
... • Problem: draw condensed structural formulas for the two aldehydes with the molecular formula C4H8O. • Solution: • First draw the functional group of an aldehyde and add the remaining three carbons; these may be bonded in two ways. • Then add the seven hydrogens necessary to complete the four bonds ...
CH 8 blackboard
... Follow the solution map, beginning with the actual amount of each reactant given, to calculate the amount of product that can be made from each reactant. Since Cu2O makes the least amount of product, Cu2O is the limiting reactant. The theoretical yield is then the amount of product made by the limit ...
... Follow the solution map, beginning with the actual amount of each reactant given, to calculate the amount of product that can be made from each reactant. Since Cu2O makes the least amount of product, Cu2O is the limiting reactant. The theoretical yield is then the amount of product made by the limit ...
Sol-Gel Science for Ceramic Materials - Sigma
... Technology, Chemistry of Materials, and Journal of Non- Crystalline Solids publish a significant number of articles that focus on silicon-based materials. These organizations, conferences, and journals highlight much of the major siliconbased research generated in academia and industry. Research inv ...
... Technology, Chemistry of Materials, and Journal of Non- Crystalline Solids publish a significant number of articles that focus on silicon-based materials. These organizations, conferences, and journals highlight much of the major siliconbased research generated in academia and industry. Research inv ...
Nickel Catalyzed Conversion of Cyclohexanol into Cyclohexylamine
... reaches a plateau for both Ni/Al2 O3 and Ni/C catalysts (Entries 4 to 6 and 10 to 12). Through the comparison between Entry 3 and 7, Entry 8 and 9, the conversion increased from 48% to 71% and 37% to 87%, respectively, indicating that NaOH can improve the catalytic activity of both Ni/Al2 O3 and Ni/ ...
... reaches a plateau for both Ni/Al2 O3 and Ni/C catalysts (Entries 4 to 6 and 10 to 12). Through the comparison between Entry 3 and 7, Entry 8 and 9, the conversion increased from 48% to 71% and 37% to 87%, respectively, indicating that NaOH can improve the catalytic activity of both Ni/Al2 O3 and Ni/ ...
Ryoji Noyori - Nobel Lecture
... drawn on silk catalyzes asymmetric (heterogeneous) hydrogenation of oximes and oxazolones[13]. This pioneering work, though not effective synthetically, was already well known throughout Japan. In 1968, two years after our asymmetric cyclopropanation in 1966, W. S. Knowles (fellow Nobel laureate in ...
... drawn on silk catalyzes asymmetric (heterogeneous) hydrogenation of oximes and oxazolones[13]. This pioneering work, though not effective synthetically, was already well known throughout Japan. In 1968, two years after our asymmetric cyclopropanation in 1966, W. S. Knowles (fellow Nobel laureate in ...
Energy is the essence of chemistry It determines which reaction can
... • no work done by the surroundings; • no heat transferred from surrounding to system. Example: expansion of a gas at higher pressure P1 into a container at lower pressure P2. The reverse process is non-spontaneous, i.e. surroundings must do work on the gas to increase its pressure from P2 to P1. A s ...
... • no work done by the surroundings; • no heat transferred from surrounding to system. Example: expansion of a gas at higher pressure P1 into a container at lower pressure P2. The reverse process is non-spontaneous, i.e. surroundings must do work on the gas to increase its pressure from P2 to P1. A s ...
chemical kinetics - Berkeley City College
... Step-1: NO2 + NO2 > NO3 + NO; [slow; rate-determining] Step-2: NO3 + CO > CO2 + NO; [fast] The rate law for the rate-determining step: Rate = k1[NO2]2, which is identical in form to the rate law obtained experimentally, in which k1 = k. The second step, which occurs very fast, does not influence ...
... Step-1: NO2 + NO2 > NO3 + NO; [slow; rate-determining] Step-2: NO3 + CO > CO2 + NO; [fast] The rate law for the rate-determining step: Rate = k1[NO2]2, which is identical in form to the rate law obtained experimentally, in which k1 = k. The second step, which occurs very fast, does not influence ...
Biosynthesis of Triglyceride and Other Fatty Acyl
... Palmitoyl CoA: 1,2-diglyceride 0-acyltransferase (EC 2.3.1.20; diglyceride acyltransferase) was assayed by the ['4C]palmitoyl CoA-dependent conversion of endogenous diglyceride to I4C-triglyceride. Assay mixtures in a total volume of 500 pl contained the following: 75 mM phosphate (K+ salt, pH 7.4), ...
... Palmitoyl CoA: 1,2-diglyceride 0-acyltransferase (EC 2.3.1.20; diglyceride acyltransferase) was assayed by the ['4C]palmitoyl CoA-dependent conversion of endogenous diglyceride to I4C-triglyceride. Assay mixtures in a total volume of 500 pl contained the following: 75 mM phosphate (K+ salt, pH 7.4), ...
H2 Chemistry Syllabus (9729)
... recognition of the need for students to develop skills that will be of long term value in an increasingly technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the tea ...
... recognition of the need for students to develop skills that will be of long term value in an increasingly technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the tea ...
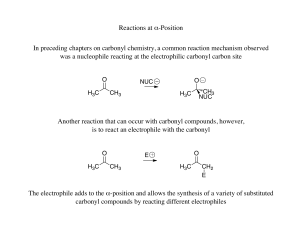
Reactions at α-Position In preceding chapters on carbonyl chemistry
... products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
... products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
Web Appendix 6
... Describe the preparation of 5.000 L of 0.1000 N Na 2CO3 (105.99 g/mol) from the primary-standard solid, assuming that the solution is to be used for titrations in which the reaction is ...
... Describe the preparation of 5.000 L of 0.1000 N Na 2CO3 (105.99 g/mol) from the primary-standard solid, assuming that the solution is to be used for titrations in which the reaction is ...
Practice Exam I FR Answers and Explanations
... 1 point is awarded for correct reactants and 2 points for correct products. All substances that ionize (strong acids, strong bases and soluble salts) must be written in ionic form. All spectator ions must be cancelled. 1 point is awarded for balancing the reaction 1 point is awarded for a correct an ...
... 1 point is awarded for correct reactants and 2 points for correct products. All substances that ionize (strong acids, strong bases and soluble salts) must be written in ionic form. All spectator ions must be cancelled. 1 point is awarded for balancing the reaction 1 point is awarded for a correct an ...
Chemistry Skills Practice Assignments
... 2. How many sublevels would you expect in the 8th energy level? 3. What is the maximum number of electrons that can fit in the 3d sublevel? 4. How many electrons can fit in a 2p orbital? 5. In the 5th energy level, there is a fifth sublevel called the “g sublevel”. Considering the trend in number of ...
... 2. How many sublevels would you expect in the 8th energy level? 3. What is the maximum number of electrons that can fit in the 3d sublevel? 4. How many electrons can fit in a 2p orbital? 5. In the 5th energy level, there is a fifth sublevel called the “g sublevel”. Considering the trend in number of ...
Chapter 4 Solution Chemistry
... negative charge (a partial negative charge, d-) while the H ends have slight positive charges (a partial positive charge, d+). – Water also has an overall bent shape. – The combined effects of polar bonds in a bent shape make water a polar molecule, having an uneven distribution of electrons. ...
... negative charge (a partial negative charge, d-) while the H ends have slight positive charges (a partial positive charge, d+). – Water also has an overall bent shape. – The combined effects of polar bonds in a bent shape make water a polar molecule, having an uneven distribution of electrons. ...
chapter4-bur.2917051..
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
alcohols (2013)
... Reagent/catalyst conc. sulfuric acid (H2SO4) Conditions reflux at 180°C Product alkene Equation ...
... Reagent/catalyst conc. sulfuric acid (H2SO4) Conditions reflux at 180°C Product alkene Equation ...
Honors Chemistry
... Moles of A to Moles of B Work the following out on a separate sheet of paper. 1. Hydrogen and oxygen react under certain conditions to product water. a. How many moles of hydrogen would be needed to produce 5.0 moles of water? b. How many moles of oxygen would be needed to produce 5.0 moles of water ...
... Moles of A to Moles of B Work the following out on a separate sheet of paper. 1. Hydrogen and oxygen react under certain conditions to product water. a. How many moles of hydrogen would be needed to produce 5.0 moles of water? b. How many moles of oxygen would be needed to produce 5.0 moles of water ...
Chapter 7 Hydrosilylation of Carbon
... A transition metal complex, MLn (L=ligand), especially an electron-rich complex of a late transition metal such as Co(I), Rh(I), Ni(0), Pd(0), or Pt(0) as a precatalyst, activates both hydrosilanes, HSiR3, and a variety of substrates, typically alkenes. A catalytic cycle is considered to involve fur ...
... A transition metal complex, MLn (L=ligand), especially an electron-rich complex of a late transition metal such as Co(I), Rh(I), Ni(0), Pd(0), or Pt(0) as a precatalyst, activates both hydrosilanes, HSiR3, and a variety of substrates, typically alkenes. A catalytic cycle is considered to involve fur ...
Unit 10 complete 2016-2017
... Moles of A to Moles of B Work the following out on a separate sheet of paper. 1. Hydrogen and oxygen react under certain conditions to product water. a. How many moles of hydrogen would be needed to produce 5.0 moles of water? b. How many moles of oxygen would be needed to produce 5.0 moles of water ...
... Moles of A to Moles of B Work the following out on a separate sheet of paper. 1. Hydrogen and oxygen react under certain conditions to product water. a. How many moles of hydrogen would be needed to produce 5.0 moles of water? b. How many moles of oxygen would be needed to produce 5.0 moles of water ...
Nikolai N. Semenov - Nobel Lecture
... R. Luther, F. Haber, A.N. Bach, S.Engler, N. A. Shilov, V. Goldschmidt, J. N. Brernsted and others. Among these workers, there were many which drew attention to the significance of often unstable intermediates to the course of chemical reaction. It was roughly about this time that Max Bodenstein sta ...
... R. Luther, F. Haber, A.N. Bach, S.Engler, N. A. Shilov, V. Goldschmidt, J. N. Brernsted and others. Among these workers, there were many which drew attention to the significance of often unstable intermediates to the course of chemical reaction. It was roughly about this time that Max Bodenstein sta ...
Alkenes 3 - ChemWeb (UCC)
... indicate that the groups being eliminated are located on adjacent atoms in the starting material as compared to a 1,1- or -elimination where both are located on the same carbon atom This, in itself, tells you nothing about the actual mechanism of the elimination process. Notice that the requirement ...
... indicate that the groups being eliminated are located on adjacent atoms in the starting material as compared to a 1,1- or -elimination where both are located on the same carbon atom This, in itself, tells you nothing about the actual mechanism of the elimination process. Notice that the requirement ...
Lab # 18
... 7. What is the equation used to convert between degrees Celsius and Kelvin? 8. In which table in your Reference Tables do you find this equation? 9. Based on your experimental procedure, should your graph be connected to the origin (point 0,0). Explain your answer. Introduction: The temperature depe ...
... 7. What is the equation used to convert between degrees Celsius and Kelvin? 8. In which table in your Reference Tables do you find this equation? 9. Based on your experimental procedure, should your graph be connected to the origin (point 0,0). Explain your answer. Introduction: The temperature depe ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.